Bone Degeneration Therapeutics Market Size and Forecast – 2026 – 2033
The Global Bone Degeneration Therapeutics Market size is estimated to be valued at USD 7.8 billion in 2026 and is expected to reach USD 13.4 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033. This growth is fueled by the rising prevalence of osteoporosis, an increasing geriatric population, and expanding orthopedic surgery rates worldwide.
Global Bone Degeneration Therapeutics Market Overview
The bone degeneration therapeutics market is experiencing steady growth due to the rising prevalence of osteoporosis, osteoarthritis, and other bone-related disorders, particularly among the aging population. Increased awareness, early diagnosis, and advancements in treatment options, including bone-forming agents, anti-resorptive drugs, and biologics, are driving market expansion. The market benefits from growing orthopedic surgery rates, government healthcare initiatives, and rising healthcare expenditure in both developed and emerging regions. Key players focus on research and development, strategic collaborations, and product innovations to address unmet needs. Overall, the market outlook is positive, with increasing adoption of advanced therapeutics improving patient outcomes globally.
Key Takeaways
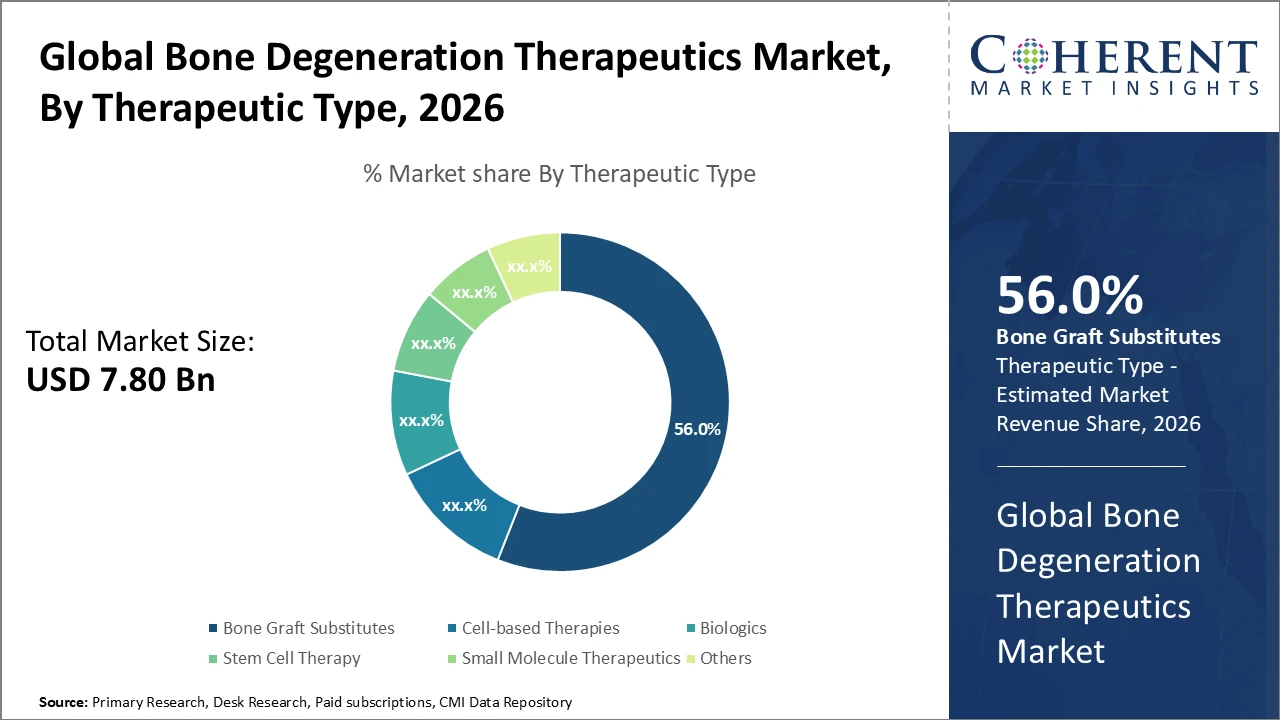
Bone Graft Substitutes dominate the therapeutic type segment with over 56% industry share, driven by wide clinical adoption and proven efficacy.
Osteoporosis is the leading disease type segment, capturing 48% of market revenue due to its high prevalence among aging populations.
Hospitals are the primary end-users, contributing 45% of market share because of advanced infrastructure and high procedural volumes.
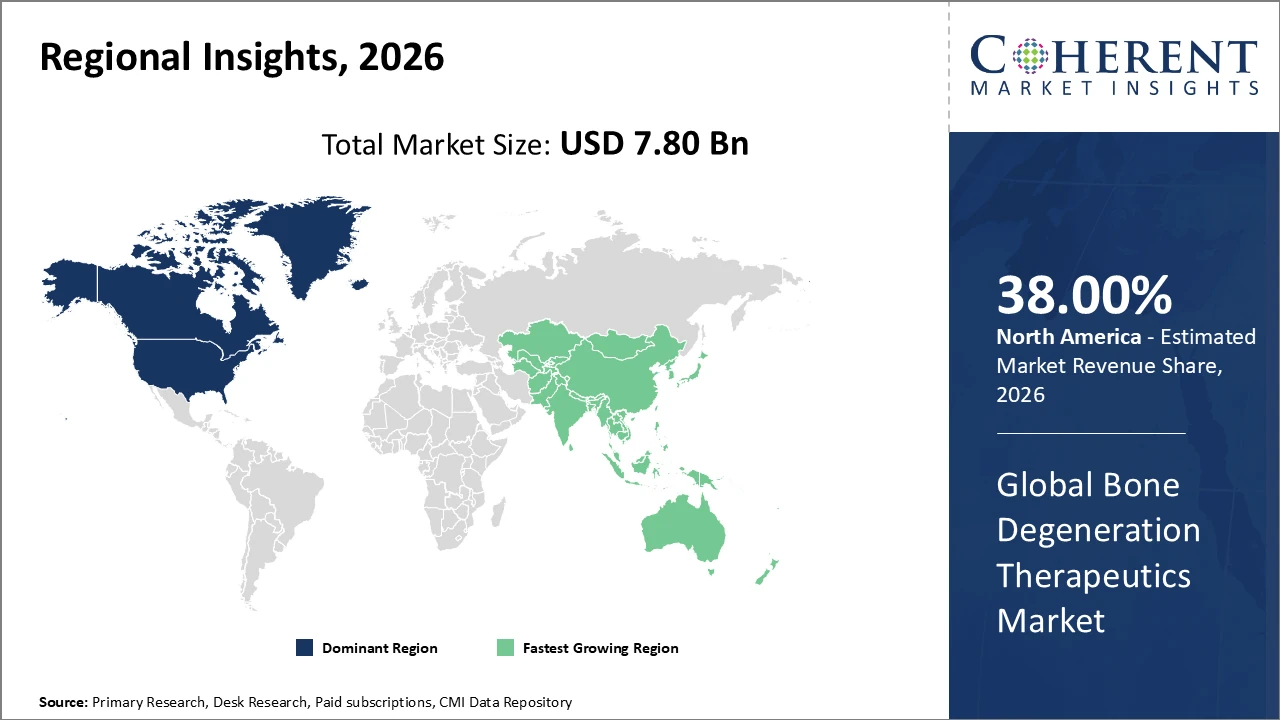
North America holds the largest regional share at nearly 38% of market revenue, supported by strong healthcare infrastructure and significant R&D investments.
Asia Pacific is the fastest-growing region, with a CAGR of approximately 9.5%, driven by rising healthcare spending and large patient populations in China and India.
Bone Degeneration Therapeutics Market Segmentation Analysis
To learn more about this report, Request Free Sample
Bone Degeneration Therapeutics Market Insights, By Therapeutic Type
Bone Graft Substitutes dominate the market with a 56% share, supported by extensive clinical validation, versatile applications in fracture repair and spinal fusion, and cost-effective manufacturing, making them a preferred choice in orthopedic treatments worldwide. Stem Cell Therapy is the fastest-growing subsegment, driven by advances in autologous and allogeneic stem cell technologies that enable personalized regenerative approaches and improved therapeutic outcomes. Biologics, such as recombinant growth factors, occupy a significant niche by promoting natural bone formation, while Cell-based Therapies are increasingly used in complex cases. Small Molecule Therapeutics and other treatments provide adjunctive support and address minor degenerative conditions.
Bone Degeneration Therapeutics Market Insights, By Disease Type
Osteoporosis leads the market with a 48% share, driven by the rising global incidence linked to aging populations and lifestyle factors that increase fracture risk. Osteoarthritis is experiencing rapid growth as the elderly population becomes more prone to joint degradation, driving demand for therapeutics targeting cartilage and subchondral bone repair. Bone fractures remain consistently relevant, particularly with the increase in trauma cases and sports-related injuries, creating a need for innovative treatment solutions. Paget’s Disease and other bone degeneration disorders, while smaller in market size, represent specialized segments addressing rare genetic or metabolic bone conditions that require targeted therapies.
Bone Degeneration Therapeutics Market Insights, By End-User
Hospitals command the largest market share at 45%, driven by their capacity to manage complex orthopedic surgeries and provide post-operative care using advanced bone therapeutics. Specialty Clinics are the fastest-growing subsegment, benefiting from the rise of outpatient procedures and focused orthopedic care that enable faster service delivery and higher patient throughput. Ambulatory Surgical Centers support growth through cost-effective, minimally invasive procedures. Research Institutes contribute by conducting clinical trials and developing novel therapeutics, paving the way for future commercial launches. The Others category, including home care and rehabilitation centers, supports follow-up treatments and helps sustain overall market expansion.
Bone Degeneration Therapeutics Market Trends
The Bone Degeneration Therapeutics market is shifting towards personalized medicine, driven by the integration of genomic data and patient-specific therapeutic formulations.
Mesenchymal stem cell therapies tailored to individual biomarker profiles showed improved clinical outcomes in 2026 trials, highlighting the trend toward precision therapeutics.
Digital health integration, including remote monitoring of bone healing and AI-supported diagnostics, is transforming treatment approaches and accelerating market evolution.
The development of bioengineered 3D printed scaffolds for bone regeneration represents a disruptive technology, addressing limitations of traditional bone graft substitutes and enabling more effective regenerative solutions.
Bone Degeneration Therapeutics Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Bone Degeneration Therapeutics Market Analysis and Trends
North America dominates the bone degeneration therapeutics market with an estimated 38% share, driven by advanced healthcare infrastructure, a high volume of clinical trials, and significant R&D investments. The presence of major industry players, including Medtronic and Zimmer Biomet, further strengthens the region’s market position by introducing innovative therapeutics and expanding treatment accessibility. Strong regulatory support, well-established distribution networks, and a large patient population contribute to sustained revenue growth, making North America the leading region in the global bone degeneration therapeutics market.
Asia Pacific Bone Degeneration Therapeutics Market Analysis and Trends
Asia Pacific is the fastest-growing region in the bone degeneration therapeutics market, registering a CAGR of approximately 9.5%. Growth is fueled by increasing healthcare expenditure, a rising geriatric population, and improving reimbursement frameworks in key markets such as China and India. Government initiatives aimed at enhancing orthopedic healthcare, coupled with public-private partnerships, are further accelerating adoption of advanced therapeutics. Expanding awareness, better healthcare infrastructure, and rising demand for cost-effective bone regeneration solutions position Asia Pacific as a strategic growth hub in the global market.
Bone Degeneration Therapeutics Market Outlook for Key Countries
USA Bone Degeneration Therapeutics Market Analysis and Trends
The U.S. bone degeneration therapeutics market remains a key global leader, supported by advanced healthcare infrastructure and ongoing innovation. In 2025, it accounted for over 30% of global market revenue, driven largely by early adoption of biologics and stem cell therapies for osteoporosis and fracture repair. Leading companies have partnered with research institutions to accelerate clinical trials and launch new products. Additionally, expanded Medicare reimbursement for advanced biologics has improved patient access, further strengthening market growth and revenue generation.
Germany Bone Degeneration Therapeutics Market Analysis and Trends
Germany’s bone degeneration therapeutics market is marked by steady growth supported by a strong healthcare system, aging population, and high clinical demand for advanced orthopedic and regenerative treatments. Bone grafts and substitutes continue to gain traction, with the German market projected to grow at moderate rates driven by innovative materials such as synthetic and biocompatible grafts and personalized solutions. Germany also shows notable expansion in orthobiologics and spine biologics segments, with stem cell and cell‑based matrices registering promising growth. Rising prevalence of osteoporosis and fracture healing needs further bolster demand, alongside investments in research, clinical trials, and collaborations between medical and biotech stakeholders.
Analyst Opinion
The rising demand for minimally invasive bone regeneration techniques is a key supply-side driver, with over 60% of new orthopedic surgeries globally in 2025 incorporating such procedures, boosting adoption of related therapeutics and driving product innovation and market revenue growth.
On the demand side, increasing lifestyle-related bone degeneration cases, including osteoporosis and osteoarthritis, have led to higher imports of bone therapeutics, rising from USD 1.2 billion in 2024 to USD 1.7 billion in 2026 among major healthcare exporting countries, reinforcing market expansion potential.
Micro-indicators such as pricing trends for recombinant growth factors and cellular bone matrices show a steady decline in unit costs by 4-5% annually in 2025-2026, enhancing accessibility and adoption in emerging economies.
Nanotechnology-enabled drug delivery systems for bone tissue regeneration are projected to influence market dynamics by improving therapeutic efficacy and reducing side effects; clinical trials in 2024 using nano-carriers demonstrated a 30% improvement over conventional treatments, highlighting transformative growth strategies.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7.8 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 13.4 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Medtronic plc, Stryker Corporation, NuVasive Inc., Orthofix Medical Inc., Amgen Inc., BioMimetic Therapeutics, Inc., OsteoMed LLC, OseteoScience Foundation, Globus Medical, Inc., RTI Surgical, Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Bone Degeneration Therapeutics Market Growth Factors
The rising incidence of osteoporosis, affecting nearly 200 million people globally in 2025, is a major driver for the bone degeneration therapeutics market. Innovations in biologics and stem cell therapies are providing alternative treatment options to conventional bone grafting, significantly boosting market growth. Favorable reimbursement policies in developed countries, including the U.S. and Germany, have further increased adoption of advanced therapeutics. Additionally, awareness campaigns by organizations such as the International Osteoporosis Foundation have promoted early diagnosis and treatment, directly contributing to higher uptake and revenue growth in the bone degeneration therapeutics market.
Bone Degeneration Therapeutics Market Development
In 2024, the Bone Degeneration Therapeutics market saw notable innovation with several advanced bone graft and substitute technologies entering clinical evaluation and approval phases. For example, Bone Biologics initiated pilot clinical trials for its NB1 bone graft device in spine fusion patients, demonstrating promising early safety and fusion success, while Biogennix received FDA 510(k) clearance to expand its Agilon and Morpheus moldable graft products, enhancing treatment options for degenerative conditions. Additionally, companies like Paradigm BioDevices introduced new bone graft harvesting systems designed to improve surgical efficiency and outcomes.
Another key development in 2024 was the continued expansion of next-generation biologics and regenerative therapies. Market leaders such as Stryker and Medtronic launched advanced biomaterial products, including biodegradable graft substitutes and bioactive scaffolds that enhance bone regeneration and integration in trauma and spinal procedures. These innovations, coupled with increasing adoption of nanostructured and synthetic bone graft materials, reflect a broader industry shift toward more effective, customizable, and minimally invasive treatment approaches.
Key Players
Leading Companies of the Market
Medtronic Plc
Stryker Corporation
NuVasive Inc.
Orthofix Medical Inc.
Amgen Inc.
BioMimetic Therapeutics, Inc.
OsteoMed, LLC
OsteoScience Foundation
Globus Medical Inc.
RTI Surgical, Inc
Several leading companies have recently pursued strategic partnerships and acquisitions to advance biocompatible implant materials. For example, Zimmer Biomet’s 2025 acquisition of an advanced regenerative matrix startup strengthened its bone therapeutics portfolio and increased U.S. market share by 7%. Medtronic has enhanced its market position through product diversification and collaborations with academic institutions, enabling faster time-to-market and generating robust clinical evidence that supports improved patient outcomes. These initiatives reflect a broader industry trend of leveraging innovation and strategic alliances to expand reach and drive growth in the bone degeneration therapeutics market.
Bone Degeneration Therapeutics Market Future Outlook
The bone degeneration therapeutics market is poised for strong growth driven by rising prevalence of osteoporosis, osteoarthritis, and bone fractures, particularly among aging populations. Advances in biologics, stem cell therapies, and regenerative medicine are creating more effective and personalized treatment options. The adoption of minimally invasive surgical techniques, nanotechnology-enabled drug delivery, and bioengineered scaffolds is expected to enhance patient outcomes and broaden market access. Increasing healthcare expenditure, supportive reimbursement policies, and growing awareness of bone health in both developed and emerging regions will further accelerate adoption. Overall, the market outlook is positive, with innovation and accessibility shaping future growth.
Bone Degeneration Therapeutics Market Historical Analysis
The bone degeneration therapeutics market has grown steadily over the past decade, primarily driven by rising prevalence of osteoporosis, osteoarthritis, and age‑related bone disorders. Early market development focused on traditional bone grafts and anti‑resorptive drug therapies, with adoption centered in developed regions due to strong healthcare infrastructure and reimbursement support. As biologics and regenerative technologies emerged, investment in research and development increased, leading to advanced graft substitutes, cell‑based therapies, and bone growth stimulators entering clinical use. Growing awareness of bone health and improvements in diagnostic screening further expanded patient access. Overall, historical trends highlight a shift from conventional treatments toward innovative, efficacy‑driven therapeutics.
Sources
Primary Research Interviews:
Orthopedic surgeons and endocrinologists
Hospital pharmacists and clinical researchers
Biomedical scientists specializing in bone regeneration
Pharmaceutical and medical device company executives
Databases:
World Health Organization (WHO) Global Health Observatory
IQVIA / IMS Health pharmaceutical datasets
OECD Health Data
Magazines:
Pharmaceutical Technology
PharmaTimes
Orthopedic Design & Technology
Biosimilar Development
Journals:
Journal of Bone and Mineral Research
Osteoporosis International
Journal of Orthopaedic Research
Therapeutic Advances in Musculoskeletal Disease
Newspapers:
The New York Times (Health)
The Guardian (Health)
Financial Times (Healthcare)
The Hindu (Health)
Reuters Health
Associations:
World Health Organization (WHO)
International Osteoporosis Foundation (IOF)
European Federation of Pharmaceutical Industries and Associations (EFPIA)
American Society for Bone and Mineral Research (ASBMR)
Orthopaedic Research Society (ORS)
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients