Biliary atresia is a rare condition which majorly occurs in infants. In this condition, the bile ducts that transport bile from the liver to the gall bladder are blocked. The symptoms of this disease appear about two to eight weeks after birth. Symptoms of the biliary atresia include jaundice, pale colored stools, slow or no weight gain, dark urine, and an enlarged spleen. Once biliary atresia is suspected, a surgery called Kasai portoenterostomy needs to be performed, as early operation is more likely to be successful. If the Kasai operation fails, liver transplantation may be needed.
The global biliary atresia treatment market is estimated to be valued at US$ 910.7 million in 2021 and is expected to exhibit a CAGR of 7.7% during the forecast period (2021-2028).
Figure 1. Global Biliary Atresia Treatment Market Share (%) in Terms of Value, by Disease Type, 2021
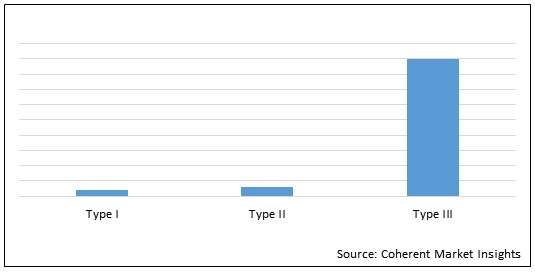
To learn more about this report, Request Free Sample
The increasing prevalence of biliary atresia and its surgery is expected to drive the market growth during the forecast period.
The increasing incidence of biliary atresia and its surgery is expected to drive the market growth during the forecast period. For instance, according to an article published by Research Gate in March 2016, babies born with biliary atresia during 1997 to 2010 were identified from the Taiwan National Health Insurance Research Database. Moreover, among 513 total biliary atresia cases, about 457 (89%) cases underwent the surgery (Kasai procedure).
Biliary Atresia Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 910.7 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 7.7% | 2028 Value Projection: | US$ 1,527.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca plc, Eisai Co., Ltd., Mirum Pharmaceuticals, Inc., Pfizer Inc., Albireo Pharma, Inc., Novartis AG, Alkem Labs, and Glenmark Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2.Global Biliary Atresia Treatment Market Share (%), by Treatment Type, 2021
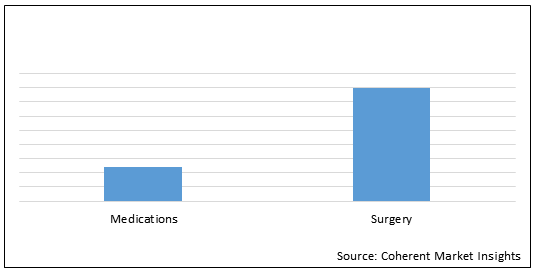
To learn more about this report, Request Free Sample
Increasing advancements in laboratory screening and diagnosis of biliary atresia is expected to drive the market growth during the forecast period.
The increasing advancements in laboratory screening and diagnosis of biliary atresia by researchers and key players are expected to drive the biliary atresia treatment market growth during the forecast period. For instance, according to an article published in the journal ‘Digestive Medical Research’ in September 2021, recent advancements in non-invasive molecular markers, such as gamma-glutamyl transpeptidase (GGT), matrix metalloproteinase-7 (MMP7), interleukin-33 (IL-33), and stool proteomics showed promising results as effective diagnostic markers in biliary atresia.
Global Biliary Atresia Treatment Market– Impact of Coronavirus (COVID-19) Pandemic
Due to the COVID-19 pandemic, pediatric liver research has faced a lot of challenges. The most impacted have been clinical research studies focusing on rare diseases like biliary atresia (BA), which is the most common indication for liver transplantation.
Biliary atresia studies done under the ‘The Childhood Liver Disease Research Network’ (ChiLDReN), an NIH-funded consortium and biorepository, has been largely interrupted during this COVID-19 pandemic, as there has been no new sample collection. Due to the failure to capture new cases of BA, the timeline of research progress will certainly be prolonged. Moreover, the diagnosis of BA has been delayed, as parents are scared of bringing their infants to their pediatrician’s hospital during this period for BA diagnosis.
As several pediatric liver diseases fall under the category of rare diseases, the unstable economic times and greatest impact of this pandemic on funding of pediatric liver research may be seen.
Global Biliary Atresia Treatment Market: Restraint
The major factors that hinder the growth of the global biliary atresia treatment market include safety concerns related to the drug, higher cost involved in the treatment, limited treatment options, and lack of skilled professional in some developing countries like India, Kenya and others.
Key Players
Major players operating in the global biliary atresia treatment market include AstraZeneca plc, Eisai Co., Ltd., Mirum Pharmaceuticals, Inc., Pfizer Inc., Albireo Pharma, Inc., Novartis AG, Alkem Labs, and Glenmark Pharmaceuticals
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients