Global immuno-oncology drugs market is estimated to be valued at USD 38.40 Bn in 2026 and is expected to reach USD 128.23 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 18.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Global immuno-oncology drugs market growth is driven by increasing adoption of immune checkpoint inhibitors across various cancer types. Biomarkers and combination therapies have gained popularity, and can drive the market growth in coming years. Further technological advancements to develop novel drug candidates with improved efficacy and faster regulatory approvals are projected to drive the immuno-oncology drugs market during the forecast period.
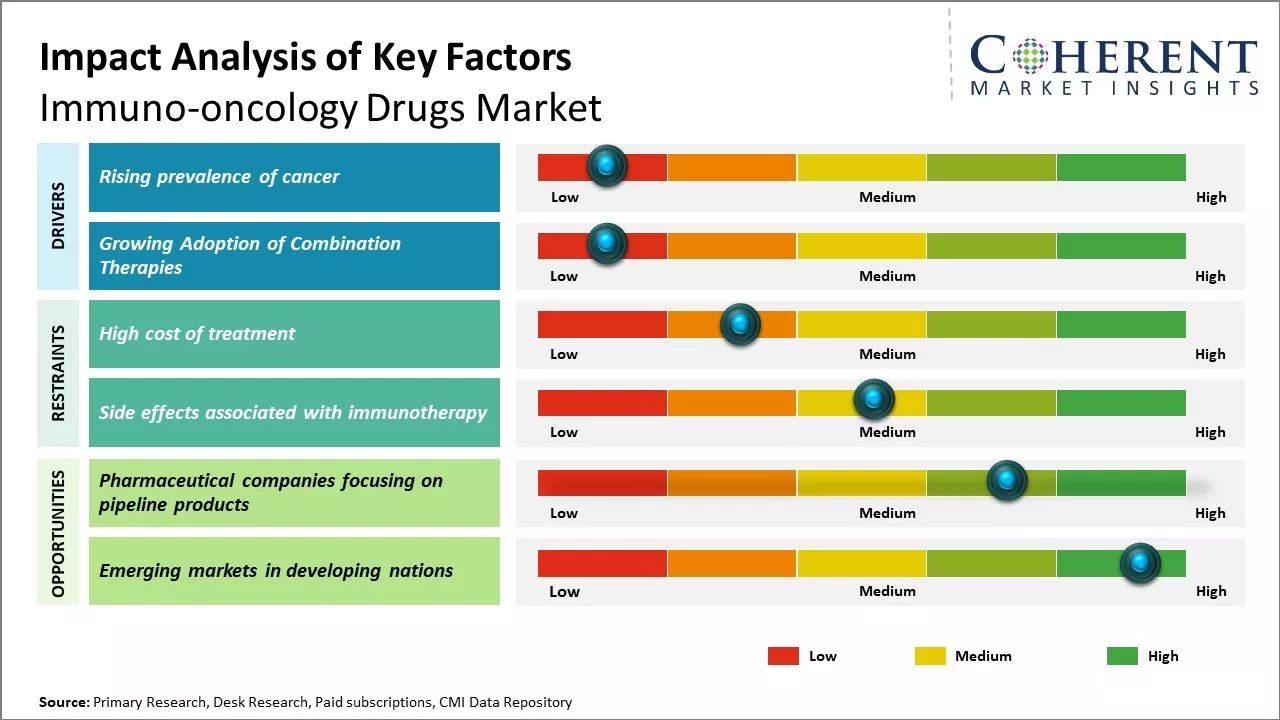
Rising prevalence of cancer
Rising prevalence of various types of cancers across the world can drive the global immuno-oncology drugs market growth. Cancer has become one of the leading causes of mortality worldwide. According to the data published by WHO, cancer burden is expected to grow exponentially in the future with the global cancer burden expected to reach about 30 million new cancer cases and over 16 million cancer deaths by 2040. This increasing incidence of cancer poses serious public health challenges. The prognosis of many cancers still remains poor due to limited treatment options. Due to significant advancements in immunotherapy and immune-oncology drugs, the treatment landscape of cancer is transforming. Immuno-oncology involves harnessing the power of the patient's own immune system to fight cancer, and it emerges as one of the most promising areas of research in oncology. Drugs which enhance the immune system's ability to recognize and destroy cancer cells like checkpoint inhibitors, monoclonal antibodies, cytokine therapies are demonstrating durable clinical responses in several cancer types including lung cancer, melanoma, kidney cancer, head and neck cancer. For instance, according to Cancer Australia's 2023 Report, approximately 162,163 cancer cases were diagnosed in Australia in 2022. This significant incidence of cancer cases boosted demand for point-of-care diagnostics. According to the data from the International Agency for Research on Cancer, in 2022, there were approximately 8,600 cases of liver cancer, and this will increase to 11,200 cases by 2030 in Mexico.
Get actionable strategies to beat competition: Request Free Sample
Growing Adoption of Combination Therapies
There has been increased use of combination immunotherapies aimed at targeting multiple immune checkpoints during cancer treatment. Combination therapies have emerged as a promising strategy to optimize clinical response rates and combat resistance to monotherapies. Early studies have shown that combined blockade of PD-1 and CTLA-4 improves survival outcomes over single-agent therapies for many cancer types. Combination regimens are also more effective than sequential therapies in eliciting durable anti-tumor responses. With initial positive clinical data, combination strategies are expected to become first-line options for cancer indications. Drug manufacturers are actively pursuing combination therapies through collaborations and pipeline expansion focused on dual or triple immune checkpoint inhibitors. The clinical benefits of combination immunotherapies over single agents drives physicians and patients to increasingly adopt such treatment approaches.
Key Takeaways from Analyst:
Researchers are continuously developing innovative immune checkpoint inhibitors and cancer vaccines that show promising results in clinical trials. North America currently dominates the market due to growing prevalence of cancer and established healthcare infrastructure that enables speedy approval and adoption of premium-priced immuno-oncology drugs. However, Asia Pacific is expected to emerge as fastest growing region due to rising healthcare spending and increasing awareness about cancer treatment alternatives like immuno-oncology.
Scientific advancements leading to discovery of new immuno-oncology drug candidates and combination therapies can drive the market growth. Successful clinical trial outcomes of immuno-oncology drugs for treatment of various cancer types can also drive the market expansion. However, high costs of these drugs can hamper its adoption. Moreover, gaps in understanding of complex interaction between tumors and immune system can limit the pipeline of novel therapies.
Key players are investing in R&D and focusing on developing off-patent drugs and biosimilars to make treatments more affordable. Partnerships between pharmaceutical companies and clinical research organizations can help expedite clinical trials and accelerate commercialization of promising drug candidates.
Market Challenges: High cost of treatment
The high cost of treatment can hamper the global immuno-oncology drugs market growth. Immunotherapy drugs that mobilize the body's immune system to fight cancer cells are considered as one of the major breakthroughs in cancer treatment. However, high cost of these drugs makes them out of reach of many patients globally. Immuno-oncology drugs often require prolonged treatment over months or years, which makes their overall cost of therapy significantly higher than traditional cancer drugs. For instance, according to a study published by American Society of Clinical Oncology in 2021, the monthly cost of popular immuno-oncology drugs like Keytruda and Opdivo ranges between US$ 12,500 to US$ 15,000. This high monthly cost poses a significant financial burden for majority of patients worldwide. Most public and private health insurance programs find it difficult to cover the complete cost of such expensive treatments. The high treatment cost is especially a roadblock in developing nations where affordability is a major concern. As per data published by World Health Organization in 2022, nearly 70% of all annual cancer deaths occur in low and middle-income countries but most national health budgets can barely afford the high cost of immunotherapy.
Market Opportunities: Pharmaceutical companies focusing on pipeline products
Focusing on pipeline products can offer opportunity for pharmaceutical companies in the global immuno-oncology drugs market. Immuno-oncology is an evolving field with significant potential as these therapies work to harness the body's own immune system to fight cancer in a more targeted manner than traditional therapies. There are many late-stage pipeline drugs currently in clinical trials that can drive the market growth. Some of the most advanced pipeline programs are in combination therapies, as combining different mechanisms, like immune checkpoint inhibitors with other immunotherapy drugs, has shown great promise in treating various cancer types more effectively than single-agent treatments. Many large pharmaceutical companies have extensive pipelines of combination therapies being tested, such as checkpoint inhibitors paired with vaccines, cellular therapies, small molecule targeted therapies, and other immune modulators. The success of these ongoing Phase III clinical trials over the next few years can drive the immuno-oncology drugs market growth.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
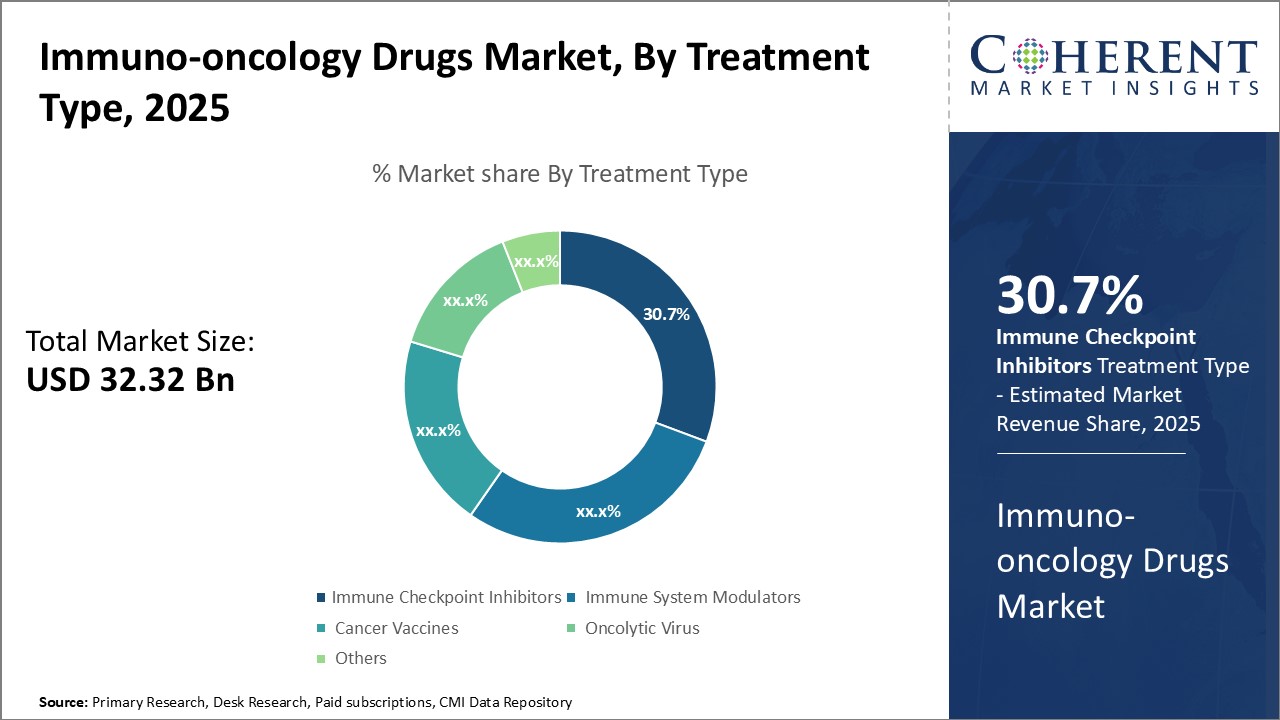
By Treatment Type - Growing prevalence of cancer boosts demand for immune checkpoint inhibitors
In terms of treatment type, immune checkpoint Inhibitors segment is estimated to contribute the highest market share of 31.7% in 2026, owing to increasing prevalence of different types of cancers globally. Immune checkpoint inhibitors work by removing brakes on the immune system T-cells to enhance their anti-tumor response and have emerged as a transformational treatment for various cancer types. Compared to other treatment types, checkpoint inhibitors provide long lasting response and have revolutionized cancer treatment. Their ability to activate the body's own immune system against cancer cells has increased their demand significantly. The success of immune checkpoint inhibitor drugs like Keytruda and Opdivo in clinical trials for cancers like melanoma, lung cancer and renal cell carcinoma has boosted physician and patient confidence in these drugs. Moreover, the approval of these drugs for wider cancer indications can drive the segment growth
To learn more about this report, Request Free Sample
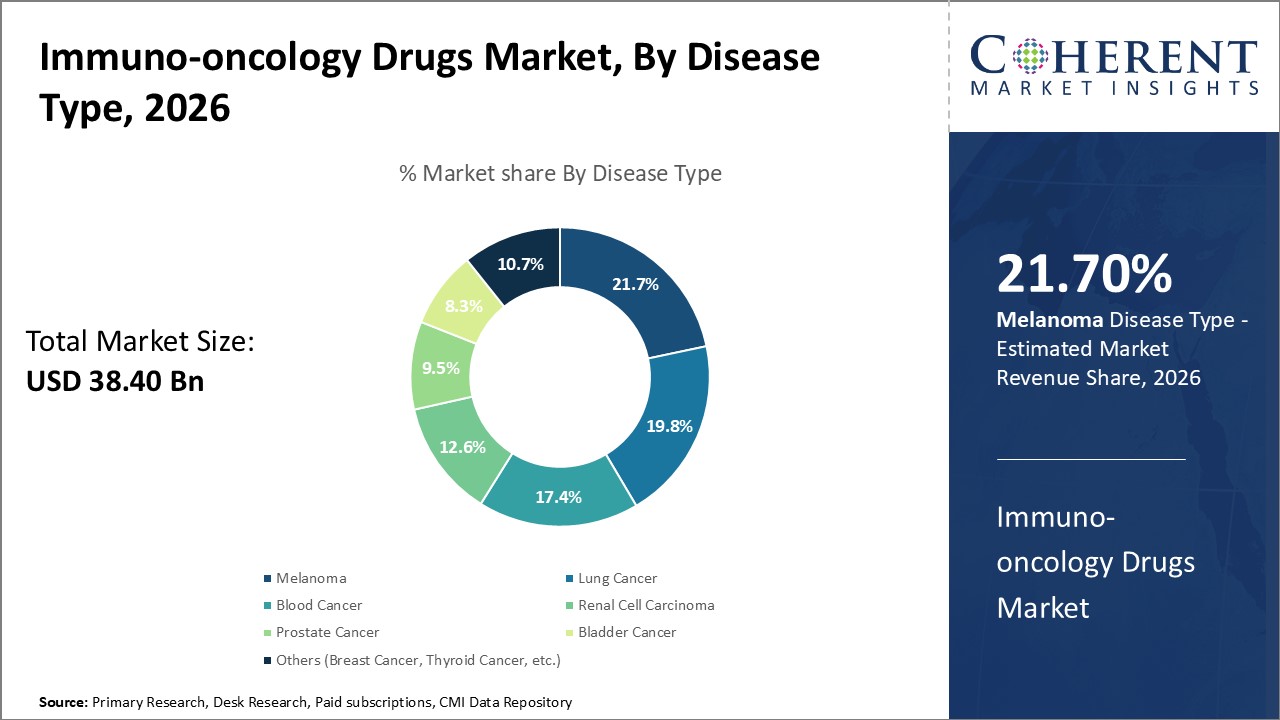
By Disease Type - Increased diagnosis boosts demand for treatments targeting lung cancer
In terms of disease type, lung cancer segment is estimated to contribute the highest market share of 21.7% in 2026. Lung cancer remains the leading cause of cancer-related deaths worldwide due to lack of early diagnostic methods for most types of lung cancers. However, growing awareness and screening programs have led to increased diagnosis of lung cancer at early stages. This has boosted demand for more effective targeted therapies with improved outcomes. Novel immunotherapies including immune checkpoint inhibitors have shown encouraging efficacy results in lung cancer, with better survival benefits than chemotherapy. Drugs like pembrolizumab and nivolumab have become the standard first-line treatment options for advanced non-small cell lung cancer. Rising incidence of lung cancer globally due to increasing tobacco consumption can drive the segment growth.
By Distribution Channel - Specialized services boost uptake through hospital pharmacies
In terms of distribution channel, hospital pharmacies segment is estimated to contribute the highest market share of 41.5% in 2026. Hospital pharmacies play a very important role in oncology treatment as most immuno-oncology drugs require specialized storage, handling and administration techniques. These also provide additional services like customized pharmacy adherence packaging and coordination with insurance. Moreover, a majority of cancer patients receive immunotherapy as part of an inpatient prescription during their hospital visit for chemotherapy or surgery. This helps hospitals to emerge as the most accessible and convenient point of procuring immuno-oncology drugs as compared to other channels. Several health systems are also investing in infusion centers and specialized cancer clinics within hospitals to provide patient-centric services. This has boosted the preference of hospital pharmacies over retail and online channels for procuring immuno-oncology drugs.
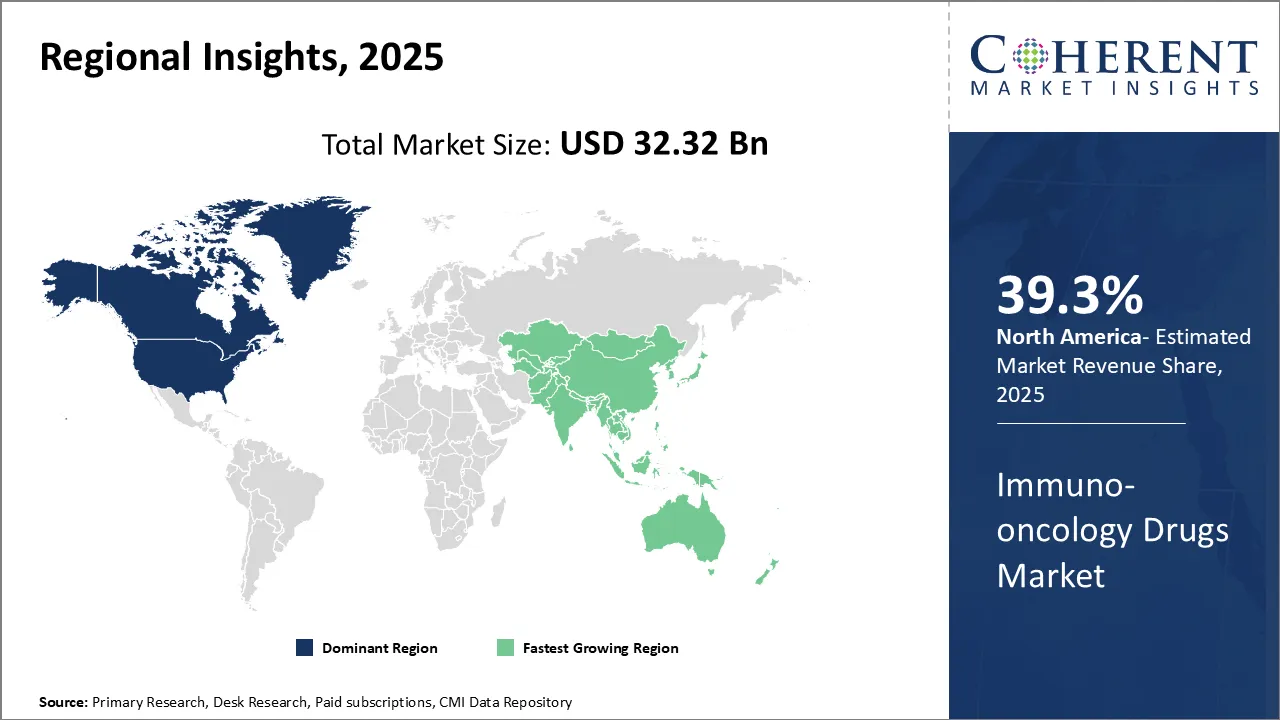
Need a Different Region or Segment? Request Free Sample
North America currently dominates the global immuno-oncology drugs market with an estimated market share of 39.3% in 2026. The U.S. is a major market for immuno-oncology drugs, owing to factors such as rising disposable income, increasing prevalence of cancer, and growing acceptance of immunotherapy in recent years among patients and healthcare professionals. Moreover, robust presence of global pharmaceutical players such as Merck, Bristol-Myers Squibb, and Roche have strengthened the supply chain of IO drugs in the region.
Asia Pacific is the fastest growing region. Rapid economic development and improving standards of living have enhanced the affordability of immuno-oncology therapies in several developing countries of Asia such as China, India, South Korea and Japan. The sizable population base also presents a massive patient pool undergoing cancer treatments. Rising medical tourism and growing penetration of health supports the proliferation of IO therapies in private healthcare settings across urban regions. Local generics manufacturers have further expanded the access of low-cost IO combination therapies, while foreign drugmakers are augmenting their presence through strategic partnerships with regional biotech firms.
Immuno-oncology Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 38.40 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 18.8% | 2033 Value Projection: | USD 128.23 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Coloplast Corp, Bristol Myers Squibb Company, Novartis AG, F. Hoffmann-LA Roche Ltd., Merck & Co., Inc., GSK Plc., Eli Lilly and Company, Fresenius Kabi AG, Pfizer Inc., AbbVie Inc., Genentech Inc., Sanofi, AstraZeneca, Bayer AG, Bluebird Bio, Inc., Regeneron Pharmaceuticals, Inc., Amgen Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients