Global cardiac rhythm management devices market is estimated to be valued at USD 19.05 Bn in 2026 and is expected to reach USD 25.76 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.4% from 2026 to 2033. The market growth is driven by rising global prevalence of cardiac arrhythmias and growing geriatric population worldwide who are more prone to developing such conditions.
To learn more about this report, Request Free Sample
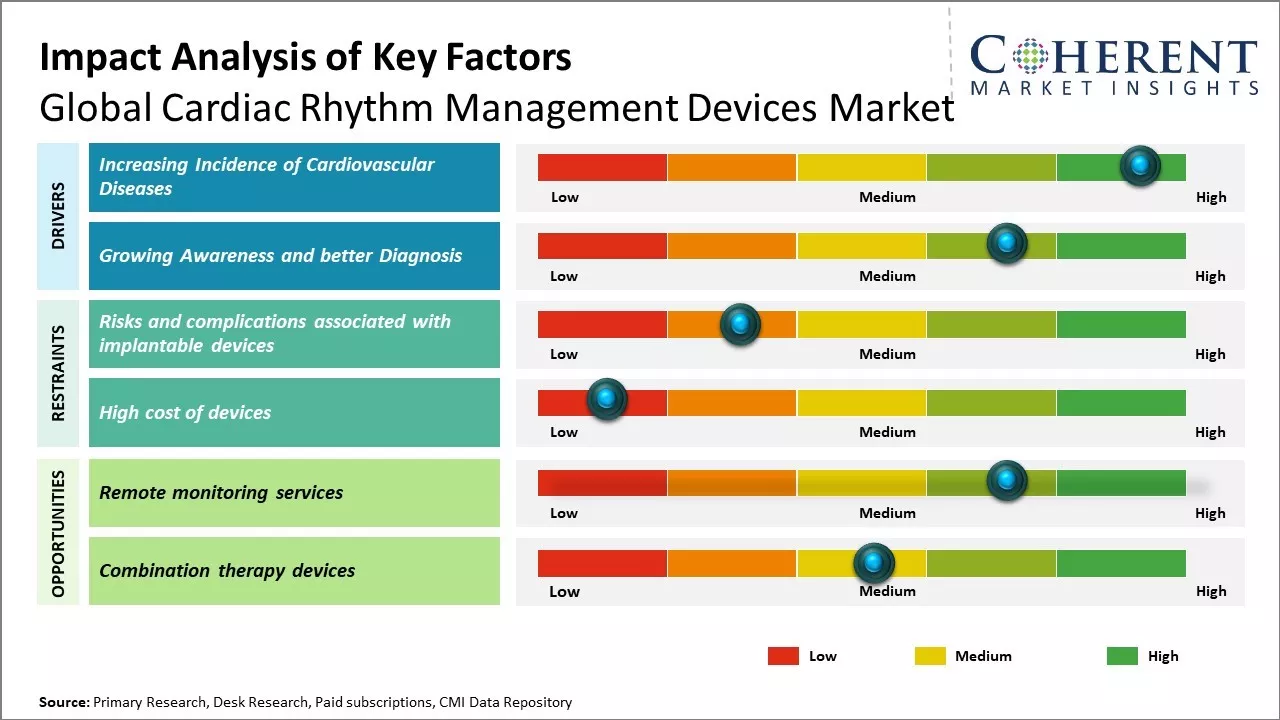
Global cardiac rhythm management devices market growth is driven by factors such as increasing focus on preventive healthcare and growing adoption of technologically advanced devices for treatment. However, risks associated with implantation of cardiac devices and pricing pressures can hamper the market growth.
|
Current Event |
Description and its Impact |
|
Regulatory Environment Changes and Market Access |
|
|
Healthcare Reimbursement Policy Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial intelligence (AI) is transforming the Cardiac Rhythm Management Devices Market by enhancing diagnostic accuracy, personalizing treatment, and enabling real-time monitoring. AI-powered algorithms are now integrated into implantable devices like pacemakers and defibrillators, as well as remote monitoring systems, to detect arrhythmias more precisely and predict adverse cardiac events. This innovation supports earlier intervention, reduces hospital readmissions, and improves patient outcomes. As healthcare systems increasingly adopt AI-driven solutions, the CRM market is witnessing accelerated growth, particularly in regions prioritizing digital health and chronic disease management.
For instance, in September 2025, Ochsner Health in New Orleans introduced AI-powered technology to enhance atrial fibrillation care. The system analyzes heart rhythm data to detect irregularities and support timely intervention. Designed for patients with rapid, erratic heartbeats, this innovation improves diagnosis and treatment planning, advancing cardiac rhythm management in clinical settings across the United States.
In terms of end user, hospitals segment is expected to contribute the highest market share of 36.5% in 2026. These institutions are equipped with the infrastructure and expertise required for complex cardiac procedures, including device implantation, monitoring, and follow-up care. Hospitals also serve as primary centers for emergency cardiac interventions, making them the most frequent users of defibrillators and other rhythm management technologies. Their ability to provide comprehensive care, from diagnosis to rehabilitation—positions them as the preferred setting for both patients and device manufacturers. Moreover, hospitals benefit from economies of scale and access to cutting-edge technologies, which further solidifies their leadership in this market.
For instance, in September 2025, Philips introduced a smart telemetry platform for cardiac monitoring, offering continuous enterprise-wide connectivity beyond the bedside. Designed for hospital environments, the system enhances real-time cardiac data access across departments, improving workflow efficiency and patient safety. This innovation supports proactive cardiac care by enabling clinicians to monitor patients remotely and respond swiftly to rhythm abnormalities.
To learn more about this report, Request Free Sample
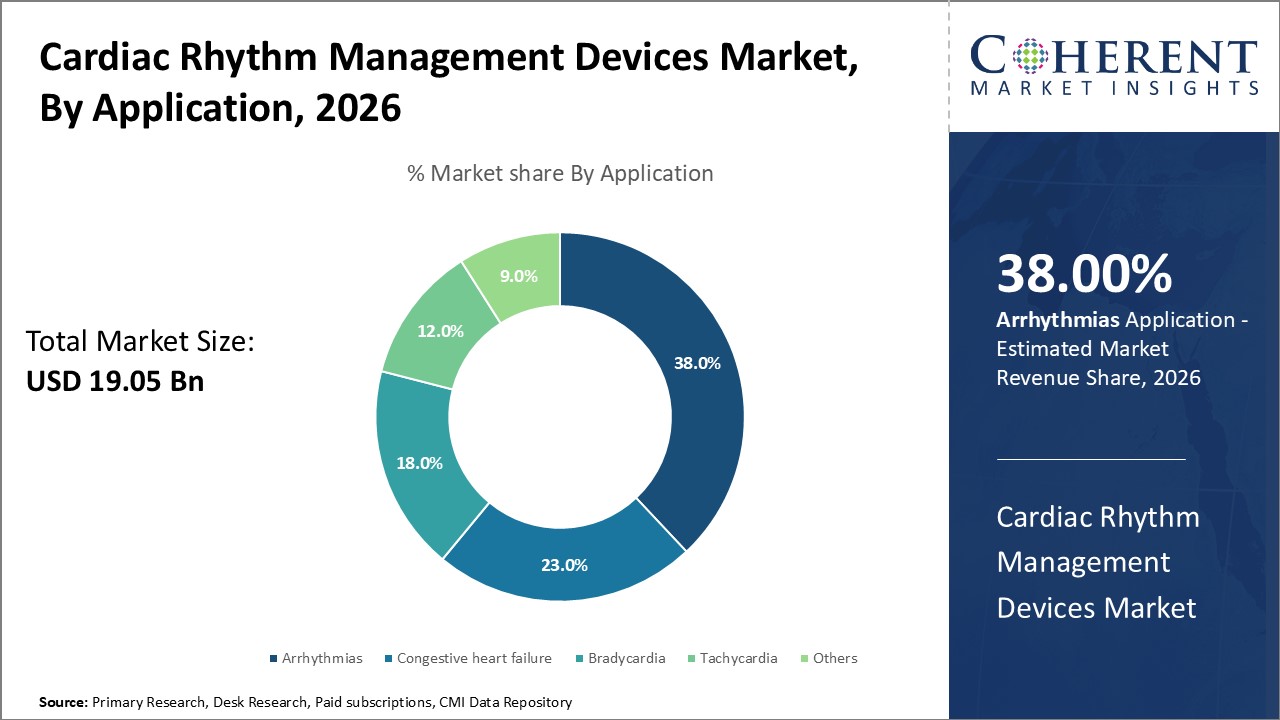
In terms of application, the arrhythmias segment is expected to contribute to the largest market share in 2026. These irregular heart rhythms, which include conditions like atrial fibrillation, bradycardia, and tachycardia, affect millions globally and are a major cause of morbidity and mortality. The rising prevalence of arrhythmias, especially among aging populations, has fueled demand for devices such as pacemakers, defibrillators, and cardiac resynchronization therapy (CRT) systems. Innovations in arrhythmia detection, such as wearable ECG monitors and AI-powered diagnostics have improved early diagnosis and treatment, further expanding this segment. As arrhythmias often require long-term management, the need for reliable and responsive rhythm control devices continues to grow.
For instance, in February 2024, Signify Health launched a new heart arrhythmia testing service, expanding its in-home diagnostic and preventive care offerings. This initiative enhances early detection of cardiac rhythm disorders through remote monitoring, aiming to improve patient outcomes and reduce hospital visits. The service reflects Signify Health’s commitment to accessible, tech-enabled healthcare solutions for chronic condition management.
To learn more about this report, Request Free Sample
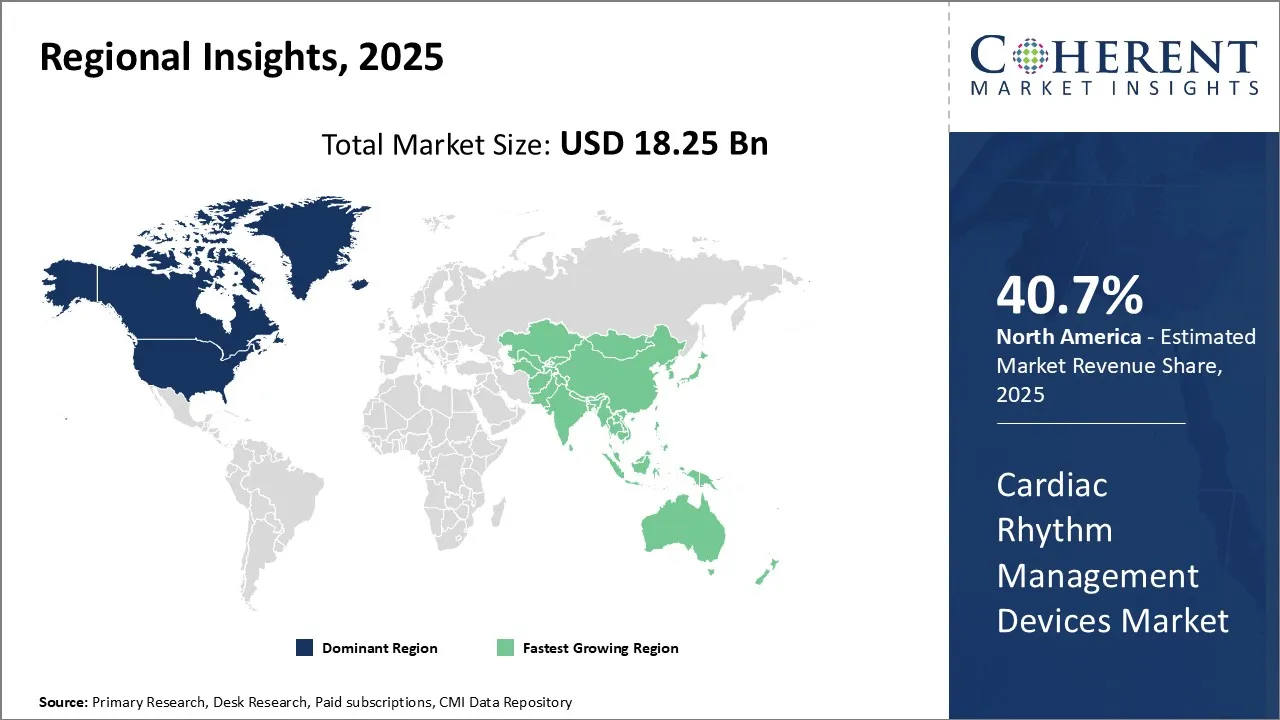
North America dominates the global cardiac rhythm management devices market with an estimated market share of 40.7% in 2026, due to highly developed healthcare infrastructure, rising prevalence of cardiovascular diseases, and growing per capita healthcare expenditure. Presence of leading global players in the region coupled with high adoption of technologically advanced devices drives the market growth. Furthermore, favorable reimbursement policies for implantable devices and rising awareness about available treatment options also drives the market growth.
For instance, in May 2026, Johnson & Johnson MedTech launched the SoundStar® eco ICE Catheter with Crystal Clear™ imaging in the U.S., enhancing 2D intracardiac echocardiography. Designed for electrophysiology procedures, the catheter delivers superior image clarity and supports real-time cardiac visualization, improving precision and safety during arrhythmia treatments and other cardiac interventions in hospital settings.
Asia Pacific is expected to be the fastest growing market for cardiac rhythm management devices. Rapidly improving healthcare infrastructure, increasing healthcare spending, and expanding private insurance coverage drives the market growth in the Asia Pacific region. Moreover, growing geriatric population suffering from heart conditions and improving economic conditions attracts leading global players to focus on developing markets within Asia Pacific. Countries like China, Japan and India offer immense opportunities due to their huge population bases and growing medical tourism industries. Rising standards of living and increasing disposable incomes have also boosts demand for quality healthcare and premium products in the Asia Pacific region. The region is witnessing growing exports of advanced devices from developed markets like the U.S. and Japan while domestic manufacturers are gaining momentum to cater to domestic needs. Favorable regulations and initiatives by governments aiming to reduce heart disease burden can drive the Asia Pacific market growth during the forecast period.
For instance, in July 2026, Johnson & Johnson MedTech launched the Varipulse™ Platform across the Asia-Pacific region, advancing atrial fibrillation treatment with pulsed field ablation technology. The platform includes a PFA catheter, mapping catheter, and integrated imaging system, offering a minimally invasive solution for arrhythmia management. This expansion supports improved cardiac care in hospitals and electrophysiology centers throughout the region.
In 2026, the U.S. Cardiac Rhythm Management Devices Market is thriving due to rising cardiovascular disease rates, an aging population, and rapid adoption of advanced technologies like implantable defibrillators and remote monitoring systems. Strong hospital infrastructure, favorable reimbursement policies, and growing demand for home-based cardiac care further fuel the nation's leadership in rhythm management innovation.
For instance, in May 2023, Biotronik unveiled the BIOMONITOR IV Implantable Cardiac Monitor at the Heart Rhythm Society Congress 2023 in the U.S. Developed with artificial intelligence, the device enhances arrhythmia detection and long-term cardiac monitoring. Its advanced algorithms and streamlined design support early diagnosis and improved rhythm management, marking a significant step forward in AI-powered cardiac care.
In 2026, China’s demand for Cardiac Rhythm Management Devices is driven by a rapidly aging population, rising incidence of arrhythmias, and expanding access to advanced cardiac care. Government healthcare reforms, local manufacturing initiatives, and increased adoption of pacemakers and defibrillators are fueling market growth, making China a key player in global rhythm management innovation.
For instance, in March 2026, MicroPort CRM launched its localized TEN Pacemaker family in China, marking a milestone in domestic cardiac care innovation. Introduced in Shanghai in March 2026, the devices are tailored to meet regional clinical needs, offering advanced rhythm management solutions. This move supports China's growing demand for accessible, high-quality cardiac rhythm management technologies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 19.05 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.4% | 2033 Value Projection: | USD 25.76 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
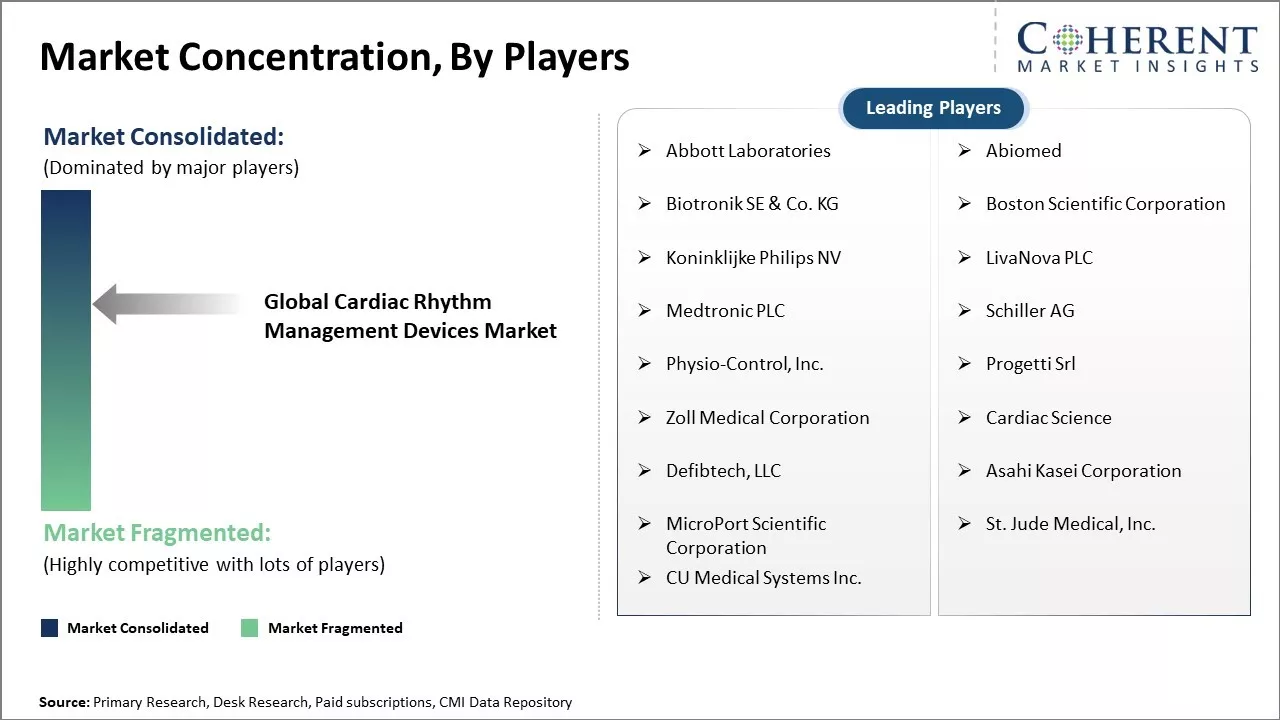
Abbott Laboratories, Abiomed, Biotronik SE & Co. KG, Boston Scientific Corporation, Koninklijke Philips NV, LivaNova PLC, Medtronic PLC, Schiller AG, Physio-Control, Inc., Progetti Srl, Zoll Medical Corporation, Cardiac Science, Defibtech, LLC, Asahi Kasei Corporation, MicroPort Scientific Corporation, St. Jude Medical, Inc., and CU Medical Systems Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence of cardiovascular diseases can drive the market growth. According to the World Health Organization, cardiovascular diseases currently account for over 17 million deaths annually, with low- and middle-income countries disproportionately impacted. Rising burden of cardiovascular diseases can be attributed to both genetic predisposing factors as well as lifestyle and environmental risk factors prevalent in modern societies. Changes in dietary patterns along with reduced physical activity contribute to increased incidence of obesity, diabetes and unhealthy cholesterol levels, all of which are known risk multipliers for developing cardiovascular conditions. Moreover, growing geriatric population can also drive the market growth as age is a significant risk factor of the disease due to deteriorating functionality of organs with time.
To learn more about this report, Request Free Sample
With greater knowledge regarding symptoms and risk factors of heart diseases, more people are becoming aware of underlying conditions in the early stages. Advancement in diagnostic technologies has enabled identification of cardiac arrhythmias and irregular heartbeats. Wearable medical devices and mobile ECG ,Electrocardiogram, recorders makes cardiac screening more convenient and accessible. Physicians can more accurately diagnose previously unknown cases of abnormal heart rhythms, valve defects or heart failures even in asymptomatic individuals. This has resulted in more patients being directed to specialized cardiac rhythm management procedures and therapies. Growing middle-class population in developing nations now has more access to sophisticated healthcare technologies and interventions. Improved medical insurance coverage has also removed financial barriers for many people, allowing for timely management according to recommended clinical guidelines.
Remote monitoring services can offer growth opportunities for global cardiac rhythm management devices market. With advanced technologies like Bluetooth and wireless connectivity, remote monitoring allows continuous surveillance of patients' heart conditions from outside the hospital setting. This offers huge benefits for patients as well as the healthcare system. Remote monitoring allows early detection of any abnormality or deterioration in patients' heart conditions. This helps intervening promptly and preventing emergency situations. It ensures patients experience less hospital visits and stay comfortable at home while getting periodic reviews of their cardiac issues. This improves their quality of life significantly. For healthcare providers, remote monitoring helps manage large patient loads efficiently with limited resources. It reduces burden on hospitals with less readmissions.
The Cardiac Rhythm Management (CRM) Devices Market is transitioning from hardware-driven competition to ecosystem-based differentiation, where device longevity, data intelligence, and clinical workflow integration determine leadership. Leadless pacemakers have matured rapidly, supported by over 300,000 global implants and long-term complication rates below 5%, making them the preferred choice for patients at risk from transvenous leads. Battery life improvements—now exceeding 15 years in next-generation systems are reshaping procurement priorities toward lifetime cost efficiency and reduced revision surgeries.
Subcutaneous ICDs (S-ICDs), led by Boston Scientific’s EMBLEM with an estimated 38–65% market share, address infection-prone and younger patients, though their lack of pacing limits full substitution of transvenous systems. Meanwhile, transvenous ICDs and CRT devices continue to dominate in complex heart-failure and resynchronization cases, aided by incremental gains in miniaturization and algorithmic intelligence.
The most transformative frontier is remote monitoring (RM): a dataset of 32,000 patients generated over 380,000 transmissions, underscoring the need for triage-level analytics to prevent clinician fatigue. Vendors integrating validated AI-driven filtering and secure connectivity will capture superior value. Overall, competitive success now depends on lifecycle economics, interoperable data ecosystems, and phenotype-driven therapy personalization—factors redefining strategic positioning across the CRM landscape.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients