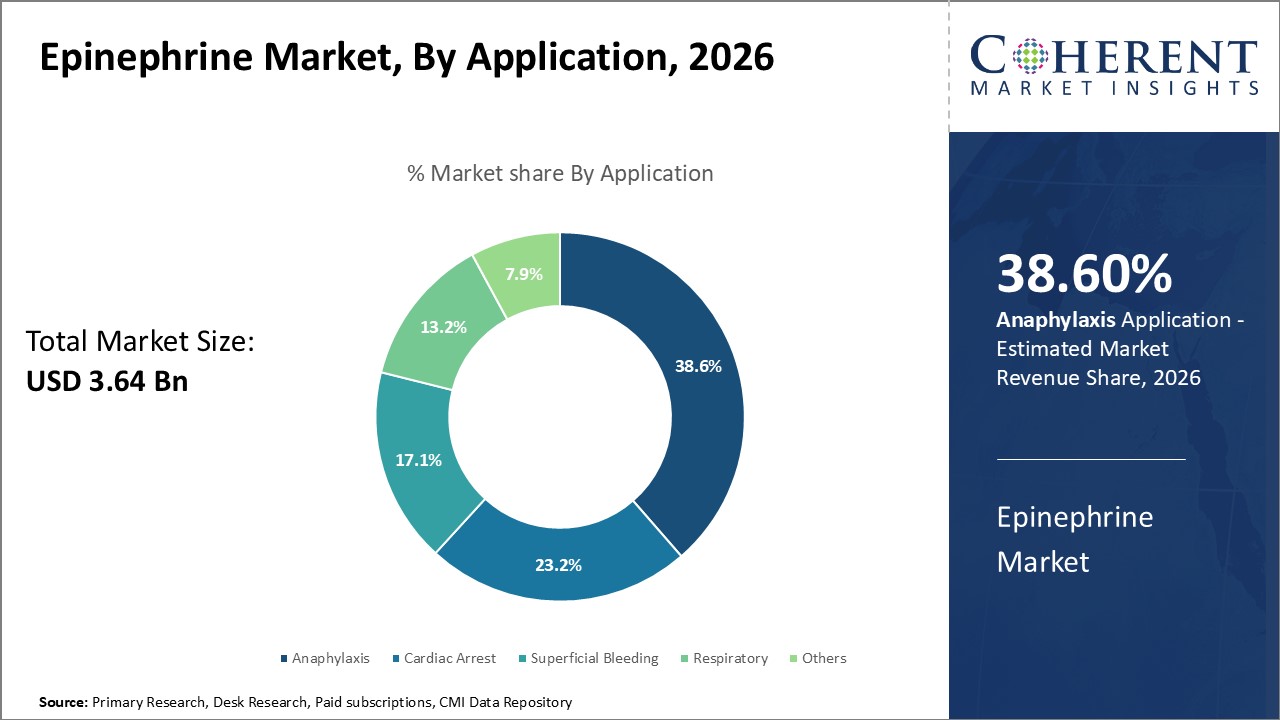
Global epinephrine market is estimated to be valued at USD 3.64 Bn in 2026 and is expected to reach USD 8.05 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 12.0% from 2026 to 2033.
To learn more about this report, Request Free Sample
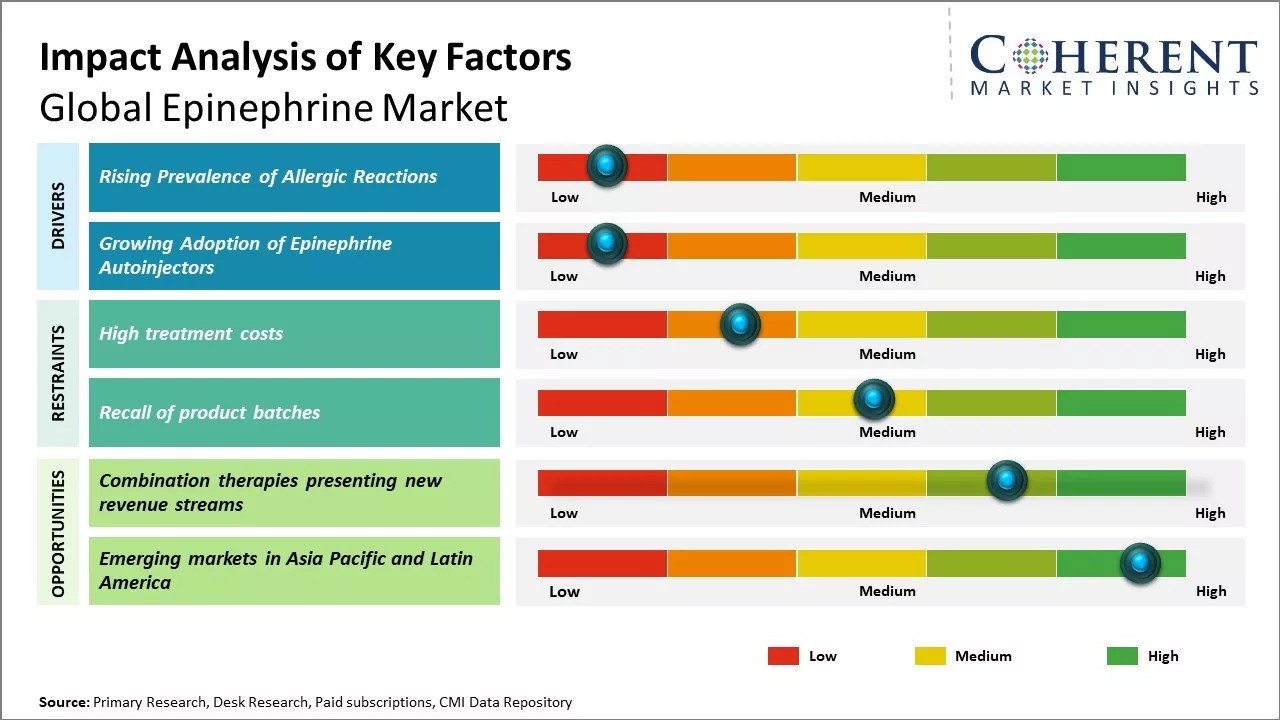
The epinephrine market is poised for growth, driven by the rising incidence of life-threatening medical emergencies such as anaphylaxis and cardiac arrests, which require immediate intervention through emergency epinephrine injections. Growing awareness about the importance of early treatment for severe allergic reactions is further boosting adoption, particularly with the increasing availability of user-friendly epinephrine auto-injectors designed for rapid, at-home use.
Additionally, initiatives by healthcare providers and advocacy groups to educate patients and caregivers on the timely use of epinephrine are contributing to the rising epinephrine market demand.
|
Current Event |
Description and its Impact |
|
Regulatory Approvals for Novel Delivery Systems |
|
|
Clinical Practice Transformations |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Reimbursement for epinephrine, particularly in the form of auto-injectors used to treat severe allergic reactions like anaphylaxis, varies widely across countries but is generally supported due to its status as a life-saving medication. In the United States, epinephrine is typically covered under Medicare Part D and state Medicaid programs. Private insurers usually cover epinephrine, but copayments can vary significantly, sometimes reaching hundreds of dollars. To ease this burden, manufacturers often offer co-pay assistance and patient support programs. In Canada, epinephrine is included in most provincial drug plans, with varying coverage rules, and is more broadly supported through private insurance.
The United Kingdom’s National Health Service (NHS) covers epinephrine auto-injectors with a nominal prescription charge, and children are eligible for free emergency access in schools. Similarly, Germany’s statutory health insurance reimburses epinephrine for individuals diagnosed with severe allergies, with additional support for training and allergy management.
In Australia, epinephrine is subsidized through the Pharmaceutical Benefits Scheme (PBS), while France reimburses around 65% of the cost through public insurance, with many patients relying on complementary plans for the balance.
In terms of product type, auto-injector segment is estimated to contribute the highest market share of 51.5% in 2026, owing to their convenience and portability. Auto-injectors allow for self-administration of epinephrine during an anaphylactic reaction without the need for assembly or handling of loose needles. This ease of use makes auto-injectors preferable for patients, especially those at higher risk such as children, or for situations away from medical supervision. Their compact size and ergonomic shape also enable auto-injectors to be easily stored and carried, in backpacks or purses, so that epinephrine treatment is immediately available when needed. Various initiatives by drugmakers to develop similar user-friendly devices have raised awareness about them. Due to their huge demand in emergency medical cases, auto-injectors remain the preferred product type of epinephrine market.
In February 2025, Rx Bandz signed a memorandum of understanding (MOU) with Zeyco to fill and distribute its MiniJect® epinephrine auto-injector across Mexico, Central, and South America. The partnership aims to expand access to the compact, user-friendly device designed for individuals with severe allergies.
To learn more about this report, Request Free Sample
In terms of application, anaphylaxis segment is estimated to contribute the highest market share of as it is primary condition treated by epinephrine. Anaphylaxis is a severe, potentially life-threatening allergic reaction that can occur within minutes of exposure to an allergen. Common triggers include foods, medications, insect stings or bites. As the prevalence of food allergies and insect venom allergies rises globally, there will cases of anaphylaxis. Epinephrine injection via auto-injector is the first-line treatment for anaphylaxis. Its alpha- and beta-adrenergic effects help reverse multiple symptoms such as low blood pressure, breathing difficulties and skin reactions. Awareness programs educate people about prompt epinephrine administration in anaphylaxis emergencies, thus, boosting their demand.
In August 2024, the U.S. Food and Drug Administration expanded its approval of Neffy®, a needle-free epinephrine nasal spray for anaphylaxis by ARS Pharmaceuticals, to include children weighing 15–30 kg, in addition to the existing 2 mg dose for those over 30 kg.
In terms of distribution channel, hospital pharmacies segment is estimated to contribute the highest market share of as patients commonly obtain epinephrine prescriptions or refills from hospital pharmacies due to factors like hospitals employ specialists such as allergists and immunologists who diagnose and manage conditions requiring epinephrine treatment such as anaphylaxis. Patients are also educated on appropriate epinephrine use and demonstrate techniques before being discharged. Hospital setting also allows direct supervision over administration technique and dosage. Even for maintenance needs, the coordinated care and insurance coverage benefits ensure continued fills through affiliated hospital outlets.
In May 2025, Endo, Inc., a diversified pharmaceutical company, introduced Adrenalin (epinephrine in 0.9% sodium chloride injection) 8 mg/250 mL premixed IV bag. This marks the first FDA-approved and currently the only commercially available manufacturer-prepared epinephrine premixed IV bag. Aimed at hospital pharmacies, the product supports streamlined operations and reduced preparation time.
To learn more about this report, Request Free Sample
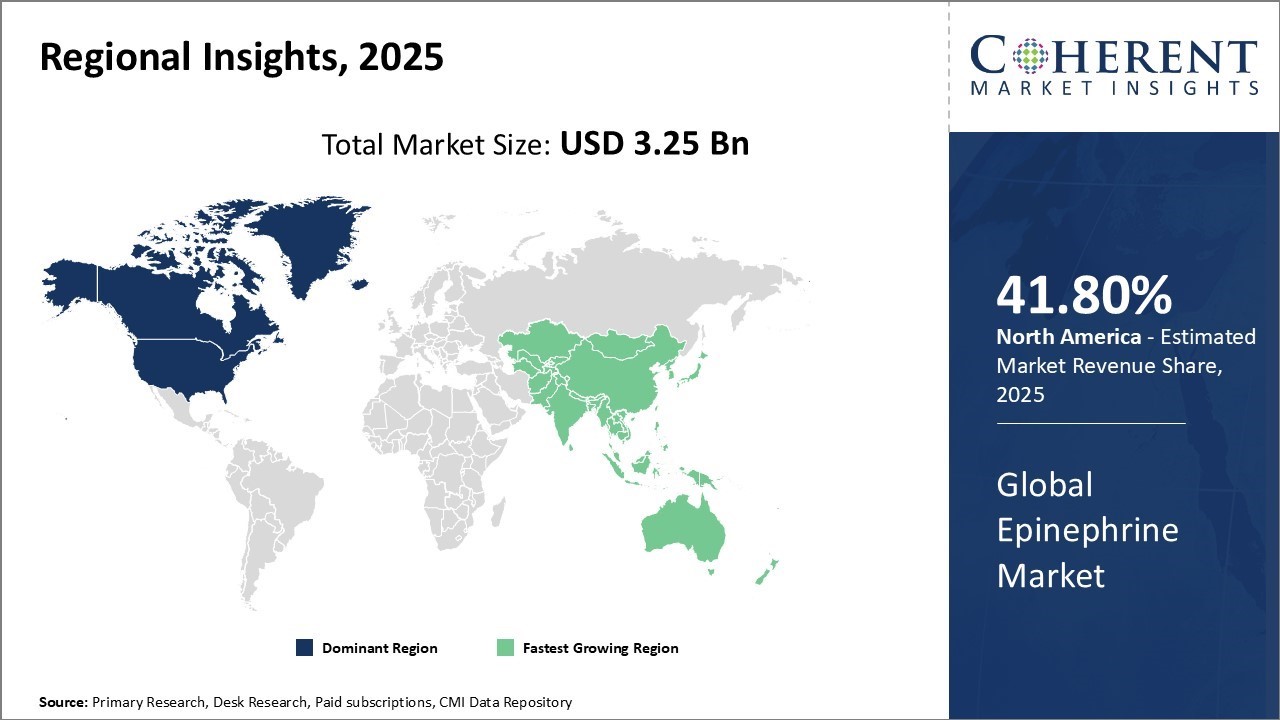
North America dominates the global epinephrine market and accounts for highest market share of 41.80% in 2026. The region has strong presence of key market players as well as suppliers and manufacturers in the industry. Major players like Sanofi and American Regent have introduced advanced delivery systems, such as voice-guided injectors and sulfite-free formulations, enhancing patient safety and broadening usage. Additionally, ARS Pharmaceuticals' USD 145 million global licensing agreement with ALK-Abelló signals the region's role in exporting innovation. With robust infrastructure, a strong presence of pharmaceutical manufacturers, and increasing awareness of anaphylaxis management, epinephrine market growth in North America is expected to remain high, reinforcing the region’s leadership in both product development and adoption.
Moreover, High epinephrine prescription rates and increasing adoption of epinephrine auto-injectors for emergency treatment of allergic reactions drives the epinephrine market growth in the region. Several product launches with improved formulations and delivery systems by major brands have augmented the market growth. Favorable reimbursement policies and high healthcare spending can boost sales in the region.
Asia Pacific has emerged as the fastest growing regional market for epinephrine globally driven by a combination of economic development and healthcare reforms. Countries like China, India, Japan and South Korea are witnessing rapid economic development that positively impacts the healthcare industry and increases affordability of new treatments. For instance, India alone accounts for over 12% of global asthma cases, creating a critical need for accessible and affordable emergency treatments. In response, awareness campaigns in hospitals and public institutions are promoting the timely use of epinephrine auto-injectors. Growing incidences of life-threatening medical conditions requiring epinephrine, rising patient awareness about availability of emergency drugs, and improving access can drive the market growth.
Governments across Asia are focusing on reducing out-of-pocket spending on healthcare through initiatives like Universal Health Coverage. In China, domestic manufacturers are offering low-cost generic epinephrine injectors, improving affordability and availability even in rural areas. Meanwhile, Japan and South Korea, have introduced subsidies for pediatric auto-injectors and are moving toward approval of needle-free delivery systems, such as nasal sprays, making emergency treatment safer and more user-friendly. These developments, combined with robust investment in healthcare infrastructure and rising allergy prevalence due to urbanization and pollution, are significantly contributing to the epinephrine market demand across the Asia Pacific region.
The United States is a leading market for epinephrine, driven by a high prevalence of severe food and drug allergies and strong public awareness of anaphylaxis treatment. Key legislation, like the School Access to Emergency Epinephrine Act, mandates the availability of auto-injectors in schools, boosting demand.
Widespread insurance coverage and a well-established emergency care system make epinephrine easily accessible. Major pharmaceutical players offer both branded and generic options, increasing affordability. With rising allergy rates, especially among children, the U.S. continues to be a key driver of epinephrine market demand.
In February 2026, Glenmark Pharmaceuticals Inc., USA, introduced its generic Epinephrine Injection USP, 10 mg/10 mL (1 mg/mL) multiple-dose vial in the United States. The product is bioequivalent and therapeutically equivalent to the reference drug from BPI Labs, LLC. This is further proliferating the epinephrine market demand.
China is seeing rising epinephrine market demand due to a growing burden of allergies and asthma, driven by urbanization and pollution. With millions affected by allergic conditions, the need for emergency treatments like epinephrine is increasing rapidly.
Government-backed healthcare reforms are improving access to essential medicines, while local generic manufacturing ensures affordable supply. Additionally, digital health investments and expanding emergency care services are raising awareness about anaphylaxis and promoting wider use of epinephrine auto-injectors. These factors position China as a key growth market in the global epinephrine landscape.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.64 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.0% | 2033 Value Projection: | USD 8.05 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Harvest Pharmaceuticals, Grand Pharma, Lincoln Medical, Ltd., Abbott Laboratories, Sanofi S.A., Mylan, Inc., ALK Abello A/S, Amedra Pharmaceuticals LLC, Impax, Emerade, Tianjin Jinyao Group, Sandoz Inc. , Bausch Health Companies Inc., Antares Pharma Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence of allergic diseases and conditions can drive the market growth. Allergic reactions can range from mild to severe and life-threatening. Epinephrine, also known as adrenaline, is the first line treatment for emergency medical situations involving severe allergic reactions or anaphylaxis. As more people around the world are developing sensitivities to various allergens such as food, drugs, insect bites and stings, there have been rising instances of anaphylaxis. This has contributed to greater awareness among the public as well as healthcare providers about the need to have immediate access to epinephrine auto-injectors, especially in developed markets.
Government-supported community health programs and school-based emergency response policies have been pivotal in driving adoption. For instance, in the United States, many states have laws requiring schools to stock epinephrine auto-injectors and train staff on their use—creating consistent demand for generic and branded epinephrine products. These regulations have significantly contributed to product uptake and wider public preparedness.
Traditionally, epinephrine for treating allergic reactions was only available in ampoules or vials that needed to be drawn up and administered via intramuscular injection. This posed several difficulties during emergency situations when seconds matter the most. The development of easy-to-use pre-filled epinephrine autoinjectors has revolutionized the management of anaphylaxis. Autoinjectors remove the need for drawing up medication from a vial and allow for hands-free self-administration through clothing, even by non-healthcare individuals.
Modern autoinjectors feature voice instructions, thus, making them suitable for use by adults as well as children experiencing symptoms. With their convenience and ease of administration, epinephrine autoinjectors have gained widespread acceptance among medical professionals and the public. Multiple device upgrades and new entrants by leading pharmaceutical companies have further expanded access and availability of these devices globally. This boosts demand for epinephrine, especially for products packaged in autoinjector formats.
Combination therapies are emerging as a promising growth avenue in the global epinephrine market, with the potential to significantly influence the epinephrine market forecast. As life sciences research advances, innovative drug delivery mechanisms are enabling more effective and patient-friendly treatment approaches. By combining epinephrine with complementary rescue medications, such as antihistamines or bronchodilators, manufacturers are creating synergistic formulations that enhance therapeutic outcomes and expand the scope of emergency care.
For instance, recent developments in combination inhalers allow for the self-administration of epinephrine during asthma exacerbations, offering faster symptom relief and greater convenience. Similarly, dual-action auto-injectors that combine epinephrine with antihistamines are showing promising results in treating severe allergic reactions, providing both immediate cardiovascular support and histamine suppression in one dose.
These advancements not only address co-occurring symptoms more effectively but also improve patient compliance, particularly in pediatric and geriatric populations. Leading pharmaceutical companies are actively investing in such multi-drug formats to broaden application areas and overcome limitations associated with traditional single-agent epinephrine products. As combination therapies gain regulatory traction and clinical acceptance, they are expected to play a pivotal role in shaping the future growth and innovation in the epinephrine market.
The epinephrine market value is currently undergoing a critical transformation, driven less by sheer volume and more by the urgency of unmet clinical needs, patent-driven competition, and technological advancements in drug delivery mechanisms. From an expert standpoint, this market is no longer just about emergency response, it is about accessibility, innovation, and regulatory dexterity.
A key concern that continues to dominate this landscape is the limited competition in the auto-injector segment, particularly in the United States. While Mylan’s EpiPen held near-monopoly status for over a decade, Teva’s generic launch in 2018 and subsequent market entries have still not meaningfully disrupted the entrenched brand loyalty or pricing strategies. Despite FDA approval of multiple generics, including those from Amneal and Adamis, access to affordable alternatives remains constrained due to production delays, insurance coverage limitations, and distribution inefficiencies. This is evidenced by periodic shortages that have occurred even as recently as 2023, creating anxiety among patient populations dependent on life-saving treatment.
In terms of innovation, the market is seeing a promising shift toward needle-free and digital-connected delivery systems, such as ARS Pharmaceuticals nasally administered epinephrine (neffy), which has gained increasing regulatory attention. If approved widely, this could redefine how patients manage anaphylaxis outside clinical settings, especially among pediatric and needle-phobic populations. It would also reduce the logistics and training burden traditionally associated with auto-injector usage.
Furthermore, intellectual property (IP) strategies are playing a central role in shaping competition. Companies continue to exploit device design patents to delay biosimilar and generic competition, effectively extending market exclusivity under the guise of innovation. This tactic, while legally sound, raises ethical and policy questions, particularly when price remains a barrier to access. Policy interventions, such as those recently observed with the U.S. Inflation Reduction Act's indirect effects on biologics, may gradually put downward pressure on device-based monopolies in the coming years.
*Definition: Global epinephrine market focuses on the sale of epinephrine (adrenaline) auto-injectors and prefilled syringes that are used for the emergency treatment of allergic reactions including anaphylaxis. The market has seen substantial growth in recent years due to the rising prevalence of food allergies as well as other forms of allergies worldwide. Major players in this market are focused on developing generic and branded epinephrine drugs with more user-friendly delivery mechanisms like auto-injectors to cater to the needs of the growing allergy patient population globally.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients