Breast Biopsy Devices Market Size and Forecast – 2026 – 2033
The Global Breast Biopsy Devices Market size is estimated to be valued at USD 1.8 billion in 2026 and is expected to reach USD 3.2 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033.
Global Breast Biopsy Devices Market Overview
The breast biopsy devices market encompasses a range of products designed for accurate diagnosis of breast abnormalities. Core offerings include core needle biopsy devices, which are minimally invasive and widely used for tissue sampling, and vacuum-assisted biopsy systems, providing larger tissue samples with reduced patient discomfort. Stereotactic biopsy devices allow precise imaging-guided procedures, while ultrasound-guided biopsy devices enable real-time monitoring and enhanced accuracy. Emerging technologies such as MRI-guided biopsy systems and automated biopsy guns further improve diagnostic efficiency. Growing awareness of early breast cancer detection and technological innovations are driving demand across hospitals, diagnostic centers, and outpatient clinics globally.
Key Takeaways
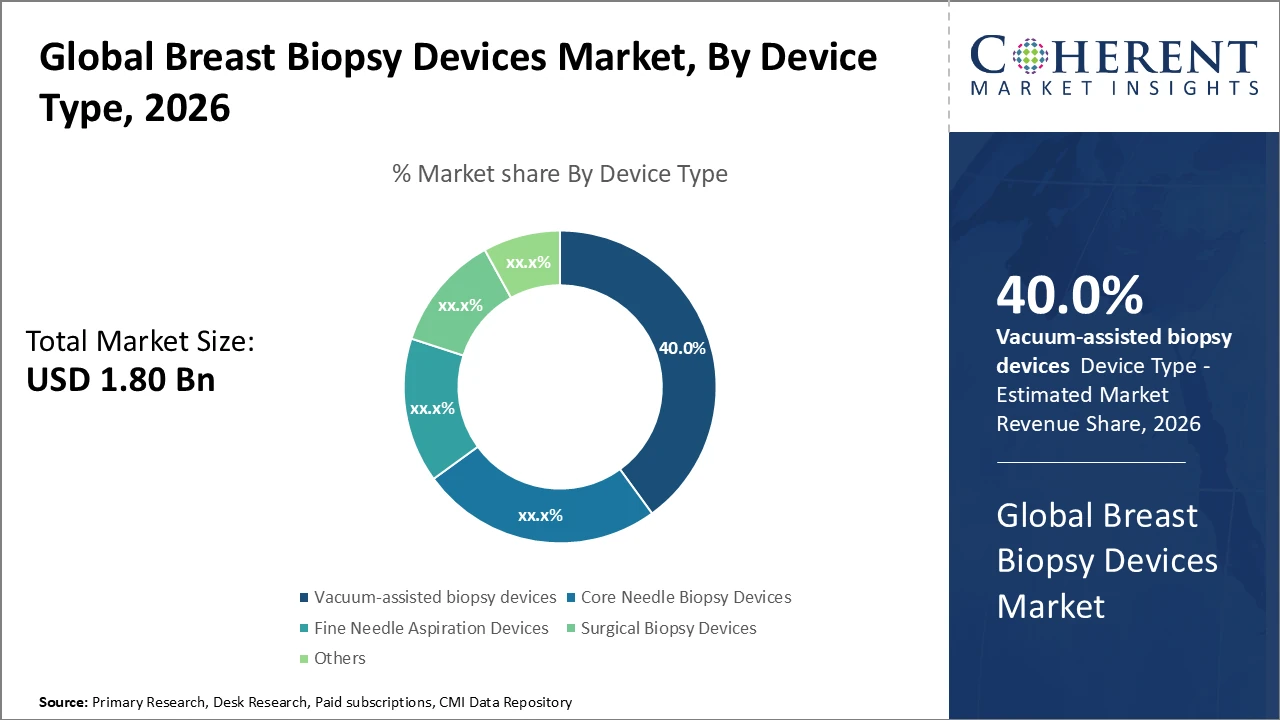
Vacuum-assisted biopsy devices continue to dominate segment share due to superior clinical accuracy and patient comfort, accounting for 40% of market revenue in 2025.
Hospitals represent the largest end-user segment, contributing approximately 55% of market volume due to advanced infrastructure and skilled personnel.
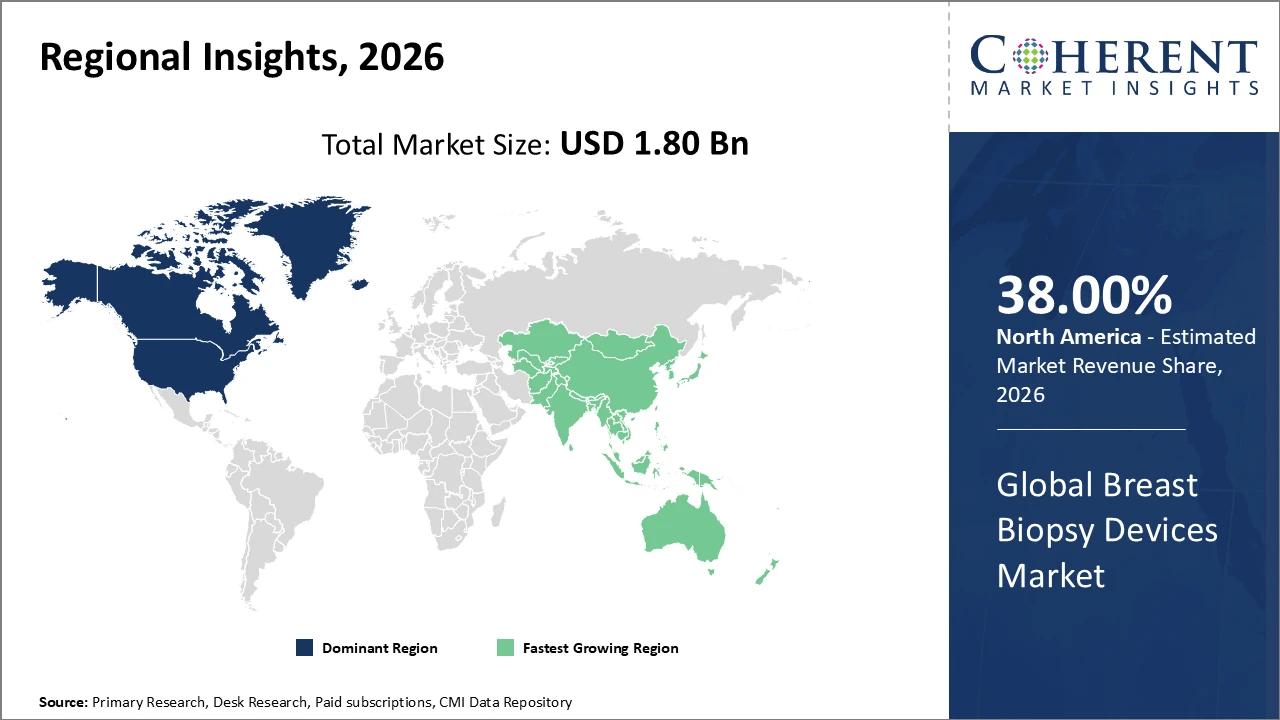
North America leads regional market share at 38% due to high healthcare expenditure and early adoption of innovative devices.
Asia Pacific exhibits the fastest CAGR, attributed to rising healthcare infrastructure investments and increasing breast cancer awareness campaigns.
Europe’s market growth is steady, fueled by reimbursement policies and expanding screening programs in Germany and the U.K.
Breast Biopsy Devices Market Segmentation Analysis
To learn more about this report, Request Free Sample
Breast Biopsy Devices Market Insights, By Device Type
Vacuum-assisted biopsy devices dominate the market due to their enhanced precision and reduced tissue trauma, making them preferred in both diagnostic and interventional radiology settings. The fastest-growing subsegment is core needle biopsy devices, which benefit from technological advancements that improve tissue yield while maintaining a minimally invasive profile. Fine needle aspiration devices are primarily used for preliminary diagnostics, offering limited tissue characterization. Surgical biopsy devices are employed for excisional purposes, particularly in complex diagnostic cases. Other niche devices address specialized clinical needs but account for a minimal share of the overall market.
Breast Biopsy Devices Market Insights, By Technology
Stereotactic-guided biopsy is emerging as the dominating subsegment, favored for its superior precision in targeting calcified lesions. Ultrasound-guided biopsy accounts for the largest volume due to its portability and real-time imaging benefits, driving the fastest growth trajectory. MRI-guided biopsy is expanding among high-risk patient groups that require superior soft tissue contrast, despite higher costs. Mammography-guided biopsy remains in niche use for assessing dense breast tissue. Other guidance modalities include emerging digital tomosynthesis-based approaches, which are gradually gaining attention for enhanced imaging and procedural accuracy.
Breast Biopsy Devices Market Insights, By End-User
Hospitals dominate the market share due to their comprehensive infrastructure and access to advanced imaging, which enable high volumes of biopsy procedures. Ambulatory surgical centers are the fastest-growing subsegment, driven by the trend toward outpatient care and the preference for minimally invasive biopsy services outside hospital settings. Diagnostic centers primarily handle routine screening referrals and perform fewer invasive procedures. Research laboratories contribute a smaller portion of the market, focusing on the development and translational application of new biopsy technologies.
Breast Biopsy Devices Market Trends
The breast biopsy devices market is integrating AI-powered imaging and robotic-assisted biopsy technologies, driving a paradigm shift.
AI algorithms improve target lesion detection, with 2026 breast cancer screening trials showing a 22% increase in diagnostic confidence.
Robotic-assisted systems reduce human error and enhance needle placement precision, leading to faster adoption in specialized oncology centers in North America and Europe.
There is growing focus on eco-friendly and single-use disposable devices to minimize infection risks and simplify sterilization processes.
These technological and sustainable innovations are expanding market scope and shaping future industry trends.
Breast Biopsy Devices Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Breast Biopsy Devices Market Analysis and Trends
In North America, the breast biopsy devices market holds about 38% of the overall market share. This dominance is driven by advanced healthcare infrastructure, early adoption of cutting-edge technologies, and comprehensive insurance coverage that supports diagnostic services. The U.S. plays a key role, with established oncology networks and reimbursement policies favoring minimally invasive biopsy procedures. Major companies such as Hologic and Bard Biopsy Systems maintain a strong presence, promoting innovation and enhancing healthcare delivery models across the region.
Asia Pacific Breast Biopsy Devices Market Analysis and Trends
Meanwhile, Asia Pacific is experiencing the fastest growth, with a CAGR exceeding 10%, driven by expanding healthcare infrastructure, increased breast cancer screening programs, and rising patient awareness. Countries such as China and India play a pivotal role, supported by government-led healthcare reforms and decreasing procedure costs that improve accessibility. Key domestic manufacturers are scaling production, while partnerships with global players facilitate technology transfer, collectively driving robust market revenue growth across the region.
Breast Biopsy Devices Market Outlook for Key Countries
USA Breast Biopsy Devices Market Analysis and Trends
The U.S. market accounts for the majority of North America’s share, contributing over 60% of regional revenue. This is supported by an extensive healthcare network, established reimbursement frameworks, and a strong focus on early breast cancer diagnosis. Leading companies such as Hologic and Bard Biopsy Systems hold significant market shares and continue investing in R&D, highlighted by Hologic’s 2025 launch of its latest vacuum-assisted biopsy platform, which improved tissue retrieval success rates by 20%. Additionally, the growing presence of outpatient and ambulatory surgical centers further supports ongoing market expansion.
Germany Breast Biopsy Devices Market Analysis and Trends
Germany’s breast biopsy devices market is experiencing steady growth, driven by advanced healthcare infrastructure, widespread breast cancer screening programs, and supportive reimbursement policies. Vacuum-assisted and stereotactic-guided biopsy devices are widely adopted due to precision and minimally invasive profiles. Increasing patient awareness and early diagnosis initiatives are boosting demand across hospitals and diagnostic centers. Technological advancements, including AI-assisted imaging and single-use disposable devices, are enhancing procedural accuracy and safety. Collaborative efforts between domestic manufacturers and global players further strengthen market innovation and expansion in Germany.
Analyst Opinion
Increasing adoption of vacuum-assisted biopsy devices has become a key growth driver, accounting for nearly 40% of the breast biopsy devices market share in 2025. This technique improves tissue sampling efficiency and reduces procedure times, with a 15% decrease in biopsy-related complications reported by leading U.S. oncology centers in 2024.
Image-guided biopsy remains central to market expansion, with ultrasound-guided devices capturing over 50% of market revenue in 2026. European hospital data from 2025 show a 12% rise in ultrasound-guided procedures due to non-invasiveness and real-time visualization.
Demand-side dynamics are shifting toward outpatient breast cancer diagnosis, driving an 18% increase in portable and handheld biopsy device adoption in North America in 2026. This growth is supported by healthcare policies promoting early diagnosis, reflected in a 10% increase in community clinics offering localized biopsy services in the U.S.
Supply-side trends indicate enhanced production capacity in Asia Pacific, with manufacturers increasing output by 25% in 2024 to meet domestic and export demand. China and India are emerging as key contributors to market revenue, highlighting the importance of scalable manufacturing.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.8 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 3.2 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Hologic Inc., Bard Biopsy Systems, Cook Medical, Becton Dickinson and Company, Siemens Healthineers, GE Healthcare, Mammotome, Inc., AngioDynamics, Inc., Pfizer Inc., C.R. Bard Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Breast Biopsy Devices Market Growth Factors
Several key factors are driving growth in the breast biopsy devices market. Rising breast cancer prevalence is increasing demand for early-stage diagnosis, with WHO reporting a 7% annual rise in new cases from 2024 to 2026. Technological advancements in minimally invasive procedures, such as stereotactic and MRI-guided biopsies, are improving diagnostic accuracy, reflected by a 15% increase in correct sample yields reported by European hospitals in 2025. Supportive healthcare policies and reimbursement programs in developed economies further encourage adoption of advanced biopsy devices. Additionally, growing demand for portable devices in emerging countries enhances accessibility, fueling market expansion.
Breast Biopsy Devices Market Development
In January 2026, the U.S. Food and Drug Administration granted 510(k) clearance to Becton, Dickinson and Company for its new breast biopsy system. The clearance covers BD’s EnCor EnCompass Breast Biopsy and Tissue Removal System, which is scheduled for commercial launch in early 2026.
Key Players
Leading Companies of the Market
Hologic Inc.
Bard Biopsy Systems
Cook Medical
Becton Dickinson and Company
Siemens Healthineers
GE Healthcare
Mammotome, Inc.
AngioDynamics, Inc.
Pfizer Inc.
C.R. Bard Inc.
Leading companies have implemented aggressive research & development initiatives focusing on enhancing device precision and patient comfort. For example, Hologic’s margin revolution platform demonstrated a 20% improvement in lesion retrieval during 2025 clinical trials, catalyzing market share expansion. Meanwhile, Bard Biopsy Systems leveraged strategic acquisitions in emerging markets like India, strengthening their distribution network and increasing regional market revenue by 13% in 2026.
Breast Biopsy Devices Market Future Outlook
The breast biopsy devices market is poised for strong growth over the coming years, driven by rising breast cancer prevalence, increasing emphasis on early detection, and technological advancements in minimally invasive procedures. AI-assisted imaging, robotic-guided biopsies, and portable handheld devices are expected to enhance diagnostic accuracy and procedural efficiency. Expanding healthcare infrastructure and screening programs in emerging regions, alongside supportive reimbursement policies in developed economies, will further boost adoption. Additionally, innovations in single-use and eco-friendly devices align with safety and sustainability trends. Overall, the market outlook reflects robust expansion, greater accessibility, and continuous technological innovation globally.
Breast Biopsy Devices Market Historical Analysis
The breast biopsy devices market has witnessed steady growth over the past decade, driven by rising breast cancer incidence and increasing awareness of early detection benefits. Initially dominated by surgical biopsy methods, the market gradually shifted toward minimally invasive technologies such as core needle, vacuum-assisted, and stereotactic-guided devices. Technological innovations improved diagnostic accuracy, procedural efficiency, and patient comfort, supporting wider adoption in hospitals and diagnostic centers. North America and Europe led market growth due to advanced healthcare infrastructure, established screening programs, and supportive reimbursement policies. Gradually, emerging regions, including Asia Pacific, began contributing to market expansion as healthcare access and awareness increased.
Sources
Primary Research Interviews:
Oncologists and Radiologists
Clinical Trial Investigators
Breast Surgeons and Pathologists
Medical Device Manufactures
Databases:
World Health Organization (WHO) Cancer and Screening Statistics
NCI Reports
Centers for Disease Control and Prevention (CDC) Breast Cancer Data
Magazines:
Medical Design & Outsourcing
Diagnostic Imaging Magazine
MedTech Outlook
HealthTech Magazine
Medical Device Network
Journals:
Journal of Breast Imaging
Journal of Clinical Oncology
Breast Cancer Research and Treatment
Radiology: Imaging Cancer
Human Vaccines & Immunotherapeutics
Newspapers:
The New York Times (Health)
The Guardian (Health)
Financial Times (Healthcare)
The Hindu (Health)
Reuters Health
Associations:
American Cancer Society (ACS)
European Society of Breast Imaging (EUSOBI)
Society of Breast Imaging
International Society of Radiology (ISR)
National Comprehensive Cancer Network (NCCN)
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients