Global electrophysiology devices market is estimated to be valued at USD 12.41 Bn in 2026 and is expected to reach USD 29.55 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 13.2% from 2026 to 2033.
To learn more about this report, Request Free Sample
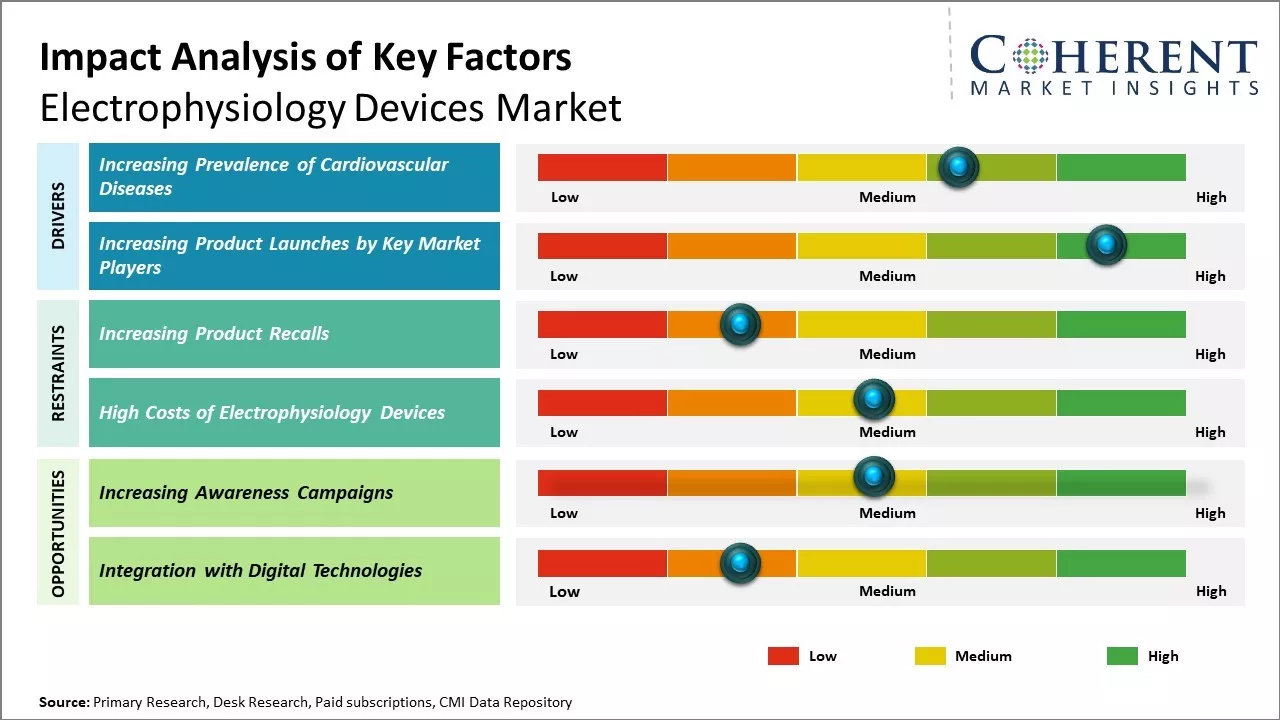
The rising incidences of cardiac arrhythmias, growing geriatric population, technological advancements in electrophysiology such as remote magnetic navigation, 3D mapping systems, and robotic navigation are expected to drive growth of the electrophysiology devices market.
Rising awareness about early disease diagnosis and treatment and availability of reimbursements are further expected to boost demand for electrophysiology devices for diagnosis and treatment of cardiac arrhythmias. However, high cost of complex cardiac electrophysiology procedures and dearth of skilled physicians can hamper the market growth.
|
Current Events |
Description and its impact |
|
Technological Advancements in Pulsed Field Ablation (PFA) |
|
|
Regulatory Shifts in the EU Medical Device Regulation (MDR) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global electrophysiology devices market growth is driven by rising prevalence of cardiovascular diseases around the world. Cardiovascular diseases have been increasing at an alarming rate over the past few decades due to changing lifestyle patterns and risk factors such as rising levels of obesity, lack of physical activity, stressful living and working conditions, and increasing consumption of tobacco, alcohol, and unhealthy foods.
Heart diseases and disorders related to heart rhythm abnormalities are becoming more widespread in both developing and developed countries. Electrophysiology procedures help to treat various types of cardiac arrhythmias using sophisticated mapping and ablation techniques. Thus, growing disease burden of cardiovascular illnesses boosts demand for electrophysiology devices.
Increasing product launches are indeed a key driver for the electrophysiology devices market. For instance, in October 2024, Medtronic received FDA approval for its Affera Mapping and Ablation System with Sphere-9 Catheter, offering an all-in-one solution for persistent atrial fibrillation. Similarly, in November 2024, Johnson & Johnson MedTech announced U.S. FDA approval of the VARIPULSE Platform for drug-refractory paroxysmal Atrial Fibrillation. These innovations highlight the continuous advancements and expanding treatment options in the field.
Increasing adoption of inorganic growth strategies such as launch of awareness campaigns can offer opportunities for the market growth. For instance, on February 20, 2024, OMRON Healthcare, Co., Ltd., a medical device company, launched a global campaign to promote awareness about atrial fibrillation (AFib), the most common type of heart arrythmia1, and how to reduce the high stroke risk associated with the condition. As a part of the campaign, the company released The Sound of AFib, a film short that was the centerpiece of the campaign.
Electrophysiology Treatment Devices segment is estimated to hold 62.8% of the market share in 2026, owing to rising prevalence of various cardiac disorders globally. Electrophysiology monitoring devices such as Electrocardiography (ECG) devices, Electroencephalography (EEG) devices, Electromyography (EMG) devices and other monitoring equipment play a crucial role in diagnosing cardiac arrhythmias and other heart conditions.
Increasing awareness about early detection of cardiac disorders and availability of advanced cardiac monitoring devices are expected to drive the segment growth. Furthermore, ongoing technological advancements in cardiac monitoring equipment like wireless and remote monitoring solutions have boosted the adoption of electrophysiology monitoring devices across hospitals and diagnostic centers.
To learn more about this report, Request Free Sample
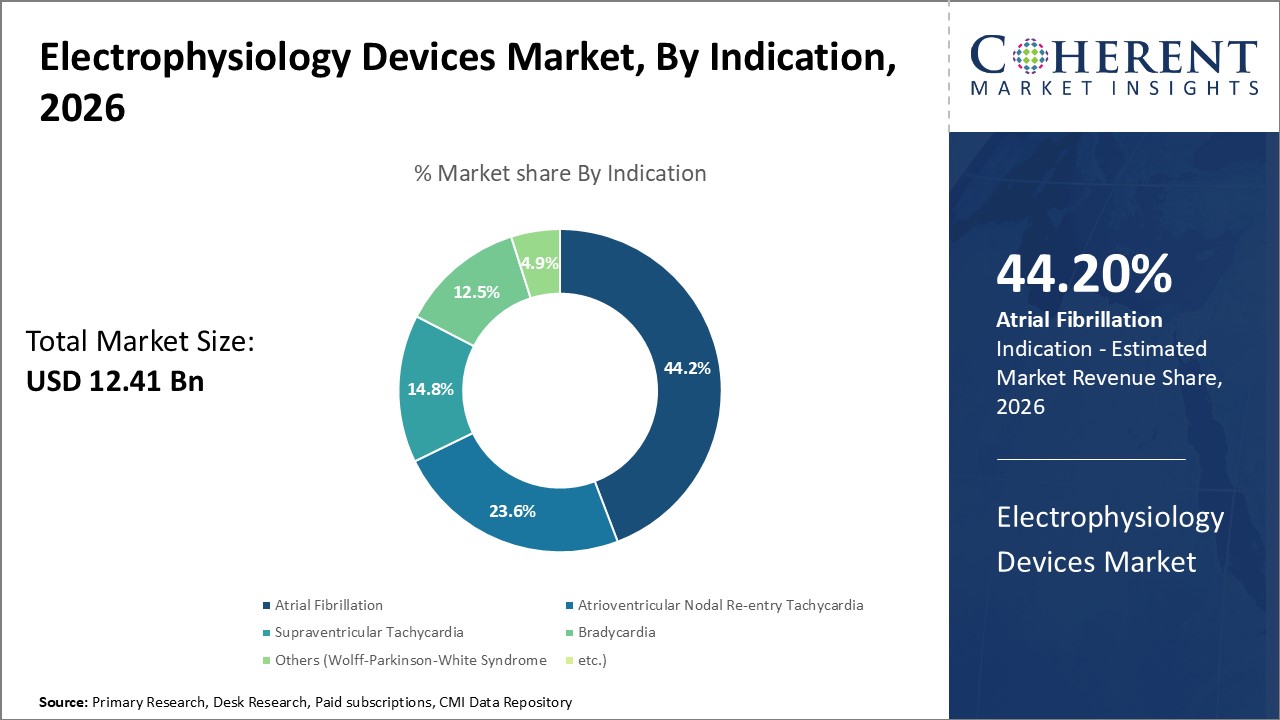
Atrial Fibrillation segment is estimated to hold 44.2% of the market share in 2026, owing to its rising global prevalence. Electrophysiology treatment devices such as ablation catheters, cardiac mapping systems, EP lab devices and other accessories play a vital role in treating various cardiac arrhythmias, especially atrial fibrillation (AFib).
The consistently increasing incidence of AFib worldwide primarily due to aging population and prevalent risk factors like obesity, high blood pressure, diabetes, and others has boosted demand for electrophysiology treatment devices for AFib treatment. The favorable reimbursement policies for cardiac ablation procedures in developed countries and growing preference for minimally invasive treatments have further augmented the growth of this segment.
Hospitals segment is estimated to hold 40.7% of the market share in 2026, owing to well-established healthcare infrastructure and availability of cutting-edge cardiac facilities. Majority of cardiac ablation and electrophysiological diagnostic procedures are performed in hospitals equipped with specialized EP labs, cardiac catheters labs and highly skilled electrophysiologists.
The rapidly growing hospital industry worldwide especially in developing nations, increasing healthcare expenditures, favorable reimbursements and heavy investments by both private and public players in developing hospital infrastructure have created an ideal environment for growth of this end-user segment. Hospitals offer one-stop solutions for cardiac patients requiring long-term care before and after complex electrophysiology procedures.
To learn more about this report, Request Free Sample
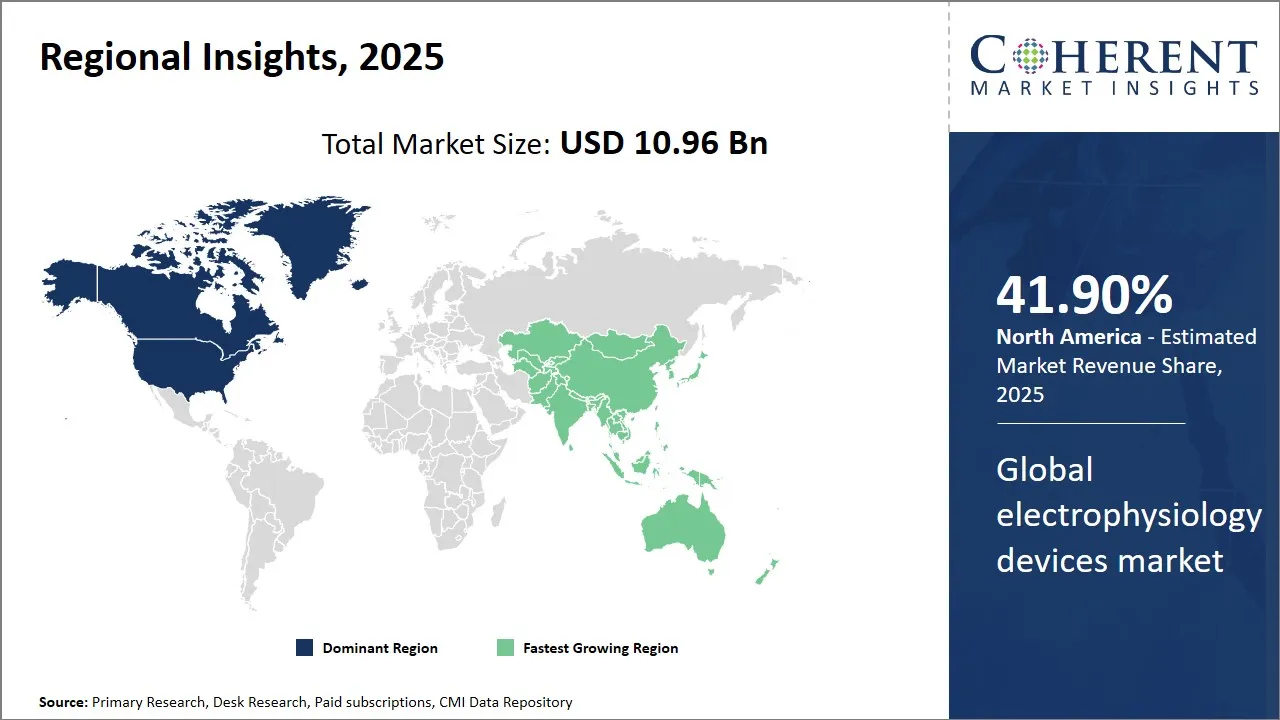
North America remains the dominant region in the global electrophysiology devices market and is estimated to hold 41.90% of the market share in 2026. With a strong presence of leading medical device companies, the region has witnessed rapid technological advancements in electrophysiology. Several new product launches catering to arrhythmias and Atrial Fibrillation treatments have cemented North America's position as the innovation hub.
Moreover, favorable reimbursement policies for complex ablation procedures in the U.S. and Canada have ensured higher procedure volumes. Leading healthcare institutions in the region are also early adopters of new electrophysiology technologies. This helps companies gauge real-world performance and drives further innovation.
The Asia Pacific region is emerging as the fastest growing market for electrophysiology devices. Countries like China, India, Japan and South Korea are witnessing significant investments by global medtech giants to tap into their huge untreated patient populations. Local manufacturing is also being incentivized through favorable regulation.
Countries in Asia Pacific present a unique growth opportunity due to rising healthcare expenditures and a growing demand for high-quality care. However, pricing pressures remain a key challenge due to price-sensitive markets and universal healthcare systems. Companies must offer highly competitive products tailored to the requirements of this cost-conscious region.
The U.S. electrophysiology devices market is experiencing robust growth, primarily fueled by the increasing prevalence of cardiac arrhythmias, particularly atrial fibrillation, and a highly developed healthcare infrastructure. Current trends include the significant uptake of pulsed-field ablation (PFA) systems, a major advancement in ablation technology, and continued innovation in sophisticated 3D mapping and navigation systems. The market is also seeing greater integration of artificial intelligence for enhanced diagnostic precision and treatment personalization. Favorable reimbursement policies continue to support the adoption of these advanced devices, reinforcing the U.S. as a leader in the global electrophysiology market. For instance, Medtronic's PulseSelect Pulsed Field Ablation System received FDA approval on December 13, 2023, marking a significant milestone in bringing this novel technology to the U.S. market.
The Chinese electrophysiology devices market is undergoing rapid expansion, driven by a growing burden of cardiovascular diseases, an aging demographic, and expanding access to specialized cardiac care. Recent trends highlight a strong adoption of advanced technologies such as three-dimensional mapping and advanced ablation techniques, including pulsed-field ablation, which are improving procedural outcomes.
Government initiatives aimed at healthcare reform and strategic investments in medical infrastructure are significantly contributing to market growth. There's also a rising interest in localized production and a growing number of collaborations between domestic and international companies to introduce cutting-edge technologies. For instance, Stereotaxis and MicroPort EP announced NMPA approval for the Magbot Magnetic Navigation Ablation Catheter in December 2024, further advancing robotic navigation in China.
Germany's electrophysiology devices market shows steady growth, supported by a sophisticated healthcare system, a significant incidence of cardiac arrhythmias, and a strong emphasis on technological innovation. Current trends indicate increasing demand for advanced ablation catheters and integrated diagnostic tools, with a focus on enhancing procedural efficiency and patient safety.
The market benefits from substantial investments in research and development and a robust regulatory environment that encourages the adoption of the latest electrophysiology solutions. The presence of leading global device manufacturers and a highly skilled medical workforce further solidifies Germany's prominent position in this specialized field. For instance, the University Medical Center Göttingen in Germany recently adopted the Optrell mapping catheter in late 2024, demonstrating their commitment to integrating high-resolution diagnostic tools for complex arrhythmia cases.
The Japanese electrophysiology devices market is demonstrating substantial growth, largely driven by its rapidly aging population, which contributes to a higher prevalence of cardiac arrhythmias, and a strong preference for minimally invasive procedures.
Key trends include the ongoing integration of advanced mapping systems, the introduction of next-generation ablation technologies like pulsed-field ablation, and the increasing application of digital health and AI in arrhythmia diagnosis and management. Despite challenges such as high equipment costs, the sustained demand for sophisticated cardiac care and continuous technological advancements are propelling the market's expansion.
For instance, Boston Scientific received Japanese regulatory approval for its FARAPULSE Pulsed Field Ablation (PFA) System in September 2024, signifying a major new non-thermal ablation option becoming available in the Japanese market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12.41 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.2% | 2033 Value Projection: | USD 29.55 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, VivaQuant, Inc., Johnson & Johnson Services, Inc., AliveCor, Inc., Bittium, Nihon Kohden Corporation, Shimmer, OMRON Healthcare, Co., Ltd., VivaLNK, Inc., Abbott, BIOTRONIK, Koninklijke Philips N.V., Omega Medical Imaging, LLC., HeartSciences, MicroPort Scientific Corporation, Siemens Healthcare GmbH |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Segment / System (Manufacturer) |
Estimated Upfront Cost (USD) |
Recurring Costs (per procedure) |
|
3D Mapping System (Carto 3, Biosense Webster) |
250,000 – 800,000 |
- Navigation patches: USD 420–USD 1,045 - Mapping catheters: USD 1,500–USD 3,000 |
|
EP Recording System |
~160,000 |
Includes disposable patches and cables (~USD 200–USD 400 per procedure) |
|
RF Ablation Catheters (open irrigated) |
1,500–2,800 |
• Cabling & tubing: USD 80–USD 400 • RF transseptal needle (e.g., NRG): ~ USD 470 |
|
Bidirectional Ablation Catheters (e.g., Blazer DX) |
1,700–3,000 |
Cable: ~USD 250 |
|
Cryoballoon Catheter (e.g., Medtronic Arctic Front) |
6,500 |
Cable & umbilical cable: USD 215 + USD 145 = USD 360 |
|
Intracardiac Echo (ICE) Catheters |
1,050 (rotational) – 2,800 (phased‑array) |
Requires echo machine (~USD 131,400) amortized over ~83 cases |
|
PFA Pentaspline Catheter (Farapulse, Boston Sci.) |
Included in console; comparable COLD catheter setups |
USD 8,500 upfront; offsets by lower repeat procedures |
|
Complete EP Lab Setup (console + mapping + recording) |
500,000 + depending on configuration |
- Consumables per procedure: ~USD 4,000–USD 6,500 (catheter ablation average) |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Company |
Key Facilities (Location) |
Manufacturing Focus |
Estimated Capacity |
Portfolio Highlights |
|
Abbott Laboratories |
- St. Paul, Minnesota, USA |
- Advanced EP catheters & mapping systems |
- Several hundred thousand catheters/year |
Diagnostic catheters, 3D mapping systems, RF ablation catheters |
|
Medtronic plc |
- Mounds View, Minnesota, USA |
- Pacemakers, defibrillators |
- ~500,000 devices/year |
EP catheters, mapping systems, ablation tools, pulse generators |
|
Boston Scientific Corp. |
- Marlborough, Massachusetts, USA |
- Cryoablation & RF ablation catheters |
- ~300,000 catheters/year |
Cryo/RF ablation catheters, mapping systems, diagnostic catheters |
|
Biotronik SE & Co. KG |
- Berlin, Germany |
- Implantable devices (pacemakers, ICDs) |
- ~150,000 units/year |
Pacemakers, ICDs, diagnostic EP catheters |
|
Siemens Healthineers |
- Erlangen, Germany |
- 3D cardiac mapping systems |
Not disclosed (software & imaging focus) |
Cardiac mapping, navigation systems, EP software (no catheter production) |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Electrophysiology devices are medical devices that are used to diagnose arrhythmias and atrial fibrillation. Atrial fibrillation is arrhythmia where the chambers of the heart begin to rapidly beat. An electrophysiology study is a test performed to assess heart's electrical system or activity and is used to diagnose abnormal heartbeats or arrhythmia. The test is performed by inserting catheters and then wire electrodes which measure electrical activity through blood vessels that enter the heart.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients