Coronary Stents Market is estimated to be valued at USD 15,078.6 Mn in 2026 and is expected to reach USD 26,060.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033.
The global Coronary Stents Market is witnessing consistent growth, fuelled by the rising prevalence of cardiovascular diseases and increasing demand for minimally invasive procedures. Key product segments include bare metal stents, drug-eluting stents, and bioresorbable stents, with bare metal stents holding the largest share. Advancements in stent technology, including improved biocompatibility and deliverability, are enhancing patient outcomes.
|
Current Event |
Description and its impact |
|
Product Innovations and Launches |
|
|
Strategic Collaborations and Investments |
|
|
Regulatory and Health Policy Developments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The coronary stents market is witnessing robust pipeline activity, with several next-generation products under development focusing on enhanced safety, efficacy, and patient compatibility. Key innovations include polymer-free drug-eluting stents, bioresorbable scaffolds, and stents with improved flexibility and deliverability for complex lesion procedures.
Leading companies such as Abbott, Boston Scientific, and Medtronic are actively conducting late-stage clinical trials to gain regulatory approvals across major markets, particularly in the U.S. and Europe. Emerging players are also entering the market with novel stent designs that reduce restenosis, improve endothelial healing, and eliminate long-term inflammation risks.
In addition, the adoption of coronary stents risk assessment tools is gaining momentum as healthcare providers aim to enhance patient outcomes through better procedural planning and post-operative monitoring. These tools, often supported by AI-driven imaging and data analytics, help clinicians identify high-risk patients, predict complications, and guide personalized stent selection strategies.
Furthermore, precision delivery systems are being integrated into future stent platforms to optimize placement and outcomes. The growing demand for personalized cardiac care, combined with regulatory support for innovation, is accelerating R&D efforts. As these products progress through the pipeline, they are expected to reshape the competitive landscape and broaden treatment options globally.
The patent landscape for coronary stents reveals intense innovation, especially in bioresorbable scaffolds, drug-eluting stents (DES), and surface-coating technologies. Second-generation DES with biodegradable polymers and polymer-free designs are attracting numerous patent filings, while advanced drug delivery systems such as nanostructured surfaces and dual‑therapy coatings are earning intellectual protection.
Significant activity is occurring in bioresorbable stent patents. Firms like Abbott, Biotronik, and emerging entities are promoting next-gen scaffolds with enhanced materials (e.g., magnesium-based) and accelerated resorption profiles to address past safety concerns. Novel coating techniques such as asymmetric drug layering and endothelial-progenitor cell capture are also protected, including patents like OrbusNeich’s Genous/Combo dual-therapy technology.
Additionally, early-stage intellectual property covers personalized stent architectures enabled by 4D printing and AI-guided placement systems. Together, these patent trends signal a shift toward smart, patient‑centric, and safer coronary stent platforms shaping future market dynamics.
Reimbursement policies profoundly influence adoption and accessibility of coronary stents across regions. In the United States, comprehensive coverage from Medicare and private insurers for percutaneous coronary interventions (PCI) facilitates widespread use of both bare-metal and advanced drug-eluting stents, helping sustain North America’s market dominance.
However, the costly nature of newer technologies like absorbable DES priced 30–50% higher than traditional stents is sometimes inadequately offset by existing payment models under DRG systems, leading hospitals to favour lower-cost alternatives.
In Germany and France, strong public insurance frameworks and dedicated device reimbursement negotiate premiums for innovative stents streamlining access for DES and bioresorbable options. Conversely, in emerging markets like India, limited insurance coverage caps penetration below 2%, restricting uptake of cutting-edge stents.
Growth in value-based reimbursement models illustrated by Japan’s premium tier for resorbable stents linked to clinical performance offers promising pathways for broader adoption of next-generation technologies.
Prescribers’ preferences in the coronary stents market are heavily influenced by clinical efficacy, patient risk profile, long-term outcomes, and ease of deployment. Drug-eluting stents (DES) remain the preferred choice among interventional cardiologists due to their lower rates of restenosis and improved long-term patency. Newer-generation DES with biocompatible or biodegradable polymers are especially favored for their enhanced safety and performance in complex lesions.
Bare-metal stents (BMS), although declining in overall use, are still prescribed in select cases—particularly for patients with high bleeding risks or those unable to undergo long-term dual antiplatelet therapy. Meanwhile, bioresorbable stents are gaining cautious interest due to their potential to eliminate long-term complications, though concerns over early thrombosis and limited long-term data have tempered widespread adoption.
Prescribers also consider procedural flexibility, stent tractability, and post-procedure patient compliance. As data from real-world evidence and clinical trials evolve, preferences continue shifting toward stents offering superior safety, efficacy, and tailored patient outcomes.
The coronary stents are mostly used to treat coronary artery diseases, hence, the increasing product approvals by regulatory authorities is expected to drive the market growth over the forecast period. For instance, in May 2022, Biosensors International Group, Ltd., a manufacturer of medical devices, announced Japanese Pharmaceuticals and Medical Devices Agency (PMDA) approval for BioFreedom Ultra and U.S. Food and Drug Administration (FDA) approval for BioFreedom.
In October 2024, Reuters highlighted that Elixir Medical’s new DynamX bioadaptor stent, which initially behaves like a typical drug-eluting stent, resorbs its polymer coating over six months to allow natural arterial motion. In a year-long Swedish trial involving 2,399 patients, DynamX showed lower rates of heart attacks, repeat procedures, and cardiac death compared to Medtronic’s standard drug-eluting stent demonstrating a potential breakthrough in adaptive stent design.
Coronary stents are used for the treatment of coronary artery diseases and thus, the increasing prevalence of coronary artery diseases is expected to drive the market growth over the forecast period. For instance, in February 2022, according to an article published by Clinical Trials Arena, a leading source of data journalism on clinical trials strategy & operations, stated that coronary artery disease (CAD), inflicts a sizeable disease burden on the U.S. and remains the country’s leading cause of death.
In March 2025, Business Standard reported that India’s drug price regulator (NPPA) approved a ~2% price increase on coronary stents to adjust for inflation. The revised ceiling pricing sets bare-metal stents at ₹10,692.69 and eluting/bioresorbable stents at ₹38,933.14. This regulatory move reflects ongoing cost pressures in the domestic stent market while ensuring patient affordability.
Bare metal stents are projected to dominate the global coronary stents market, accounting for 46.5% of the total market share in 2026. This dominance is largely attributed to their affordability, ease of deployment, and reduced need for prolonged dual antiplatelet therapy, making them especially suitable for patients at high risk of bleeding complications.
Despite the increasing popularity of drug-eluting stents, bare metal stents continue to play a vital role in specific clinical scenarios, particularly in healthcare settings with limited access to advanced medical infrastructure. Recent innovations in stent architecture such as enhanced radial strength and the use of biocompatible materials are improving clinical outcomes in acute coronary syndrome interventions.
Furthermore, emerging economies are experiencing a surge in percutaneous coronary intervention (PCI) procedures due to rising cardiovascular disease prevalence and improving healthcare access. These factors collectively strengthen the position of bare metal stents as a critical component in the global coronary stents market, especially in cost-sensitive and high-volume healthcare environments.
To learn more about this report, Request Free Sample
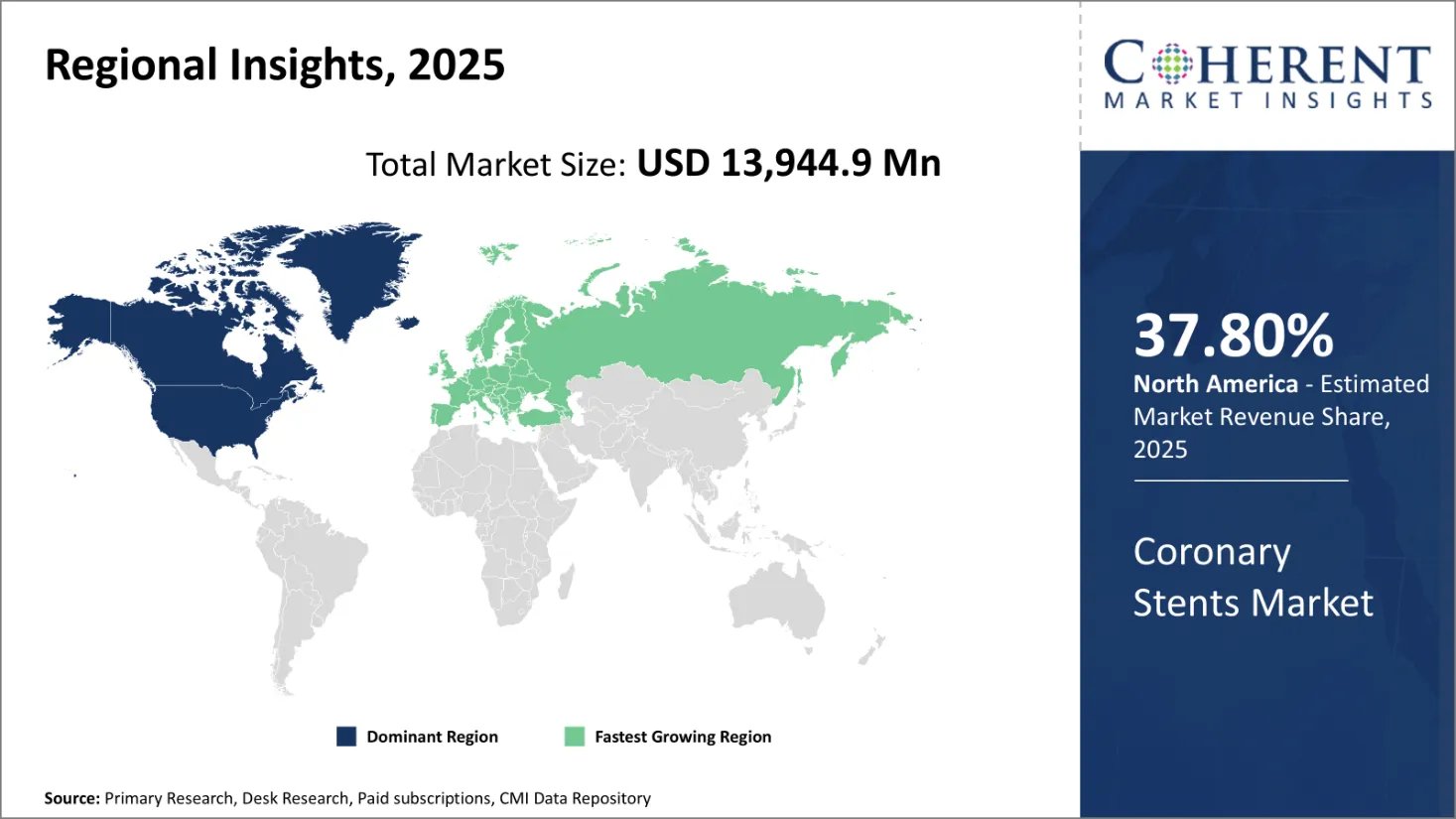
North America is expected to lead the global coronary stents market with a projected 37.80% market share in 2026, reflecting the region’s advanced healthcare ecosystem and strong clinical focus on cardiovascular disease management. This regional leadership is primarily driven by a high incidence of coronary artery disease, early adoption of next-generation stent technologies, and favourable reimbursement frameworks.
The U.S. remains at the forefront of coronary stent innovation, supported by continuous R&D investment, streamlined FDA approval pathways, and robust funding for interventional cardiology research. Healthcare providers across the region are increasingly integrating bioresorbable scaffolds and polymer-free drug-eluting stents into treatment protocols, offering enhanced patient outcomes and reduced long-term complications.
Moreover, the region’s aging population, growing burden of lifestyle-related cardiac disorders, and emphasis on minimally invasive procedures are accelerating procedural volumes. Strategic collaborations between hospitals, academic institutions, and medical device companies are fostering innovation, expanding access, and reinforcing North America’s dominant position in the global coronary stents market.
The United States leads the global coronary stents market, supported by its highly advanced healthcare infrastructure, strong R&D ecosystem, and high procedural volumes for cardiovascular interventions. The U.S. accounts for the largest share of North America’s projected 37.8% market share in 2026, driven by the widespread prevalence of coronary artery disease and the growing adoption of minimally invasive procedures.
Government support through Medicare and private insurance coverage makes coronary stenting widely accessible. The FDA’s accelerated approval pathways and continued investment in clinical trials have enabled the rapid commercialization of next-generation stents, including bioresorbable and polymer-free technologies.
Moreover, increasing emphasis on coronary stents and blood clot prevention has led to the development of drug-eluting and antithrombotic-coated stents aimed at reducing post-procedural complications. Major players like Abbott, Boston Scientific, and Medtronic continue to innovate and expand their product portfolios, reinforcing the U.S.'s dominant position in the global coronary stents industry.
Canada complements the U.S. market leadership with a strong focus on evidence-based cardiology practices, public healthcare accessibility, and nationwide cardiac care programs. Provincial healthcare systems are actively investing in improving cardiovascular care infrastructure, leading to steady growth in percutaneous coronary interventions across the country.
Research institutions and government agencies such as the Canadian Institutes of Health Research (CIHR) are collaborating with medical device companies to support innovation in stent technologies. With increasing awareness of cardiovascular health, a rising elderly population, and support for early diagnosis and intervention, Canada continues to strengthen its role in the North American coronary stents landscape.
Together, the United States and Canada drive North America’s dominance in the coronary stents market by combining technological innovation, clinical expertise, and broad healthcare access to meet growing global demand for effective cardiovascular treatment solutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 15,078.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 26,060.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, iVascular, Alvimedica, Alta Biomaterials, Japan Medical Device Technology Co., Ltd., Veritas Bioventions Pvt Ltd, Nano Therapeutics Pvt. Ltd, Abbott, SLTL Group, Translumina Therapeutics LLP, DSM, Arterius, Terumo Corporation, SMT, Boston Scientific Corporation, Cook, Biotronik, B. Braun SE, MicroPort Scientific Corporation, Meril Life Sciences Pvt. Ltd, C. R. Bard, Inc, KYOTO MEDICAL PLANNING Co., Ltd, QualiMed, Elixir Medical Corporation, ENDOCOR GmbH & CO. KG, Amaranth Medical, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients