The health supplementary food OEM and ODM market is anticipated to grow at a CAGR of 7.4% with USD 67.0 Bn in 2026 and is expected to reach USD 110.5 Bn in 2033. The market is growing as people are getting chronic lifestyle diseases (1.3 Bn people globally), and many brands prefer to have their products made by other companies instead of building expensive factories themselves. Using OEM (Original Equipment Manufacturer) and ODM (Original Design Manufacturer) models has become an important strategy in the health supplement industry. These models let brands make new as well as unique products quickly without spending a lot of money on production.
Diet Supplement is projected to account for the largest share of nutrition type in 2026, representing approximately 55% of the total volume. Owing to its deeply rooted consumer health trends, lifestyle changes, as well as increasing awareness about personalized nutrition. Obesity is a chronic disease that affects over one billion people in the world and is a major risk factor for cardiovascular disease, type 2 diabetes, as well as cancer.
Diet Supplements, which include vitamins, minerals, herbal extracts, and performance enhancers, have gained high acceptance due to their ability to target specific health needs such as weight management, energy boosting, immunity enhancement, chronic disease prevention, etc.
For example, government health programs, like the U.S. NIH Office of Dietary Supplements, say micronutrients are important for good health. This has made more people want products that give real nutritional benefits. The government of India initiated national programs to prevent, control and combat these deficiencies and their serious consequences. The interventions involved distribution of iodized salt in the endemic areas, administration of semiannual massive dose of vitamin A to young children, and distribution of iron-folic acid tablets to the vulnerable groups.
To learn more about this report, Request Free Sample
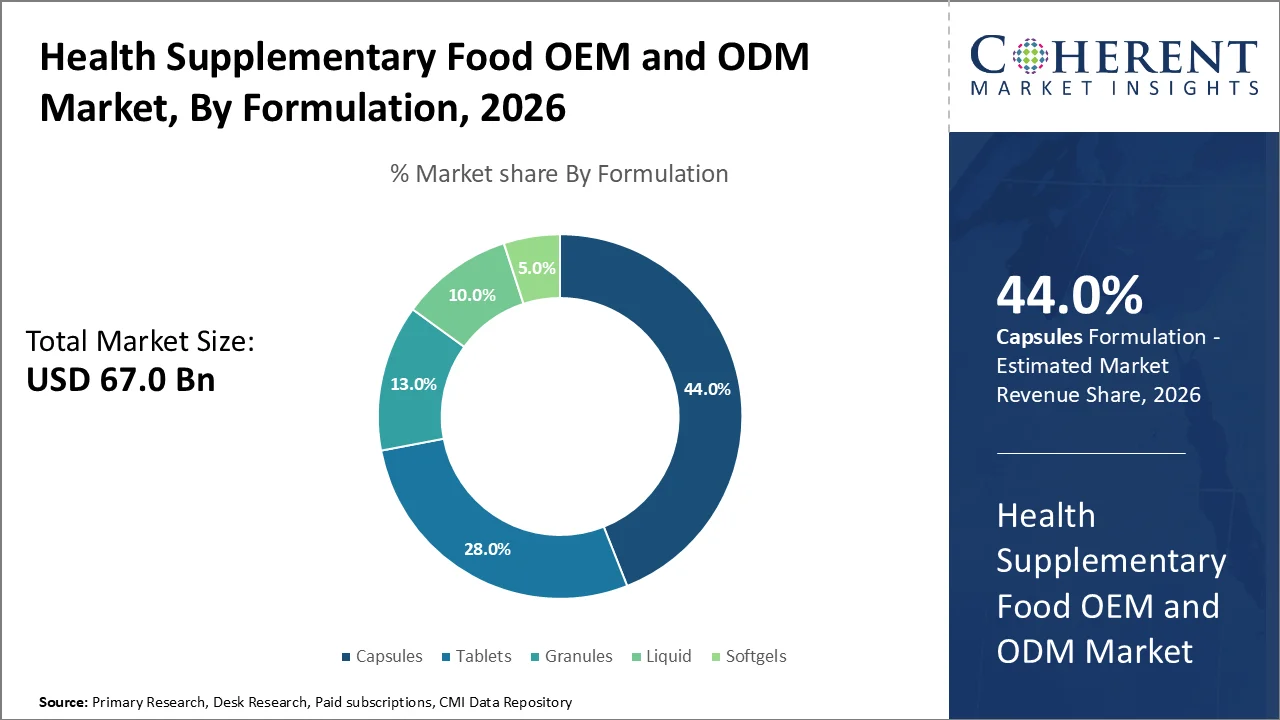
Based on formulation, capsules dominate the market, accounting for a significant 44% share in 2026, primarily due to their widespread consumer acceptance, ease of manufacturing, as well as ability to encapsulate a variety of supplement formulations that are either sensitive to air and light or have unpleasant tastes and odors. Capsules, especially gelatin and vegetarian variants, provide an effective barrier that protects active ingredients from oxidation as well as degradation, thereby improving product stability and shelf-life.
In October 2025, Lonza Capsugel said they are launching a new Organicaps™ capsule at SupplySide Global 2025. It is the only plant-based, USDA organic, quick-release capsule made in North America. Featuring a high oxygen barrier, non-reactive shell, as well as excellent polymer stability, Organicaps™ capsules are designed to help brands stay one step ahead with organic product claims in the growing organic supplements market.
This is particularly important for supplements containing probiotics, oils rich in omega-3 fatty acids, or volatile herbal extracts. Health organizations such as the U.S. Food and Drug Administration (FDA) has clear rules for capsules, making them easy to use and accepted in many markets, especially in North America and Europe, which are usually leaders in health supplements.
Based on packaging type, bottle packaging dominates the market, accounting for a significant 45% share in 2026. Bottle Packaging has held its dominance primarily due to its strong alignment with consumer preferences for health supplements that needs careful dosing as well as secure storage. The durability along with reusability of bottles makes them a perfect choice for supplements such as vitamins, minerals, herbal extracts, etc., where preservation from air, moisture, as well as contaminants is critical.
Additionally, bottles allow for controlled dispensing caps, droppers, or measuring spoons can be integrated, enabling precise dosage which is vital in health supplements. For instance, major supplement manufacturers such as Nature’s Bounty and Garden of Life package their multivitamins as well as specialty supplements predominantly in plastic or glass bottles, showing trust in this format.
Advanced technologies like automated capsule filling, continuous tablet manufacturing, robotic packaging, as well as AI-driven production optimization are transforming supplement production. Leading companies such as Lonza Group, Catalent, Thorne Health, etc., have adopted these systems, achieving high-speed capsule filling, continuous solid dosage manufacturing, and precise robotic dosing.
The impact of these technologies is significant: production time is lowered by 40–60%, dosage accuracy reaches 99.8%, labor costs drop by 35%, as well as risks of human error and contamination are minimized. This inclination is driving faster, safer, as well as more efficient manufacturing in the health supplement industry.
3D printing is enabling personalized supplements with custom shapes, dosages, and complex release profiles. Multi-material printing allows combination products in a single tablet. Companies like FabRx, Aprecia Pharmaceuticals, and Multiply Labs are leading the way, making tailored vitamins, rapidly dissolving tablets, as well as robotic 3D-printed supplements.
Using 3D printing has many benefits: it lets companies make custom products for many people, makes testing new designs twice as fast, as well as creates new types of doses that were not possible before. Making products only when needed also reduces storage and makes supplement production easier and faster.
|
Current Event |
Description and its Impact |
|
FDA Updates Dietary Supplement Regulations (2026) |
|
|
EU Novel Foods Regulation Expansion (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
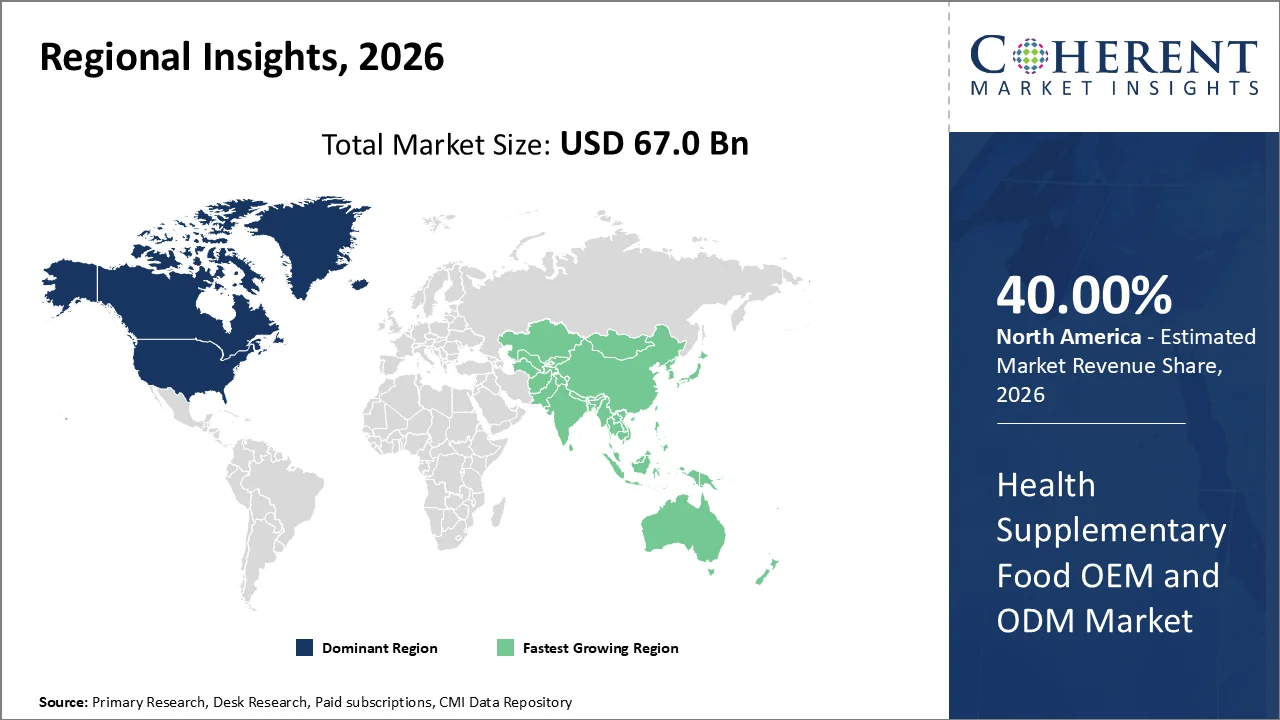
North America account 40% market share in 2026, owing to its well-established healthcare infrastructure, stringent regulatory framework, as well as high consumer awareness regarding nutritional supplementation. According to the CHPA Survey, 92% Say Supplements Have Had a Positive Impact on Their Health. 83% of voters use dietary supplements to support their wellness needs.
The region benefits from the presence of major pharmaceutical as well as nutraceutical companies that have established robust OEM and ODM partnerships with supplement manufacturers.
The U.S. FDA has clear rules for dietary supplements under the DSHEDA law, making it easier for OEM and ODM companies to operate. These rules have encouraged many contract manufacturers to open factories in states like California, New York, and Texas.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. Driven by the rapid expansion of domestic manufacturing capabilities as well as increasing consumer awareness about health and wellness. This accelerated growth is fueled by high investments from local government authorities in biotechnology as well as nutraceutical manufacturing infrastructure, particularly in countries like China, India, South Korea, etc. the Singapore government have committed USD28 billion under the RIE2025 plan, with a strong pipeline of translational as well as AI-driven biomedical sciences initiatives expected under RIE2030.
China's 13th Five-Year plan gave a lot of support to build local health supplement factories, helping companies like By-Health and Infinitus create top-quality OEM and ODM facilities. In India, the PLI program for medicines helped the nutraceutical OEM sector grow, and companies like Hindustan Unilever and Dabur increased their contract manufacturing.
The U.S. leads the market because of strong research and development. Groups like the NIH and universities research supplements and help create new products. The American Botanical Council and the Council for Responsible Nutrition give advice to help companies follow rules and work together, making the industry more professional and bringing in business.
Additionally, the U.S. has strong supply chains that link raw materials from countries like China and India with local manufacturers who make as well as package products for sale in North America.
The proximity to major population centers as well as established distribution channels through retailers like CVS, Walgreens, Whole Foods Market, etc., creates compelling value propositions for brands seeking comprehensive OEM and ODM services that extend beyond manufacturing to include packaging, labeling, along with logistics support.
China contributes the highest share in the Health Supplementary Food OEM and ODM market in APAC owing to its well-established manufacturing infrastructure as well as comprehensive supply chain ecosystem. The dominance of the country is supported by its massive production capacity, with over 3,000 certified health supplement manufacturing facilities as reported by the China Food and Drug Administration (CFDA).
China's leadership position is supported by its advanced pharmaceutical as well as nutraceutical manufacturing capabilities, supported by Special Economic Zones like Guangdong and Jiangsu provinces that house numerous international OEM and ODM operations.
The Chinese government's "Made in China 2025" initiative has majorly boosted the country's pharmaceutical as well as health supplement manufacturing sector, with state-backed investments exceeding USD50 billion in biotechnology and pharmaceutical infrastructure development.
Some of the major key players in Health Supplementary Food OEM and ODM Market include, Cosmax Inc., Kolmar Korea, Japan Supplement Foods Co., Ltd., Health Sources Nutrition Co., Ltd., Goerlich Pharma GmbH, API Co., Ltd., Syngen Biotech Co., Ltd., OriBionatureSdnBhd, Nutricare Co.,Ltd, and NOVAREX Co., Ltd
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 67.0 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.4% | 2033 Value Projection: | USD 110.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cosmax Inc., Kolmar Korea, Japan Supplement Foods Co., Ltd., Health Sources Nutrition Co., Ltd., Goerlich Pharma GmbH, API Co., Ltd., Syngen Biotech Co., Ltd., OriBionatureSdnBhd, Nutricare Co.,Ltd, and NOVAREX Co., Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients