COVID-19 Detection Kits Market is estimated to be valued at USD 4.74 Bn in 2026 and is expected to reach USD 5.67 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 2.6% from 2026 to 2033.
Analysts’ Views on Global COVID-19 Detection Kits Market:
The COVID-19 Rapid Antigen Self Test is an in vitro diagnostic test intended for qualitative detection of SARS-CoV-2 antigen from human nasal samples. It provides results within 15 minutes, making it a convenient and efficient tool for rapid screening and early detection of COVID-19. The test is designed to be user-friendly, allowing individuals to perform the test themselves in the comfort of their own homes. The COVID-19 Rapid Antigen Self Test kit includes all necessary components and clear instructions for accurate and reliable results. This self-testing option not only reduces the burden on healthcare systems but also promotes early identification of infected individuals, helping to prevent further spread of the virus in communities.
Figure 1. Global COVID-19 Detection Kits Market Share (%), By Product Type, 2026
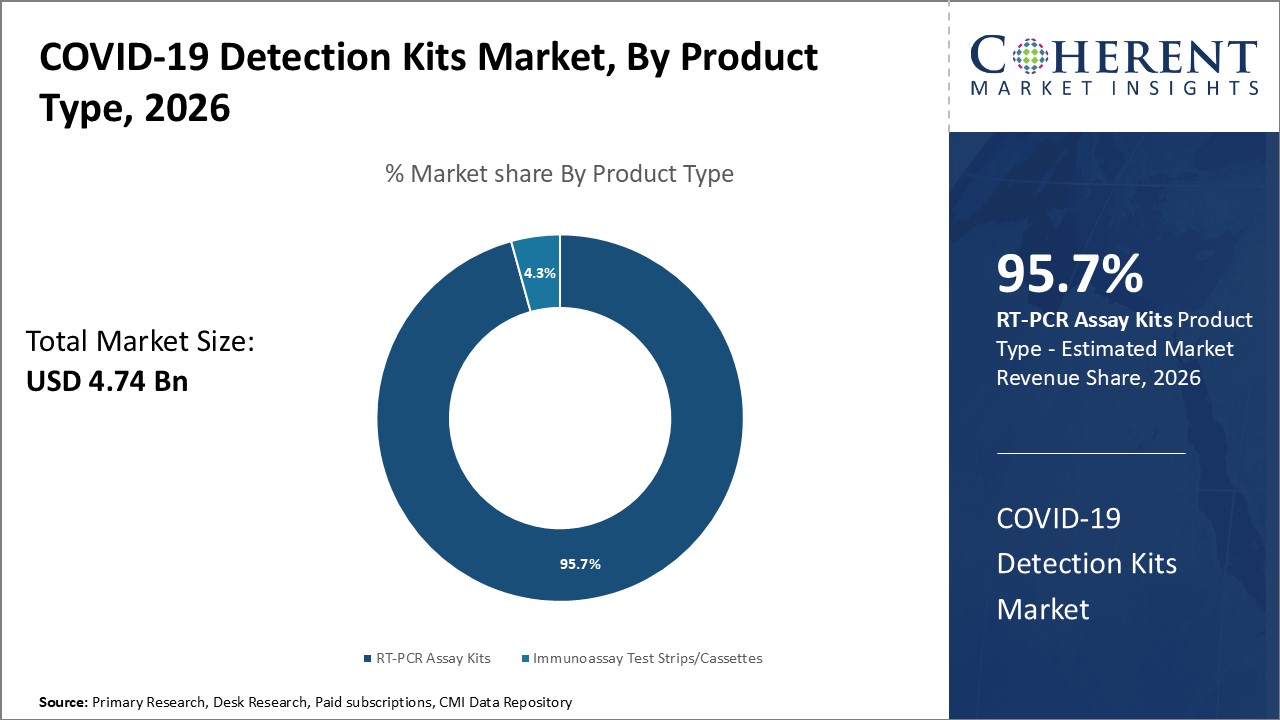
To learn more about this report, Request Free Sample
Figure 1. Global COVID-19 Detection Kits Market Share (%), By Product Region, 2026
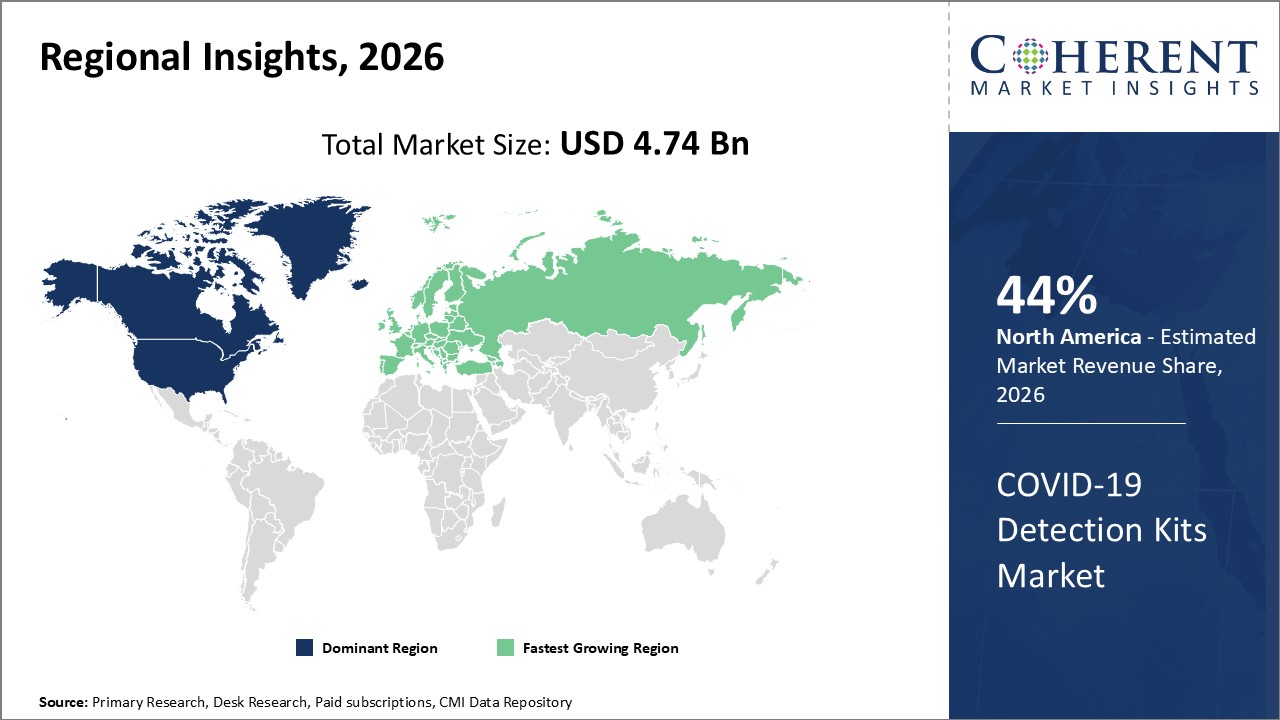
To learn more about this report, Request Free Sample
Global COVID-19 Detection Kits Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global COVID-19 detection kits market over the forecast period. North America is estimated to hold 44% of the market share in 2026. The global COVID-19 detection kits market is expected to witness significant growth in the coming years, due to involvemne of government authority for the market expansion.
For instance, on April 20, 2023, it was reported that approximately one-third of American families used the free COVID-19 at-home test site that the Biden administration developed
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.74 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.6% | 2033 Value Projection: | USD 5.67 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Co-Diagnostics, Inc., F. Hoffmann-La Roche AG, Siemens Healthineers AG, Thermo Fisher Scientific, Abbott Laboratories, BGI, Cepheid Inc., BioFire Diagnostics, GenMark Diagnostics, Qiagen, Quidel Corporation, Randox Laboratories Ltd., SD Biosensor, INC., Shenzhen Bioeasy Biotechnology Co., Ltd., Seegene Inc., JOYSBIO Biotechnology, Cipla Limited, Bione, Virax Biolabs Group Limited, SD BioSensor, Inc., NanoSpot.ai, J. Mitra & Co. Pvt. Ltd., Genes2Me, Ubio Biotechnology Systems Pvt Ltd, Bharat Biotech, Gennova Biopharmaceuticals, QRX Pharma, Angstrom Biotech Pvt. Ltd., and Detect, Inc. |
||
| Growth Drivers: | |||
| Restraints & Challenges: | |||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global COVID-19 Detection Kits Market- Cross Sectional Analysis:
Key players, along with U.S. government research institutes, are making COVID-19 detection kits with advanced technology, which is expected to boost demand for the COVID-19 detection kits market. For instance, on June 25, 2023, Gennova Biopharmaceuticals, a biotechnology company, launched Gemcovac, the country's first indigenously made coronavirus vaccine that uses the mRNA method. The vaccine, branded Gemcovac, was developed with funding from DBT and the Biotechnology Industry Research Assistance Council (BIRAC).
Global COVID-19 Detection Kits Market: Key Developments
In December 2021, QRX Pharma, a clinical-stage specialty pharmaceutical company, in collaboration with Healthians.com, a leading health test at home service offering company, launched rapid testing kits for COVID-19 that can provide instant credible results in 30 minutes, offering much faster turnaround times than the conventional RT-PCR testing kits currently in use.
Global COVID-19 Detection Kits Market: Key Trends
Global COVID-19 Detection Kits Market: Restraint
Therefore, now key players are not focusing on product development and the integration of new technology and methods to avoid the false-result issue.
Global COVID-19 Detection Kits Market - Key Players
Major players operating in the global COVID-19 detection kits market include Co-Diagnostics, Inc., F. Hoffmann-La Roche AG, Siemens Healthineers AG, Thermo Fisher Scientific, Abbott Laboratories, BGI, Cepheid Inc., BioFire Diagnostics, GenMark Diagnostics, Qiagen, Quidel Corporation, Randox Laboratories Ltd., SD Biosensor, INC., Shenzhen Bioeasy Biotechnology Co., Ltd., Seegene Inc., JOYSBIO Biotechnology, Cipla Limited, Bione, Virax Biolabs Group Limited, SD BioSensor, Inc., NanoSpot.ai, J. Mitra & Co. Pvt. Ltd., Genes2Me, Ubio Biotechnology Systems Pvt Ltd, Bharat Biotech, Gennova Biopharmaceuticals, QRX Pharma, Angstrom Biotech Pvt. Ltd., and Detect, Inc.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients