The Gm1 Gangliosidosis Treatment Market is anticipated to grow at a CAGR of 36.7% with USD 194.2 Mn in 2026 and is expected to reach USD 1,658.9 Mn in 2033. The Global GM1 Gangliosidosis Treatment Market is gaining momentum due to high unmet clinical need, rare disease policy support, and progress in gene therapy and substrate-reduction therapies. GM1 gangliosidosis is a rare inherited lysosomal storage disorder caused by GLB1 gene mutations, leading to progressive nervous system damage. NIH MedlinePlus data published in 2023 estimates GM1 gangliosidosis frequency at 1 in 100,000 to 200,000 newborns, with Type I reported more frequently than other forms. The market is also supported by broader rare disease attention, as the WHO World Health Assembly update published in 2025 recognized that over 300 million people globally live with more than 7,000 rare diseases, most beginning in childhood. Current treatment remains largely supportive, creating demand for disease-modifying options. Clinical activity is advancing through gene therapy candidates such as PBGM01, which received FDA orphan, rare pediatric disease, and fast track designations, as reported in 2023 SEC filings. In addition, clinical-stage programs such as LYS-GM101, approved for adaptive clinical trial initiation in 2021, further indicate a developing treatment pipeline.
Source: ClinicalTrial.gov; Passage Bio
PBGM01 is projected to account for the largest share of cognitive systems spending in 2026, representing approximately 45.0% of the total volume. PBGM01 appears to be the dominant product type in the GM1 Gangliosidosis Treatment Market because it has the clearest late-stage clinical visibility among named candidates. According to ClinicalTrials.gov, the Imagine-1 Phase 1/2 study was listed as active, not recruiting, with 2026-02 shown as the estimated primary completion timing and 2029-02 as estimated study completion; the study evaluates a single intracisterna magna dose of PBGM01 for Type 1 and Type 2a pediatric GM1. Futhermmore, according to MedlinePlus Genetics, published in 2023, GM1 occurs in 1 in 100,000–200,000 newborns, and Type I is reported more frequently, supporting strong demand for infantile-focused therapies. Moreover, according to Passage Bio, Inc., in 2024, GEMMABio, a new therapeutics company, received exclusive worldwide PBGM01 development and commercialization rights, strengthening its 2026 pipeline continuity.
Source: ClinicalTrials.gov; MedlinePlus; Passagebio.com
To learn more about this report, Request Free Sample
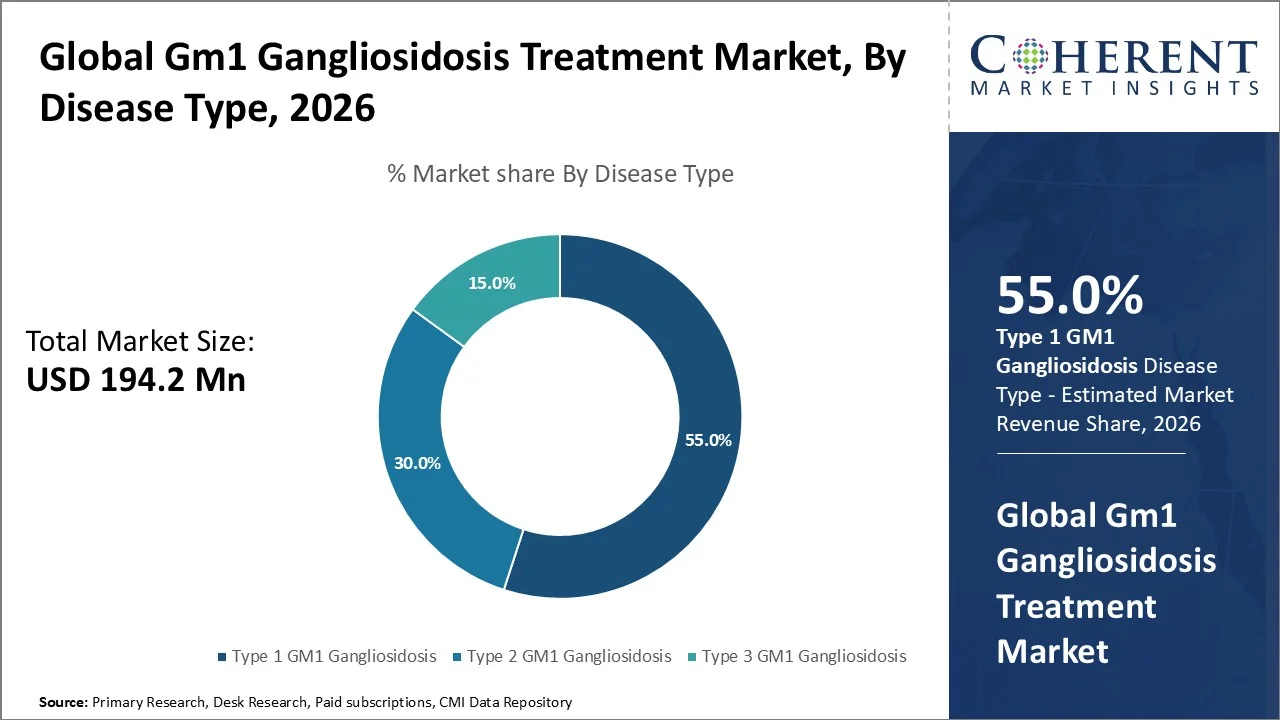
Based on disease type, type 1 GM1 Gangliosidosis dominate the market, accounting for a significant 55.0% share in 2026, Type 1 GM1 Gangliosidosis holds the largest share in the GM1 gangliosidosis treatment market due to its severe infantile onset, high clinical urgency, and stronger focus in gene therapy development. A 2026 NIH-indexed publication states that Type I is the most aggressive GM1 form, typically emerging before 6–12 months with hypotonia, neurodevelopmental regression, organ involvement, and rapid disease progression. This creates higher diagnostic attention, faster treatment-seeking, and stronger eligibility for early-intervention trials. ClinicalTrials.gov lists PBGM01 as a GM1 gene therapy designed to deliver a functional GLB1 gene copy to the brain and peripheral tissues, with evaluation in pediatric participants including early-onset infantile GM1. Type 1 therefore dominates because it represents the highest unmet need, greatest mortality risk, and clearest target population for disease-modifying therapies, encouraging companies and regulators to prioritize infantile GM1 programs. Moreover, in 2026, Azafaros B.V., biotechnology company also reported Phase 3 development activity for nizubaglustat in GM1/GM2 gangliosidoses. These factors make Type 1 the priority segment for diagnosis, trial enrollment, and disease-modifying therapy development.
Increasing research and development is driving the GM1 gangliosidosis treatment market size by shifting the field from supportive care toward disease-modifying therapies, especially gene therapy and oral substrate-reduction approaches. For instance, in 2026, FDA’s Rare Disease Innovation Hub agenda prioritized rare-disease regulatory science, novel endpoints, and biomarker development, supporting faster evaluation of ultra-rare genetic therapies. ClinicalTrials.gov also lists active GM1 programs, including PBGM01 gene therapy, designed to deliver a functional GLB1 gene to the brain and peripheral tissues. Moreover, in January 2026, Azafaros B.V., biotechnology company reported that two pivotal Phase 3 studies of nizubaglustat for GM1/GM2 gangliosidoses and NPC were enrolling, with data expected in 2027. These developments expand pipeline visibility, attract rare-disease investment, and strengthen future commercialization potential.
Rising inorganic strategies such as licensing and collaborations are strengthening the GM1 gangliosidosis treatment market by helping small biotech innovators share clinical, regulatory, and commercialization risk. For instance, in 2026, the New England Journal Medicines-published Phase 1–2 study on AAV9 gene therapy for Type II GM1 gangliosidosis showed the value of collaboration-led development. The program involved academic and government-linked research groups, including NHGRI/NIH, UMass Chan Medical School, and Auburn University, after earlier commercial sponsorship challenges. This supports the trend that partnerships and licensing models help sustain ultra-rare gene therapy programs where small patient pools create high development risk. Furthermore, in August 2024, Passage Bio, biotech company out-licensed PBGM01 for GM1 gangliosidosis to GEMMA Biotherapeutics with exclusive worldwide development and commercialization rights, showing how licensing helps continue rare pediatric gene therapy programs. Moreover, in 2026, the AAV9/GLB1 gene therapy study for Type II GM1 showed clinical progress from a long academic-government collaboration involving NHGRI/NIH, UMass Chan Medical School, and Auburn University.
Growing early diagnosis through newborn screening and increasing investment in rare disease research and development has created significant opportunity for the Gm1 gangliosidosis treatment market growth over forecast period. Growing early diagnosis through newborn screening and higher rare-disease R&D investment can drive the GM1 gangliosidosis treatment market by expanding the identifiable patient pool before irreversible neurological damage occurs. For instance, in March 2026, HRSA: Health Resources and Services Administration stated that U.S. newborn screening is state-based and most states screen for some or all RUSP conditions, while RUSP inclusion depends on screening benefit, state feasibility, and effective treatment availability. This supports future GM1 screening readiness as therapies advance. Furthermore, in 2026, NIH/NCATS (NCATS- National Center for Advancing Translational Sciences – NIH) reported that more than 10,000 rare diseases affect millions in the U.S., and around 30% of children with rare diseases die before age five, strengthening urgency for early diagnosis and R&D. Azafaros B.V. company’s 2026 Phase 3 nizubaglustat program for GM1/GM2 further validates commercial and clinical momentum.
|
Current Event |
Description and its Impact |
|
FDA Rare Disease Innovation Hub Strategic Agenda 2026 |
|
|
FDA Plausible Mechanism Framework for Ultra-Rare Genetic Diseases |
|
|
NIH/NCATS Rare Disease Research and Gene Therapy Collaboration Push |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
North America account 40.0% market share in 2026, supported by advanced infrastructure for rare disease research, strong U.S. regulations, and increasing government initiatives. North America dominates the GM1 gangliosidosis treatment market due to strong U.S. rare-disease regulation, clinical trial infrastructure, newborn screening systems, and gene therapy R&D. For instance, in 2026, U.S.FDA’s Rare Disease Innovation Hub released a strategic agenda to advance rare-disease regulatory science and improve coordination across drug, biologic, and device centers, supporting therapies for small patient groups such as GM1. Furthermore, in 2026, NIH/NCATS reported that more than 10,000 rare diseases affect millions of Americans and around 30% of children with rare diseases die before age five, strengthening funding urgency. The U.S. also has broad early-diagnosis capacity, as Health Resources and Services Administration (HRSA) states newborn screening reaches nearly 4 million babies each year. ClinicalTrials.gov lists U.S.-linked PBGM01 gene therapy for GM1, designed to deliver a functional GLB1 gene copy to brain and peripheral tissues. These factors support faster diagnosis, trial recruitment, and therapy commercialization.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033. Asia Pacific is the fastest-growing region in the GM1 gangliosidosis treatment market due to expanding rare-disease diagnosis, newborn screening, and government-backed genetic medicine infrastructure. According to India’s PIB, in 2026, the government highlighted early detection, affordability, genomics, biotechnology, and preventive healthcare as priorities for genetic and rare diseases. This directly supports diagnosis-led demand for GM1 therapies. According to Lok Sabha data published in 2026, India provides financial support of up to Rs. 50 lakhs per rare-disease patient under NPRD 2021, improving treatment access. Clinical readiness is also rising, as ClinicalTrials.gov lists PBGM01’s Phase 1/2 study with February 2026 as the estimated primary completion date. MedlinePlus Genetics reports GM1 occurs in 1 in 100,000–200,000 newborns, and Type I is reported more frequently, supporting strong pediatric treatment need.
The U.S. dominates the North America GM1 gangliosidosis treatment market due to its stronger rare-disease regulatory ecosystem, clinical-trial infrastructure, and active gene-therapy pipeline. For instance, in April 2026, U.S.FDA’s Rare Disease Innovation Hub released its Strategic Agenda to improve rare-disease regulatory science and coordination across FDA centers, especially for small patient populations with high unmet need. Furthermore, in 2026, NIH/NCATS reported that over 10,000 rare diseases affect millions of Americans, and nearly 30% of children with rare diseases die before age five, supporting urgent R&D funding. ClinicalTrials.gov lists PBGM01, a GM1 gene therapy designed to deliver a functional GLB1 gene copy to the brain and peripheral tissues. In addition, in 2026, Azafaros B.V. reported Phase 3 nizubaglustat studies for GM1/GM2 gangliosidoses, further strengthening U.S.-linked rare-disease development momentum.
China dominates Asia Pacific due to its stronger rare-disease policy, large diagnosed pool, and faster drug-access reforms. According to China’s State Council, in 2025, China expanded its rare-disease catalog to 207 conditions, built a national network of 419 medical institutions, and registered 1.64 million rare-disease cases across a reporting system covering 1.15 billion people. In 2026, China revised drug regulations to give market exclusivity to eligible pediatric and rare-disease drugs, supporting GM1 therapy development and faster market entry.
Some of the major key players in Global Gm1 Gangliosidosis Treatment Market are Axovant Gene Therapies Ltd, Passage Bio and Lysogene.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 194.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 36.7% | 2033 Value Projection: | USD 1,658.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Axovant Gene Therapies Ltd, Passage Bio and Lysogene. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients