Chemotherapy Induced Thrombocytopenia Therapeutics Market is estimated to be valued at USD 1,775.8 Mn in 2026 and is expected to reach USD 2,532.2 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.2% from 2026 to 2033.
Analysts’ Views on the Global Chemotherapy-induced Thrombocytopenia Therapeutics Market:
Increasing advancements in chemotherapy and cancer therapies may drive the use of chemotherapy and, subsequently, the demand for thrombocytopenia therapeutics to fuel the global chemotherapy-induced thrombocytopenia therapeutics market growth over the forecast period.
Figure 1. Global Chemotherapy-induced Thrombocytopenia Therapeutics Market Share (%), By Drug Class, 2026
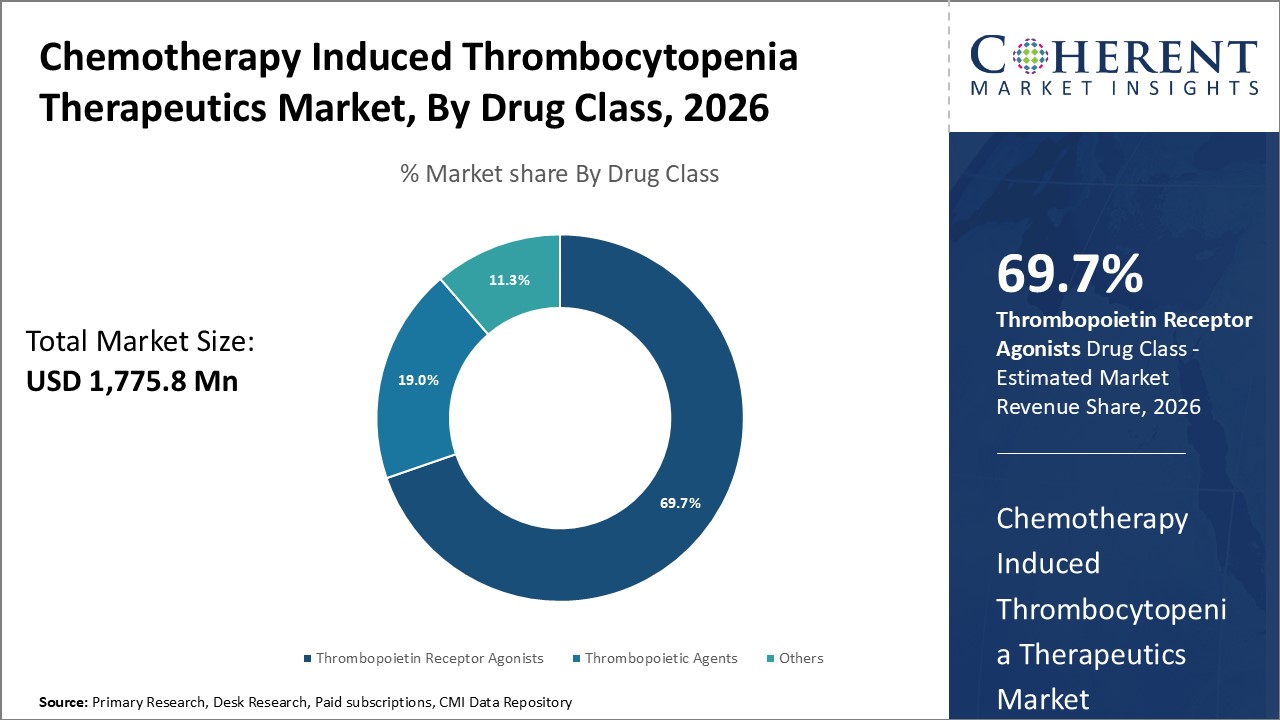
To learn more about this report, Request Free Sample
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market - Drivers
Figure 2. Global Chemotherapy-induced Thrombocytopenia Therapeutics Market Share (%), By Region, 2026
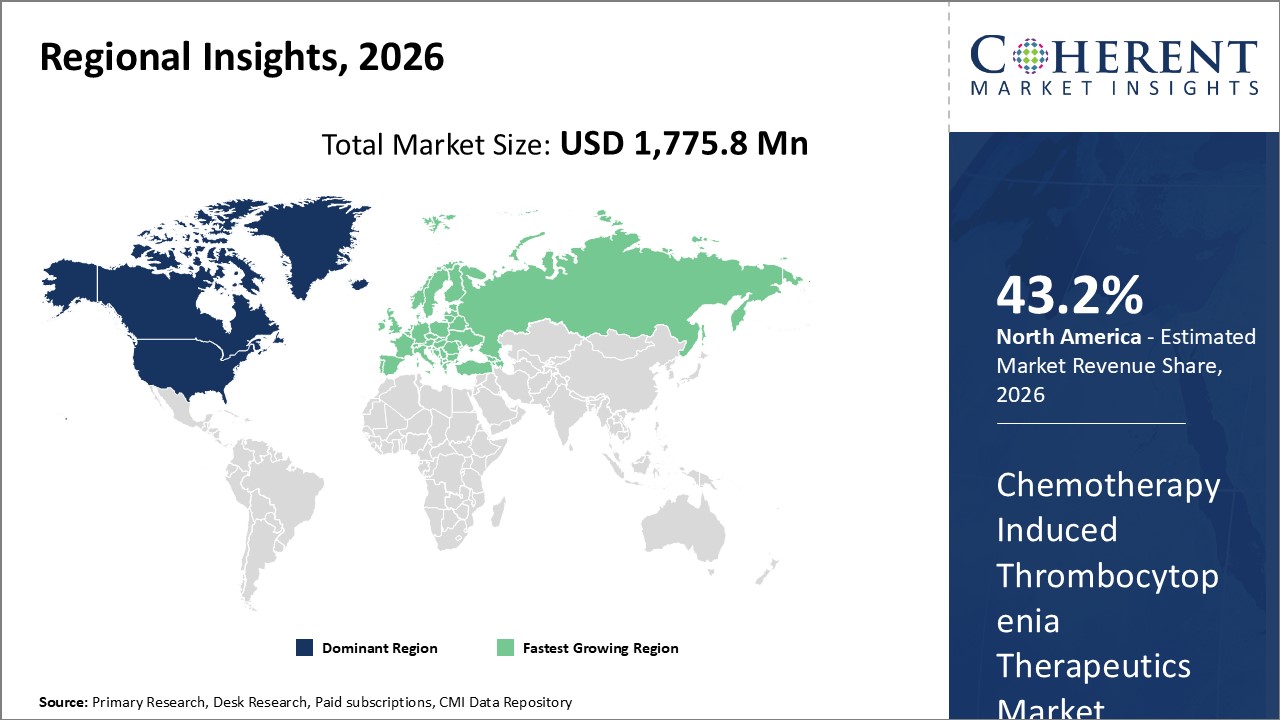
To learn more about this report, Request Free Sample
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global chemotherapy-induced thrombocytopenia therapeutics market over the forecast period. North America is estimated to hold 43.2% of the market share in 2026. The market is expected to witness significant growth in the near future due to rising cancer initiatives in the region. For instance, in June 2023, the U.S. government provided US$ 50 million to launch the Persistent Poverty Initiative, which aims to reduce the cumulative effects of persistent poverty on cancer outcomes by increasing research capacity, encouraging cancer prevention research, and encouraging the implementation of community-based programs. The Persistent Poverty Initiative is the first significant program to address persistent poverty's structural and institutional determinants in the context of cancer.
Chemotherapy Induced Thrombocytopenia Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,775.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 2,532.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Amgen, Inc., Novartis AG, Pfizer Inc., Swedish Orphan Biovitrum AB, Jiangsu HengRui Medicine Co., Ltd., Teva Pharmaceutical Industries Ltd., and Mylan N.V. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market - Segmentation
The global chemotherapy-induced thrombocytopenia therapeutics market report is segmented into drug class, route of administration, distribution channel, and region.
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market - Cross Sectional Analysis
Among the distribution channel segment, the hospital pharmacies segment is expected to be dominant in the Asia Pacific region owing to the increasing launch of hospital pharmacies. For instance, on September 26, 2023, the Lilavati Hospital, a private hospital in Mumbai, India, launched a chain of Lilavati Pharmacy outlets around India. The Lilavati Pharmacy chain will also launch a community wellness programs. Customers will be able to consult with a doctor at the drugstore and receive proper treatment.
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market: Key Developments
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market: Key Trends
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market: Restraint
To counterbalance the restraint, key players should focus on increasing research and development activities of therapeutics.
Global Chemotherapy-induced Thrombocytopenia Therapeutics Market - Key Players
The major players operating in the global chemotherapy-induced thrombocytopenia therapeutics market include Amgen, Inc., Novartis AG, Pfizer Inc., Swedish Orphan Biovitrum AB, Jiangsu HengRui Medicine Co., Ltd., Teva Pharmaceutical Industries Ltd., and Mylan N.V.
Definition: Chemotherapy-induced thrombocytopenia (CIT) is a medical disorder caused by chemotherapy that results in a considerable decrease in the number of platelets in the blood. Platelets are little blood cells that aid in blood clotting and the prevention of excessive bleeding. When the platelet count falls to excessively low levels as a result of chemotherapy, there is an increased risk of bleeding and bruising. Chemotherapy is a frequent cancer treatment, but it can have negative effects on bone marrow, which produces blood cells such as platelets. Chemotherapy drugs can impair the bone marrow's capacity to produce sufficient platelets, resulting in thrombocytopenia.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients