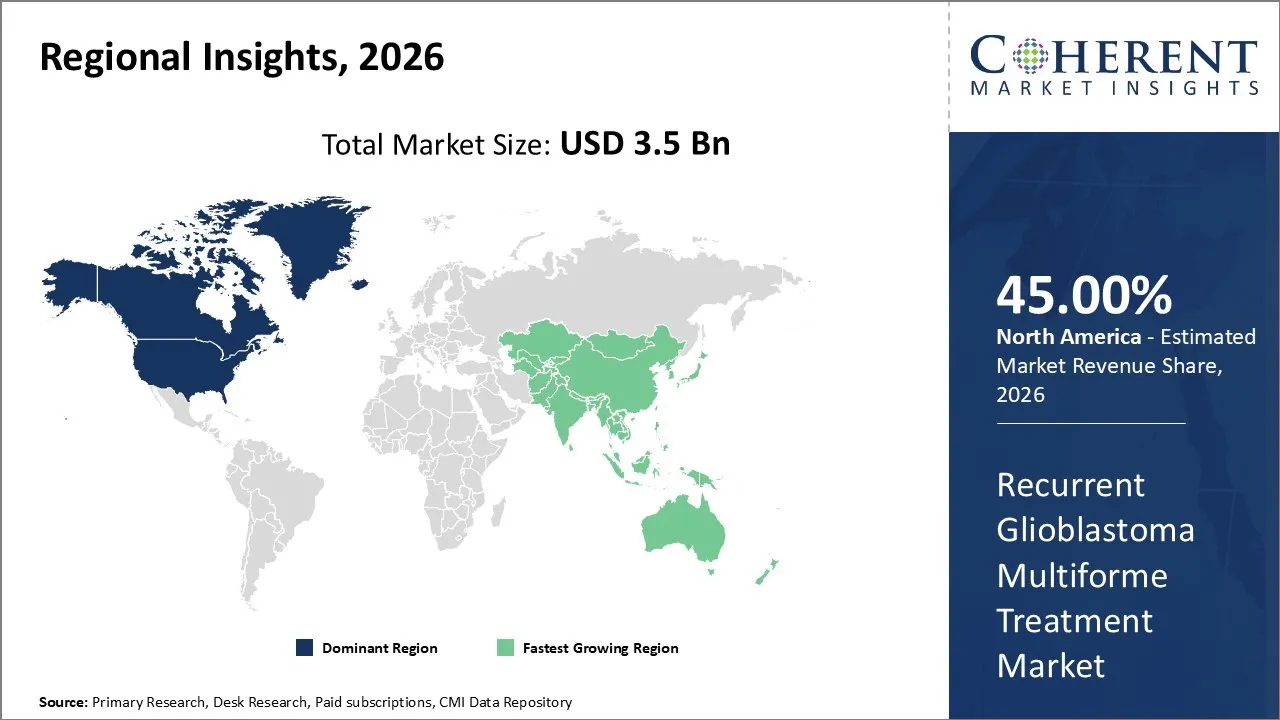
The recurrent glioblastoma multiforme treatment market was valued at USD 3.5 Bn in 2026 and is forecast to reach a value of USD 5.37 Bn by 2033 at a CAGR of 8.35% between 2026 and 2033.
Glioblastoma is a type of brain tumor that arises from the glial or supportive brain tissue called ‘glioma’. Glioblastoma multiforme (abbreviated as GBM) is an aggressive malignant brain tumor that has a very poor prognosis. The commercially available treatment option for this condition faces many challenges, including the crossing of blood brain barrier, which are limiting the concentration of drugs at the tumor site. Key companies in this market are actively focused on research & development activities to develop, and commercialize novel drugs for the treatment of such condition.
|
Current Event |
Description and its Impact |
|
FDA Clears New Clinical Trials for Innovative GBM Therapies |
|
|
Growth in Clinical Research and Trials in Asia-Pacific |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of treatment, the chemotherapy segment is expected to lead the market with 55% share in 2026. Chemotherapy has remained the main treatment for GBM due to decades of clinical research showing its effectiveness, both alone and combined with other therapies. Studies and treatment protocols consistently show that chemotherapy can slow tumor growth and improve patient survival, making it the cornerstone therapy for GBM globally.
Temozolomide, one of the most broadly used chemotherapeutic agents for GBM, has been extensively validated through numerous clinical trials as well as real-world applications, establishing a robust foundation for chemotherapy's market leadership position.
In terms of type, the grade IV segment is expected to hold 75% share of the market in 2026. Grade IV glioblastoma multiforme represents the most malignant form of brain tumor, characterized by rapid proliferation, extensive vascularization, as well as profound resistance to conventional therapies. The World Health Organization's classification system designates Grade IV as the most severe category, as well as clinical data from major cancer centers consistently demonstrates that approximately 80-85% of all glioblastoma cases fall into this category.
The dominance of this segment is fundamentally stimulated by the tumor's inherent biological characteristics, including its tendency to develop treatment resistance through multiple molecular pathways, necessitating continuous innovation in therapeutic approaches and creating sustained demand for specialized treatment protocols.
To learn more about this report, Request Free Sample
North America is expected to dominate the Recurrent Glioblastoma Multiforme Treatment market with 45% share in 2026, owing to its robust healthcare infrastructure, extensive research and development capabilities, as well as favorable regulatory environment that accelerates the approval and adoption of innovative recurrent glioblastoma multiforme treatments. The region's dominance is exemplified by the significant concentration of world-class medical institutions such as Mayo Clinic, Johns Hopkins Hospital, Memorial Sloan Kettering Cancer Center, etc., which serve as pioneers in developing and implementing cutting-edge therapeutic approaches for recurrent GBM.
For instance, in August 2025, a new clinical trial for patients with recurring glioblastoma multiforme (GBM), a very aggressive and hard-to-treat brain cancer, has been approved by the US FDA. Starlight Therapeutics, part of Lantern Pharma, said its new drug trial combining STAR-001 (LP-184) with the FDA-approved drug spironolactone has been approved. The trial will include adult patients whose GBM got worse after their first treatment.
Asia Pacific is expected to exhibit the fastest growth, owing to rapidly expanding healthcare capabilities as well as increasing investment in oncology infrastructure across key countries. The remarkable growth trajectory is exemplified by China's National Health Commission's recent approval of CAR-T cell therapy for brain tumors, marking a major milestone in locally developed advanced treatments.
Japan's Pharmaceuticals and Medical Devices Agency has accelerated approval pathways for innovative GBM treatments, resulting in faster market entry for novel therapeutic approaches developed by domestic companies like Takeda Pharmaceuticals and Daiichi Sankyo.
The U.S. contributes the highest share to the recurrent glioblastoma multiforme treatment market in North America due to its robust healthcare infrastructure, as well as extensive research capabilities, as well as the major presence of leading pharmaceutical companies actively developing innovative treatment modalities. The United States benefits from having premier cancer research institutions such as the National Cancer Institute (NCI), which has been instrumental in advancing glioblastoma research through initiatives like The Cancer Genome Atlas project that has offered crucial insights into GBM molecular characteristics.
For instance, in July 2025, , a biotech company working on new treatments for aggressive cancers, said that the U.S. FDA has given Orphan Drug Designation (ODD) to GLIX1. This drug is for treating malignant glioma, a serious type of brain cancer that includes glioblastoma.
Japan contributes the highest share in the recurrent glioblastoma multiforme treatment market due to its sophisticated healthcare infrastructure, advanced regulatory framework, as well as established pharmaceutical ecosystem that facilitates rapid adoption of innovative therapeutic approaches. The Japan Pharmaceuticals and Medical Devices Agency (PMDA) have implemented streamlined approval processes for oncology treatments, particularly for rare and aggressive cancers like recurrent glioblastoma multiforme, which has accelerated the availability of cutting-edge therapies including tumor treating fields (TTFields) and novel immunotherapeutic agents.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.5 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.35% | 2033 Value Projection: | USD 5.37 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
GlaxoSmithKline plc., AstraZeneca, F. Hoffman-La Roche, Ltd., Pfizer, Inc., Merck & Co., Inc., Vascular Biogeneics, AngioChem, Inc., Boehringer Ingelheim GmbH, Bristol-Myers Squibb Company, Boston Biomedical, Inc., Cantex Pharmaceuticals, Inc., Celldex Therapeutics, Inc., Cavion LLC, Coherus BioSciences, Inc., Eisai, Eli Lilly and Company, Cortice Biosciences, Inc., EnGeneIC Ltd., GenSpera, Inc., ERC Belgium SA, Genzyme Corporation, ImmunoCellular Therapeutics, Ltd., and GW Pharmaceuticals Plc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising incidence of glioblastoma multiforme (GBM), particularly recurrent glioblastoma multiforme, is significantly propelling the growth of treatment options worldwide. Glioblastoma, a highly aggressive and malignant brain tumor, poses substantial therapeutic challenges due to its rapid progression and high recurrence rate after initial treatment.
Increasing investment in oncology research and development (R&D) by pharmaceutical companies is significantly accelerating advancements in the treatment landscape for Recurrent Glioblastoma Multiforme (rGBM). Given the aggressive nature of rGBM and its poor prognosis, there remains an unmet medical need for more effective therapies beyond the current standard of care. Pharmaceutical companies are channeling substantial resources into exploring innovative treatment modalities such as immunotherapies, targeted therapies, and gene therapies that hold promise in enhancing patient outcomes.
The development of combination therapy approaches represents a significant opportunity in the treatment of Recurrent Glioblastoma Multiforme (rGBM), a notoriously aggressive and difficult-to-treat brain tumor. Current monotherapies, including surgery, radiation, and chemotherapy, have shown limited success in extending patient survival, primarily due to tumor heterogeneity and the blood-brain barrier’s impediment to drug delivery. Combination therapies, integrating modalities such as immunotherapy, targeted molecular agents, and standard chemoradiation, seek to address these limitations by simultaneously attacking multiple pathways involved in tumor growth and resistance.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients