Global Clinical Data Management Market Overview
The process of collection, documentation, and data storage, which are critical for clinical trials or procedures and to biotechnology companies and pharmaceutical manufacturers is termed as Clinical Data Management (CDM). Clinical data management tool is employed in clinical research for managing the data of a clinical trial. Data management is a process of collection, integration, and management of subject data in compliance with regulatory standards. Clinical data management is a fundamental phase in clinical research leading to the generation of high-quality, reliable, and statistically comprehensive data to reduce time duration from drug development to marketing. The clinical data is captured in a paper or electronic data capture format. Pharma companies, contract research organizations, and IT companies are involved in CDM activities. Data is collected from multiple sources and stored in a patient-centric fashion in a data repository commonly known as clinical data repository.
The major goal of CDM is to assure that the data support deductions drawn from the research to protect public health and trust in marketed therapeutics. Outsourcing of clinical data management to IT service providers is done by the organizations to focus on core competencies of clinical research and pharmaceuticals, reduction in operational expenditure, and investment in research and development activities.
Clinical Data Management Market Size and Forecast – 2026 – 2033
The Clinical Data Management Market size is estimated to be valued at USD 3.91 billion in 2026 and is expected to reach USD 8.54 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 11.8% from 2026 to 2033.
Key Takeaways
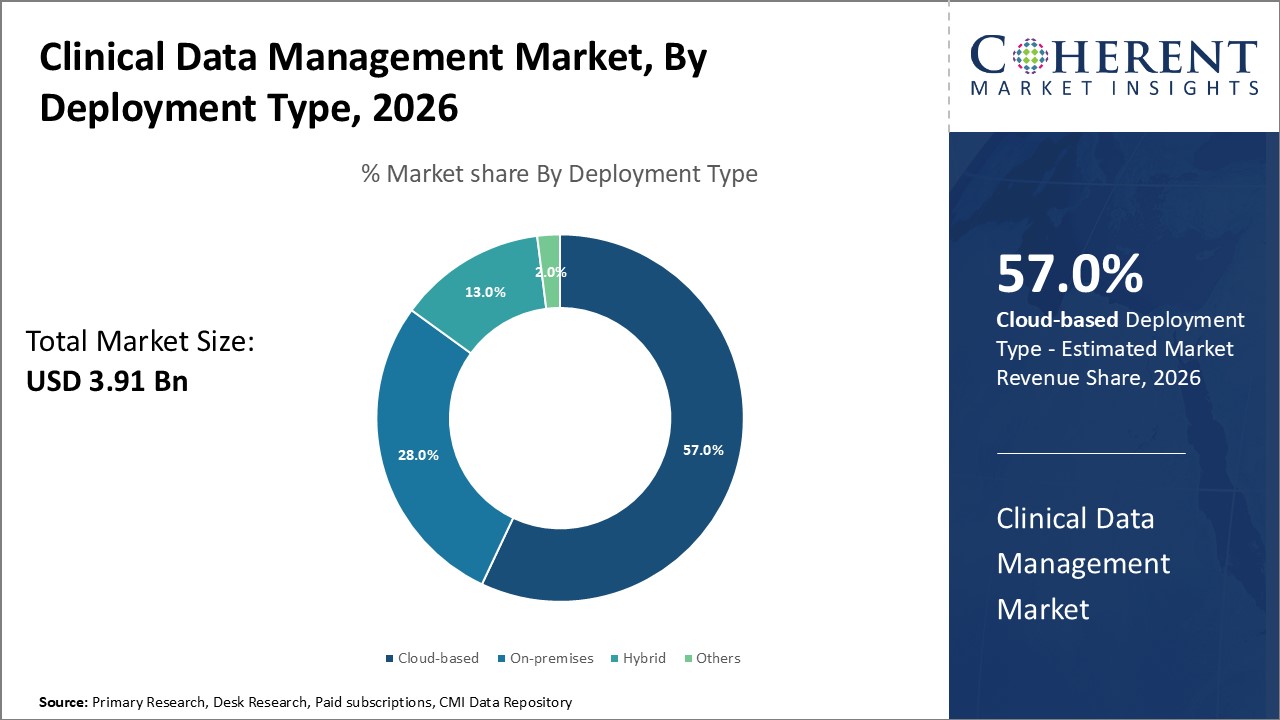
Cloud-based deployment leads with 57% market share, supported by its scalability and integration with AI. Hybrid models are gaining traction in pharmaceutical companies due to flexible data control options.
Oncology remains the dominant therapeutic area segment, commanding approximately 36% share, driven by increasing R&D investments. Neurology is the fastest-growing sub segment, with a 14% growth rate accelerated by rising prevalence of neurodegenerative diseases.
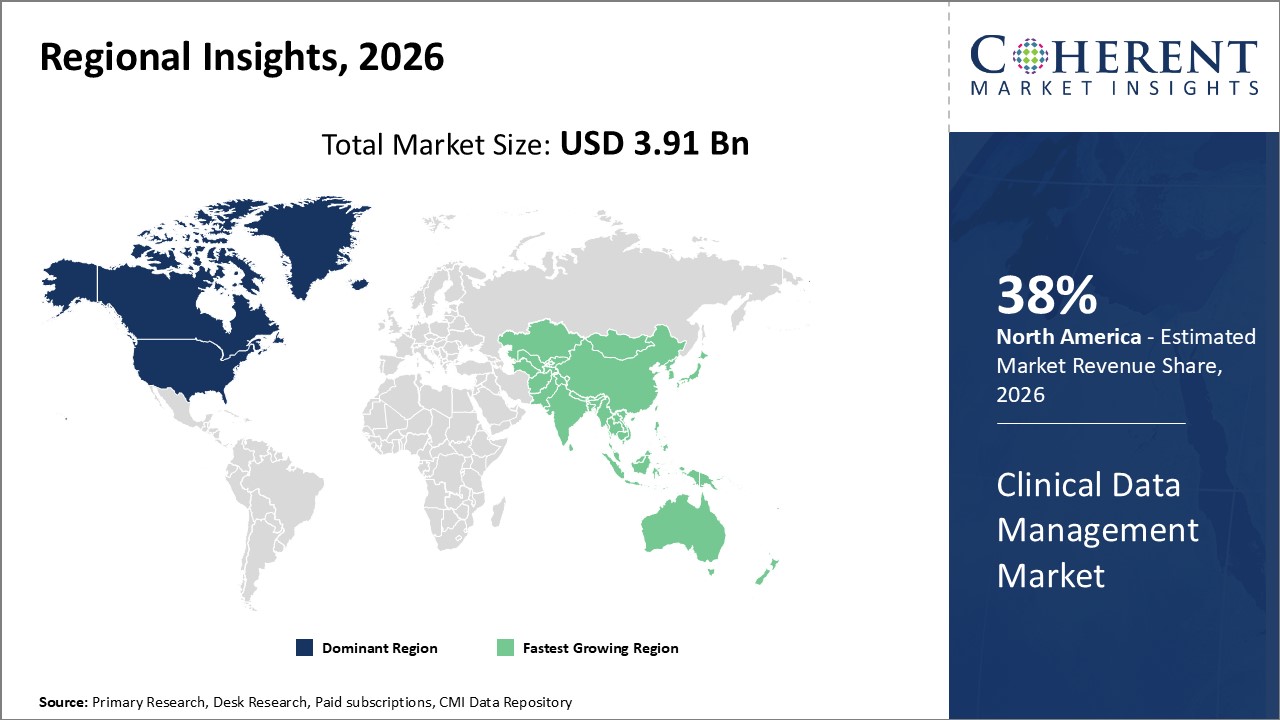
North America dominates clinical data management market share at 38%, primarily due to mature regulatory frameworks and technological ecosystem. Asia Pacific is the fastest-growing region with a CAGR exceeding 14%, propelled by expanding clinical trial activity, digital infrastructure improvements, and local manufacturing growth.
Clinical Data Management Market – Segmentation Analysis
To learn more about this report, Request Free Sample
Clinical Data Management Market Insights, By Deployment Type
The market is divided into Cloud-based, On-premises, Hybrid, and Others deployment types. With 57% of the market, cloud-based deployment is the most popular among them, indicating widespread use because of its scalability, affordability, and ease of integration. Particularly during the epidemic and after, cloud platforms have made remote monitoring and data access possible.
Clinical Data Management Market Insights, By Therapeutic Area
In terms of Therapeutic Area, the market segments into Oncology, Cardiovascular, Neurology, Infectious Diseases, and Others. Oncology leads the market with a 35% share, driven by extensive R&D pipelines and high-volume, multi-center trial designs demanding robust data management infrastructures.
Clinical Data Management Market Insights, By End-User
The market segmented by End-User comprises Pharmaceutical Companies, Contract Research Organizations (CROs), Biotechnology Firms, Academic Research Institutes, and Others. CROs lead with 43% market share, driven by escalating outsourcing trends and the growing complexity of clinical trials managed externally. Pharmaceutical companies represent a significant portion as well, continuously investing in internal CDM capabilities to innovate and reduce timelines.
Clinical Data Management Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Clinical Data Management Market Analysis and Trends
Dominance in the clinical data management market in North America is a result of a strong healthcare system, strict regulations such as those set by the FDA, and significant R&D expenditures. Due to investments made by large corporations with headquarters in the United States and Canada in cloud-based and AI-integrated CDM platforms, further, the region currently has around 38% of the market share.
Asia Pacific Clinical Data Management Market Analysis and Trends
Asia Pacific, on the other hand, is growing at the quickest rate, with a CAGR of more than 14%. Government incentives for pharmaceutical research, growing healthcare IT infrastructure, and rising clinical trial volumes in nations like China and India are some of the factors causing this. Prominent businesses that are actively creating regional centers help to accelerate market growth.
Clinical Data Management Market Outlook for Key Countries
United States Clinical Data Management Market Analysis and Trends
A state-of-the-art healthcare ecosystem and the existence of numerous leading industry players, including Medidata Solutions and Veeva Systems, are advantages for the clinical data management market in the USA. More than 35% of clinical trials registered worldwide in 2025 were conducted in the United States, highlighting the nation's crucial position in market income. Furthermore, the FDA's changing regulations regarding electronic data capture and data integrity have sped up the adoption of cutting-edge CDM solutions, creating an atmosphere that is conducive to company expansion.
China Clinical Data Management Market Analysis and Trends
Inpatient services, increased clinical trial programs, cross-border partnerships, and pro-pharmaceutical innovation government regulations have all contributed to the market's rapid growth in China. Together, leading domestic businesses and foreign firms growing their operations in China have supported steady market revenue development. Clinical trial registrations increased by 18% in 2025, indicating a greater need for trustworthy data management solutions. China's significance in the Asia Pacific market is further enhanced by the concurrent rise of digital health initiatives.
Analyst Opinion
The substantial rise in global clinical trials, especially in oncology and rare diseases, is a primary demand-side driver. For instance, over 8,500 oncology clinical trials were registered globally in 2025 alone, reflecting a 13% year-over-year increase, underlining escalating needs for efficient data management to handle voluminous patient and trial data sets.
From a supply-side perspective, enhanced production capacity of AI-integrated CDM tools has led to 20% faster data validation cycles compared to traditional manual processes, with trials in Europe reporting up to a 30% reduction in data entry errors in 2025.
A growing trend is the rise in cross-industry collaborations, with pharmaceutical companies partnering with IT providers to co-develop tailor-made clinical data management platforms, as evidenced by over 25 such alliances formed in 2025, which impact overall market volume substantially.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.91 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.8% | 2033 Value Projection: | USD 8.54 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Medidata Solutions, Oracle Health Sciences, Veeva Systems, IBM Watson Health, Parexel International, BioClinica, PAREXEL International, Signant Health, IQVIA Holdings, CRF Health | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Growth factors
Key drivers powering market growth include the digital transformation of clinical trials through advanced electronic data capture (EDC) and integrated CDM platforms, which raised clinical trial efficiency by nearly 25% in 2026. Stringent regulatory mandates worldwide, including FDA’s push for data integrity and compliance, are propelling demand for standardized data management solutions globally.
Growth in biologics and personalized medicine increases data complexity, driving adoption of adaptive CDM technologies that support multifactorial datasets. Lastly, rising outsourcing trends with CROs managing trial data fuels global market revenue, where CROs contributed to over 40% of clinical trial management in 2025, thus magnifying clinical data management service demand.
Clinical Data Management Market Development
Market Trends
The clinical data management market is rapidly aligning with AI and machine learning algorithms to enhance predictive data analytics and automate risk-based monitoring. Recent deployments of AI-driven data systems in Phase III trials for cardiovascular diseases have reduced monitoring costs by 18% in 2025.
There is also a discernible shift to decentralized clinical trials (DCTs), with DCTs projected to constitute nearly 30% of all trials by 2027, increasing demand for interoperable and cloud-based CDM systems that integrate remote data collection. Blockchain technology is another emerging trend to improve data traceability and security, with pilot projects in Europe reporting 25% improvement in audit readiness during 2026.
Key Players
Oracle Health Sciences
Veeva Systems
IBM Watson Health
Parexel International
BioClinica
Signant Health
IQVIA Holdings
CRF Health
Several market companies such as Medidata Solutions expanded their cloud-based platform capabilities through acquisitions, which resulted in a 17% revenue increase in 2025, strengthening their leadership position. Oracle Health Sciences adopted an AI-centric growth strategy, focusing on predictive modelling, which enhanced customer retention rates by approximately 22% in recent years.
Clinical Data Management Market Future Outlook
The clinical data management market refers to the industry focused on providing software and services to facilitate the collection, integration, and management of clinical trial data. This market includes solutions for electronic data capture, data validation, and overall data management to ensure the integrity and regulatory compliance of clinical study information. It supports pharmaceutical and biotechnology companies, clinical research organizations, and other stakeholders in accelerating drug development and improving clinical research efficiency.
The market is experiencing significant growth driven by technological advancements such as artificial intelligence, machine learning, and cloud-based platforms, which enhance data accuracy, accessibility, and scalability. Increasing complexity and volume of clinical trials, stringent regulatory frameworks, and the globalization of clinical research activities further propel the demand for robust clinical data management systems. Geographically, North America leads with a strong healthcare infrastructure and R&D investment, while emerging markets such as Asia Pacific are witnessing rapid expansion. The market overall is characterized by ongoing innovation, outsourcing trends, and a strong emphasis on quality and compliance across clinical research processes.
Historical Analysis
The Clinical Data Management Market evolved significantly over the decades, beginning with manual, paper-based processes that dominated clinical data handling during the early and mid-20th century. Initially, data management was heavily reliant on physical documentation, emphasizing meticulous record keeping and manual analysis, which was time-consuming but necessary for clinical research accuracy. The introduction of computers in the 1970s marked a turning point, enabling digital data processing and improved efficiency. The 1990s saw specialized software designed for clinical data capture and management emerge, which greatly reduced errors and accelerated trial timelines.
Over time, the market expanded alongside the increasing complexity and volume of clinical trials, which pushed the demand for more advanced, automated, and cloud-based solutions. Technological advances such as electronic data capture, artificial intelligence, and machine learning revolutionized data accuracy and management capabilities. The market also adapted to growing regulatory requirements and the globalization of clinical trials, shifting towards real-time data access and decentralized trial management. Recent trends focused on integrating diverse data sources including wearable devices and electronic health records. Geographically, the market initially developed strongest in North America and Europe, with significant expansion occurring in Asia Pacific and emerging regions. Overall, the historical growth of the Clinical Data Management Market reflected ongoing digital transformation and innovation to meet the increasing demands of modern clinical research.
Sources
Primary Research interviews:
CTOs (Chief Technology Officers)
Data Scientists
Database Administrators (DBAs)
Databases:
Web of Science
ACM Digital Library
ProQuest
Magazines:
CIO Magazine
Database Trends and Applications (DBTA)
Computerworld
Journals:
Journal of Data and Information Quality
Journal of Big Data
International Journal of Information Management
Newspapers:
The Economic Times
The New York Times (Technology section)
Financial Times
Associations:
ACM (Association for Computing Machinery)
IEEE Computer Society
International Association for Information and Data Quality (IAIDQ)
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients