The global intraosseous infusion devices market is expected to grow from USD 600.1 Mn in 2026 to USD 972.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.1% from 2026 to 2033. The market for intraosseous infusion devices is poised for significant expansion, fueled by the rising number of trauma-related injuries, cardiac emergencies, and critical care instances that demand quick vascular access.
According to the World Health Organization, of the 4.4 million injury-related deaths, unintentional injuries take the lives of 3.16 million people every year and violence-related injuries kill 1.25 million people every year. Roughly 1 in 3 of these deaths result from road traffic crashes, 1 in 6 from suicide, 1 in 10 from homicide and 1 in 61 from war and conflict.
(Source: World Health Organization)
Manual is projected to hold a market share of 43.1% in 2026, because of its reduced costs, easy operating procedure, and universal application in emergencies and other limited resources. Such devices have great popularity among ambulances, military healthcare centers, and healthcare facilities that cannot afford advanced equipment. Manual devices involve less maintenance, use no batteries or charge, which makes them convenient for emergency situations. For instance, Cook Medical Dieckmann Intraosseous Infusion Needle is created in order to ensure fast and efficient intraosseous access for infusion of liquids and drugs in emergencies where IV access is either difficult or delayed. (Source: Cook Medical) The sustained availability of cost-effective manual intraosseous systems are aiding their widespread uptake globally.
To learn more about this report, Request Free Sample
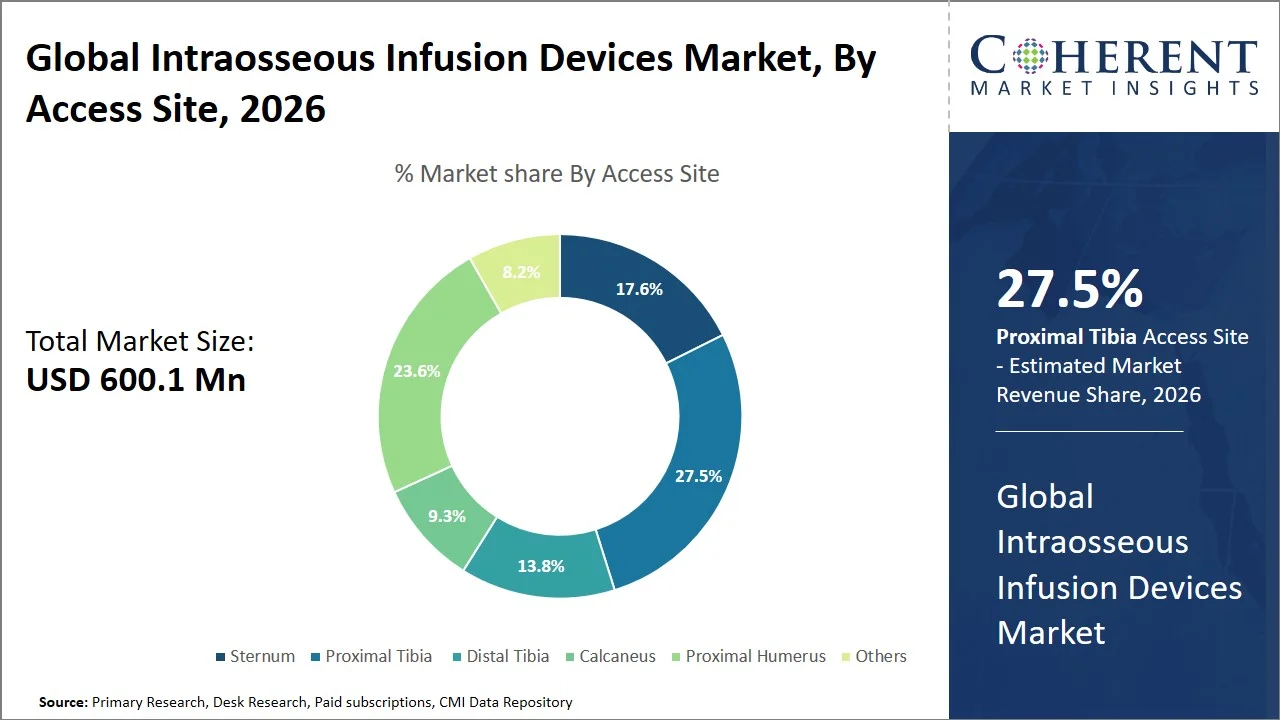
Proximal tibia is projected to hold a market share of 27.5% in 2026, attributed to the ease of anatomic access, first-pass success rate, and rapid insertion in emergency cases, like cardiac arrest, injury, and dehydration cases. This site presents little soft tissue coverage and landmark identification, making it possible for the clinicians to gain immediate vascular access irrespective of the condition of the patient. According to research published by ResearchGate, proximal tibial intraosseous (IO) access proves more convenient and efficient as compared to humeral access, especially for paramedics and in emergency care scenarios. (Source: ResearchGate)
Furthermore, the proximal tibia makes up the commonest choice in emergency care centers and emergency medical technician services due to reduced risks of needle dislodgment and reliable infusion. For instance, a review article from the British Association of Critical Care Nurses, the proximal tibia was identified as the preferred intraosseous access point due to its simplicity, speed, and lower incidence of failed attempts relative to other access points. (Source: University of Huddersfield)
The adults segment is projected to hold a market share of 38.8% in 2026, due to an increasing incidence of trauma, cardiac arrest, sepsis, and emergency surgeries. Peripheral venous access is not feasible in adult patients due to the occurrence of obesity, low blood pressure, blood loss, or vascular collapse. Therefore, intraosseous access serves as an alternative route in emergencies. The market segment is driven by an increasing adoption of advanced cardiac life support techniques and growing utilization of intraosseous devices in hospitals such emergency departments, intensive care units, and ambulance services.
Moreover, medical practitioners favor the use of intraosseous infusions in adult patients due to rapid fluid administration, medication delivery, and higher success rates on their first attempt in case of failure to establish peripheral venous access in emergencies. For instance, a systematic review conducted and presented by the National Library of Medicine indicated the increasing trend towards intraosseous access in adult emergency situations due to rapidity, efficiency, and efficacy. (Source: National Library of Medicine)
|
Current Events |
Description and its Impact |
|
Regulatory Expansion of FDA Emergency Use and Device Modernization Pathways for Critical Care Devices (February 2026) |
|
|
Implementation of the European Union Medical Device Regulation (EU MDR) Strengthening Compliance Requirements (December 2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Food and Drug Administration, European Commission)
To learn more about this report, Request Free Sample
The rising use of intraosseous infusion devices in the field of emergency medicine services and military medicine will significantly contribute to the overall growth of the market owing to the high demand for fast vascular access in cases of trauma, battlefield injuries, cardiac arrests, and emergencies. There is an increasing tendency among emergency medical service providers and military medical staff to integrate the use of intraosseous infusion technology into advanced life support practices for the fast delivery of fluid and medication when IV access is not easily available. One of the prominent players, Cook Medical Emergency Medicine Solutions, is continually expanding its portfolio of emergency vascular access devices that are applicable in trauma and critical care environments.
For example, the company provides innovative products for emergency medicine applications that have been developed specifically to facilitate rapid vascular access in acutely ill patients, thus ensuring the wider use of intraosseous technology in emergency situations. (Source: Cook Medical) With the increasing use of advanced emergency medical equipment technology among both ambulances and militaries, the widespread use of intraosseous infusion technology continues to grow.
To learn more about this report, Request Free Sample
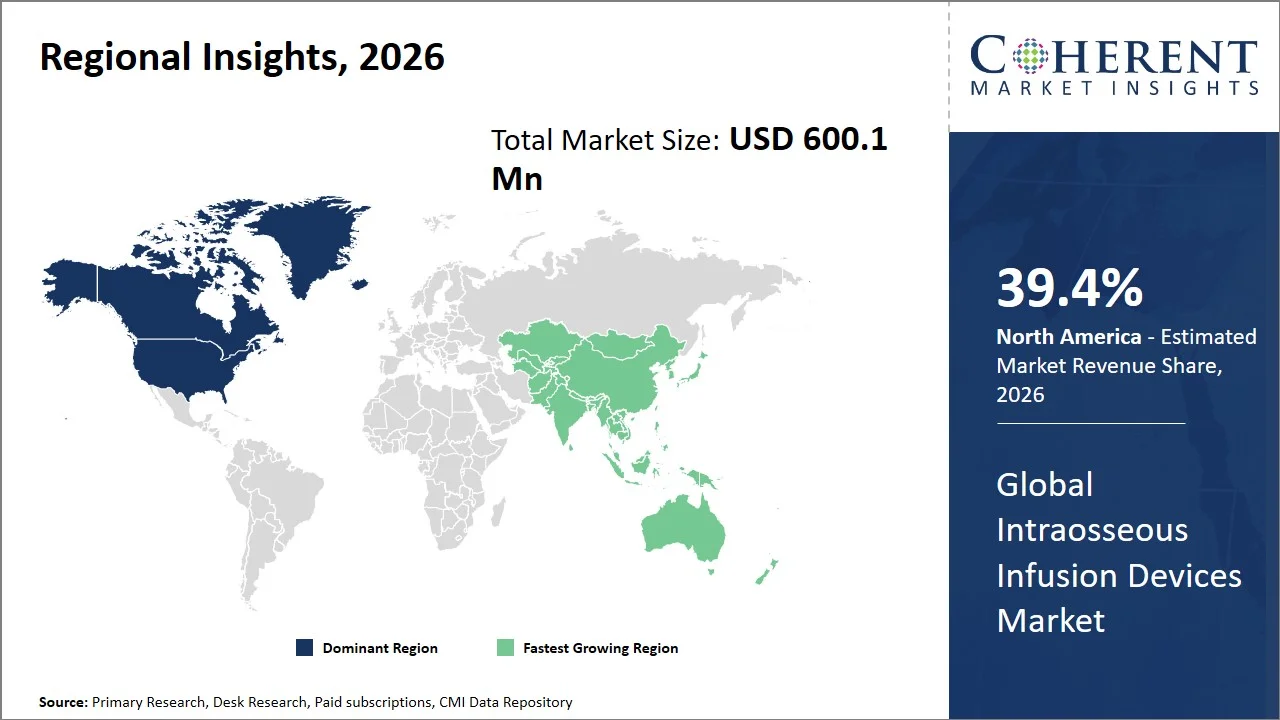
North America leads the global intraosseous infusion devices market, accounting for an estimated 39.4% share in 2026, owing to the increasing adoption of technological innovations within emergency care procedures conducted in trauma centers, air ambulances, and combat casualty care units. The application of technological innovation within the North America region aims at increasing the effectiveness of vascular access in emergencies through advanced infusion devices. For instance, SAM Medical continues to adopt an increased number of SAM IO intraosseous access system within emergency applications in the U.S. to ensure effective fluid delivery in emergency scenarios. (Source: SAM Medical) Moreover, regional healthcare providers are including intraosseous access protocols in advanced life support training programs, further reinforcing the use of the product in emergency medicine scenarios.
The Asia Pacific intraosseous infusion devices market is expected to exhibit the fastest growth with an estimated contribution of 20.5% share to the market in 2026, owing to the development of emergency healthcare infrastructures and adoption of advanced critical care technologies in emerging countries. Countries like China, India, and Japan are enhancing their trauma care programs, ambulance systems, and emergency medical service operations, which results in a need for advanced rapid vascular access products. Furthermore, disaster management and training in emergency medicine are aiding in promoting intraosseous access technologies among healthcare settings. For instance, Pyng Medical Corp. (now part of Teleflex) FAST1 Intraosseous Technology is increasingly being promoted for use in military and emergency medical settings, leading to a rise in demand for advanced intraosseous access devices in healthcare markets globally, including those in the Asia Pacific region. (Source: Teleflex Incorporated)
The U.S. has the potential for leadership in innovation and adoption in the field of intraosseous infusion devices due to the high product adoption rate owing to the presence of established medical device companies (such as Teleflex Incorporated, Becton, Dickinson and Company (BD), and Cook Medical) and constant development of new technologies in the field of emergency vascular access devices.
Several companies are working in the development of innovative products in terms of usability and portability of intraosseous access systems in emergency settings. The encouraging U.S. FDA policies and regulations (such as the FDA 510(k) Clearance Pathway and Breakthrough Devices Program) are also helping in increasing the approval rate of innovative medical devices and thereby driving the growth of this market. Additionally, the extensive use of intraosseous access systems in hospitals, emergency medical services (EMS), and military healthcare establishments are contributing positively to the market growth.
The U.K. is an ideal market for intraosseous infusion devices attributed to the widespread use of highly sophisticated emergency care equipment in hospitals as well as in ambulance services. The existence of highly developed healthcare facilities and the rising emphasis on rapid vascular access techniques have led to the expansion of the market. Regulatory backing from the Medicines and Healthcare products Regulatory Agency (MHRA) has been helpful in promoting innovations in the development of emergency care devices in the country. Furthermore, the rising application of intraosseous access systems for trauma care, critical care, and pre-hospital emergency treatments has contributed to the growth of the market in the country.
The intraosseous infusion devices market in China is currently experiencing significant development attributed to the evolution of its healthcare infrastructure during emergencies as well as the introduction of advanced healthcare technology in hospitals and trauma centers. The application of rapid vascular access devices is also on the rise in cases of emergencies and in critical care units. The efforts of the National Medical Products Administration (NMPA) through regulation have been encouraging the adoption of new devices and technologies into the healthcare system. In addition, advancements being made in emergency medical services (EMS) and critical care units have been contributing to the growth of the market.
Germany dominates the intraosseous infusion devices market in Europe owing to the country’s advanced health care infrastructure and high adoption rates of emergency and critical care devices in hospitals and trauma centers (Germany has more than 1,900 hospitals, according to the Federal Statistical Office of Germany). There is an established network for emergency medical services in Germany, which facilitates the use of fast vascular access devices in pre-hospital and critical care scenarios. Regulations under the European Union Medical Device Regulation (EU MDR) are conducive to the adoption of technological advancements in intraosseous devices. Moreover, the presence of top medical devices manufacturers (such as Becton, Dickinson and Company (BD), Teleflex Incorporated, and Cook Medical) and increased attention towards emergency care advancements are fueling the market growth.
Japan is gradually evolving into a prominent market for intraosseous infusion devices owing to the rising uptake of sophisticated emergency and critical care equipment at hospitals and healthcare facilities. The country’s commitment to technological advancements in medical devices and precision healthcare has led to the use of rapid vascular access devices in emergency medicine. Furthermore, the regulatory endorsement by the Pharmaceuticals and Medical Devices Agency (PMDA) has helped in promoting technologically advanced intraosseous systems. Besides, the rising use of intraosseous devices in trauma treatment, disaster relief operations, and emergency medical services (EMS) is aiding the growth of the market.
|
Region |
Key Regulatory Bodies |
Approval & Compliance Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA), Health Canada |
FDA 510(k) clearance, Quality System Regulation (QSR), ISO 13485 compliance |
Strong regulatory oversight ensures product safety and accelerates adoption of technologically advanced intraosseous infusion devices across hospitals and EMS settings |
|
Europe |
European Medicines Agency (EMA), European Commission, Notified Bodies |
EU Medical Device Regulation (MDR 2017/745), CE Mark certification, ISO 13485 standards |
Strict compliance requirements improve product quality and patient safety while increasing entry barriers for new manufacturers |
|
Asia Pacific |
National Medical Products Administration (NMPA - China), Central Drugs Standard Control Organization (CDSCO - India), Pharmaceuticals and Medical Devices Agency (PMDA - Japan) |
Country-specific medical device approvals, product registration, safety and quality compliance |
Expanding healthcare infrastructure and improving regulatory harmonization are supporting market growth and foreign manufacturer entry |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
Medical device registration, import approvals, local compliance and quality standards |
Growing healthcare investments and evolving regulatory frameworks are increasing adoption of emergency medical devices across the region |
|
Middle East & Africa |
Saudi Food and Drug Authority (SFDA), South African Health Products Regulatory Authority (SAHPRA), UAE Ministry of Health |
Device registration, import licensing, regional quality and safety regulations |
Increasing focus on emergency healthcare preparedness and healthcare modernization is driving demand for compliant intraosseous infusion devices |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Technology-enabled intraosseous infusion system devices that are battery-powered and easy-to-use are creating new growth opportunities in the global intraosseous infusion devices market owing to features like fast insertion, accuracy, portability, and usability during emergencies. Technological advancements in battery-operated devices enables health professionals to get rapid vascular access within few seconds in conditions that include trauma care, cardiac arrest, and other emergencies, where vascular access becomes difficult using traditional methods of infusion. The focus for manufactures in designing the device has been mainly on ergonomic devices, safety features, and portability for enhanced efficiency in emergencies. Moreover, technological advancements witnessed in the clinical sector have been pushing manufacturers to design advanced intraosseous devices.
For instance, a retrospective observational study conducted in December 2024 in the National Library of Medicine found that the safety and effectiveness of the Arrow EZ-IO System for pediatric emergency medicine is high and has no adverse events, which proves the increasing acceptance of intraosseous devices with innovative technology in clinical practice. (Source: National Library of Medicine) The continual advancement in the area of powered intraosseous access technologies are anticipated to improve the performance of emergency care systems and drive the market expansion.

To learn more about this report, Request Free Sample
The global intraosseous infusion devices market is moderately competitive due to the presence of well-known companies in the sector that emphasize the development of fast vascular access technology. Companies within the market continue to invest in innovations, drill-driven products, and portable intraosseous access products to increase their market share in hospitals, EMS services, and military healthcare markets. Other strategic initiatives being employed by companies to improve their market share include product launches and regulatory approvals. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 600.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.1% | 2033 Value Projection: | USD 972.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teleflex Incorporated, BD, Cook Medical, SAM Medical, Argon Medical Devices, Inc., PerSys Medical, Pyng Medical Corp., Aero Healthcare, PAVmed Inc., and BIOPSYBELL S.R.L. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients