Ischemia Reperfusion Injury Therapeutics Market is estimated to be valued at USD 2.45 Bn in 2026 and is expected to reach USD 3.74 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
Analysts’ Views on Global Ischemia Reperfusion Injury Therapeutics Market:
Increasing research & development activities by the market players, in turn is expected to drive the market growth over the forecast period. For instance, Amyndas Pharmaceuticals, a pharmaceutical company, had developed a proprietary drug delivery platform called Nano-EMulsion Delivery System (NEDS), which allows for targeted delivery of drugs to the affected tissues or organs. This platform technology enhances the efficacy of the drugs and reduces its toxicity. Thus, increasing use of these kind of platform can further help the company in strengthening its drug efficacy.
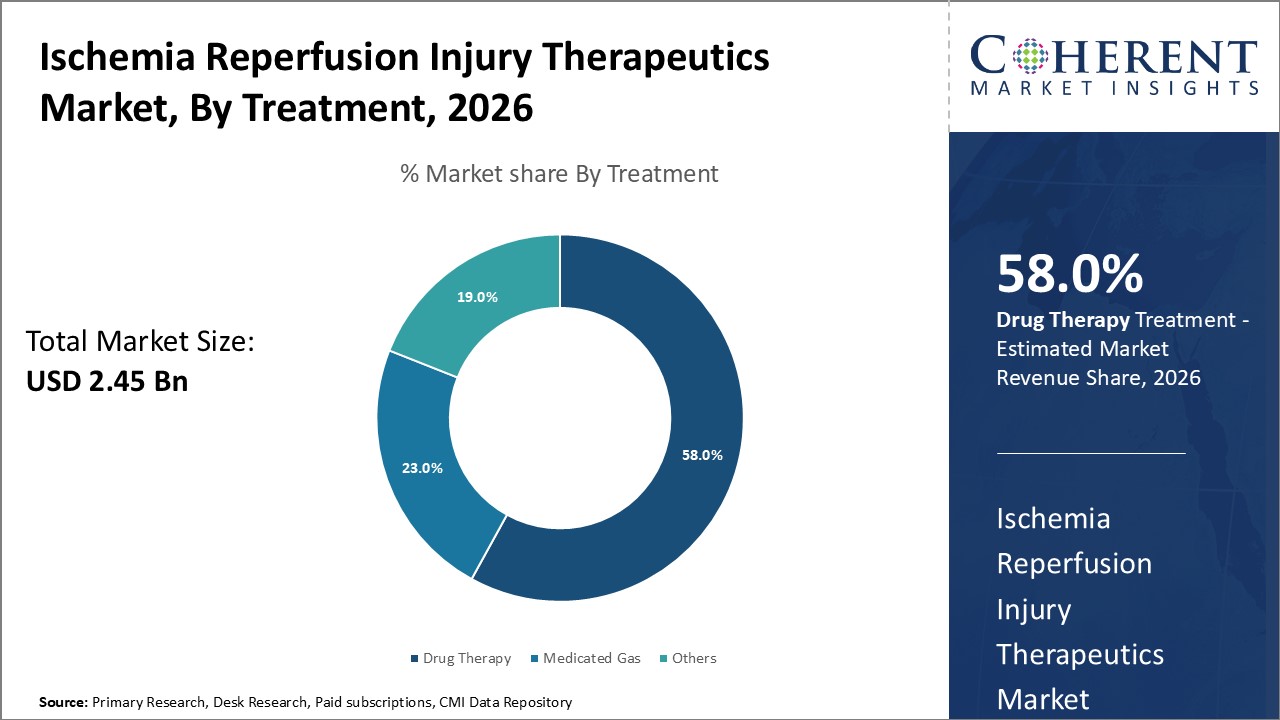
Figure 1. Global Ischemia Reperfusion Injury Therapeutics Market Share (%), by Treatment, 2026
To learn more about this report, Request Free Sample
Global Ischemia Reperfusion Injury Therapeutics Market– Driver
Increasing prevalence of heart injuries
Increasing prevalence of various kinds of heart injures, such as cardiac, great vessel, pulmonary injuries and others, is expected to fuel the market growth over the forecast period. For instance, the American Heart Association has released Heart and Stroke Statistics- 2022 update, according to which it reported that cardiac arrest (CA) remains a public health crisis. There are more than 356,000 out-of-hospital cardiac arrests (OHCA) annually in the U.S., out of which nearly 90% of them are fatal.
Similarly, in March 2022, according to the report published by National Center for Biotechnology Information Center, reported that annual mortality from CA (cardiac arrest) is as high as 4–5 million deaths worldwide, with approximate 544,000 deaths in China.
Increasing research & development activities by the market players
The key market players are focused on research and development activities used treatment of patients suffering from various disease such as organ injury, acute kidney injury and others, which in turn is expected to drive the market growth over the forecast period. For instance, Angion Biomedica, a pharmaceutical company, has developed a drug candidate called ANG-3777, which is a hepatocyte growth factor (HGF) mimetic. ANG-3777 has been shown to reduce inflammation, promote tissue regeneration, and improve organ function in preclinical studies of various types of acute organ injuries caused by ischemia reperfusion injury. ANG-3777 has been granted orphan drug designation by the U.S. Food and Drug Administration (FDA) for the treatment of acute kidney injury.
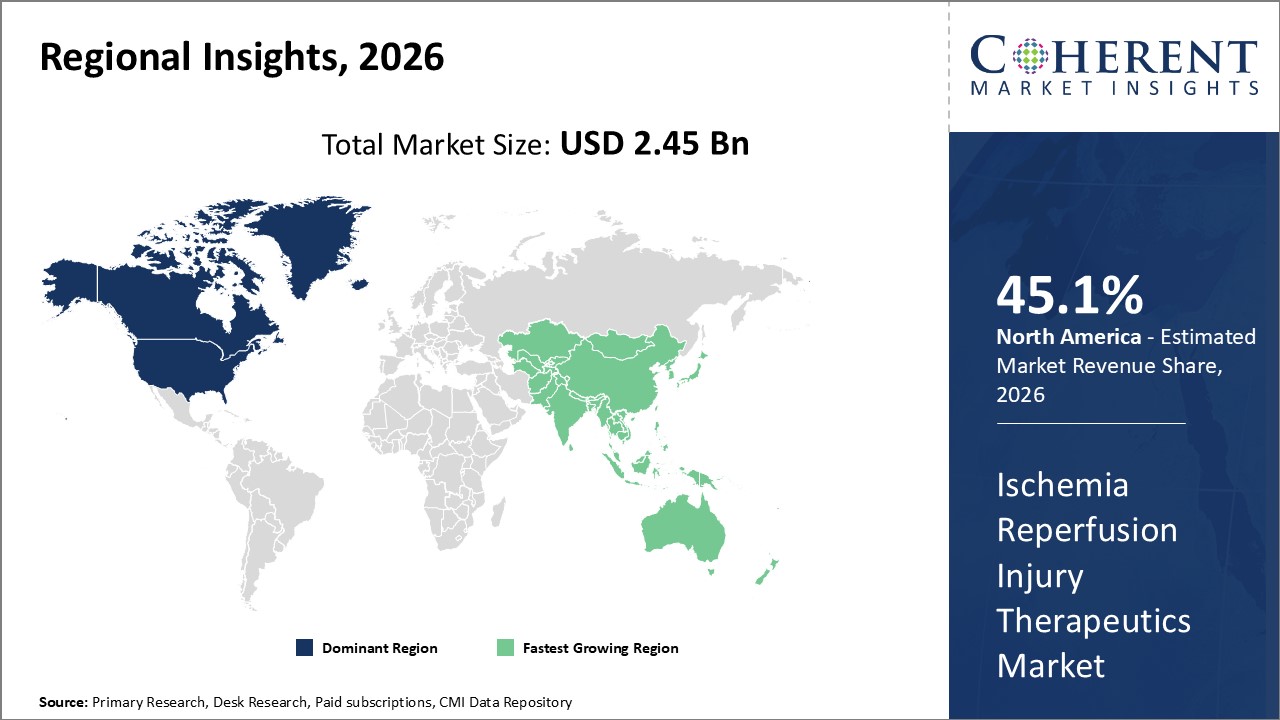
Figure 2. Global Ischemia Reperfusion Injury Therapeutics Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Ischemia Reperfusion Injury Therapeutics Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global ischemia reperfusion injury therapeutics market over the forecast period. This is attributed to the increasing prevalence of various injuries such as heart injuries, brain injuries, kidney injuries and others. For instance, in November 2021, according to the report published in the National Center for Biotechnology Center, reported that, Heart failure (HF) is a major public health problem, with a prevalence of over 5.8 million in the U.S., and over 23 million worldwide in the year 2021.
For instance, in June 2022, according to the report published by the National Institute of Cardiovascular Outcomes Research (NICOR), in the U.K. number of operations performed were down by 34% in the year 2020/21 with only 19,333 adult heart operations, and 21% increase in waiting times in England, 61% increase in Northern Ireland, and 94% increase in Wales.
Global Ischemia Reperfusion Injury Therapeutics Market Segmentation:
The global ischemia reperfusion injury therapeutics market report is segmented into treatment, injury type, end user and region.
Based on Treatment, the global ischemia reperfusion injury therapeutics market is segmented into Drug Therapy, Medicated Gas, and Others (Vitamin therapy and among others). Out of which, Drug Therapy segment is expected to dominate the global ischemia reperfusion injury therapeutics market during the forecast period and this is due to increasing adoption of various growth strategies, such as product launch, partnership, and others by the market players.
Based on Injury Type, the global ischemia reperfusion injury therapeutics market is segmented into Heart Injury, Kidney Injury, Intestine Injury, and Others. Out of which, heart injury segment is expected to dominate the global ischemia reperfusion injury therapeutics market during the forecast period owing to increasing prevalence of various types of heart injuries such as coronary heart disease and others.
Based on End user, the global ischemia reperfusion injury therapeutics market is segmented into hospitals, specialty clinics, ambulatory surgical centers, and others (Academic and Research Institutes and among others). The hospitals segment is expected to dominate the market over the forecast period and this is due to the prevalence of cardiac arrest across the globe.
Ischemia Reperfusion Injury Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.45 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 3.74 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Revive Therapeutics Ltd., Faraday Pharmaceuticals, Abiomed, Omeros Corporation, Pharming Group N.V., Nyken B.V., Orexo AB, Opsona Therapeutics Limited, PledPharma AB, Prothix BV, Proteo, Inc., Amyndas Pharmaceuticals LLC, Prolong Pharmaceuticals, Stealth BioTherapeutics Inc., Zealand Pharma A/S, Antipodean Pharmaceuticals, Inc., Bayer AG, Angion Biomedica Corp., Bolder Biotechnology, Inc., Curatis Pharma GmbH, Biomedica Management Corporation, Ensemble Therapeutics Corporation, Gilead Sciences, Inc., Erimos Pharmaceuticals, LLC. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Ischemia Reperfusion Injury Therapeutics Market Cross Sectional Analysis:
Increasing initiatives taken by the non-profit organization such as awareness campaign is expected to drive the growth of the product type segment in North America region. For instance, on May 2, 2023, American Heart Association, Inc., a non-profit organization, conducted an awareness campaign in the U.S. in order to urge people to learn the signs of stroke and how to prevent one.
Global Ischemia Reperfusion Injury Therapeutics Market: Key Developments
In March 2022, SBI Pharmaceuticals, a pharmaceutical company in collaboration with the University of Oxford, announced that SBI Pharmaceuticals had initiated a phase II clinical trial to develop a cardio protective agent combining 5-ALA hydrochloride and the cardiac surgery under cardiopulmonary bypass, the sodium ferrous citrate is response for the mitigation of ischemia reperfusion.
In April 2021, PharmaDrug Inc., a specialty pharmaceutical company, announced that U.S. FDA (Food and Drug Administration) had granted Orphan Drug Designation to dimethyltryptamine (DMT) for the Prevention of Ischemia-Reperfusion Injury from Organ Transplantation, which includes the liver, kidney, heart, and lung.
Global Ischemia Reperfusion Injury Therapeutics Market: Key Trend
Increasing number of clinical trials by the market players
The market players are focused on conducting clinical trials for development of drugs used in treatment of acute ischemia reperfusion, is expected to fuel the segmental growth over the forecast period. For instance, in December 2021, Faraday Pharmaceuticals, Inc., a muscle health-focused biopharmaceutical company, announced that the publication of results from its Phase 2 trial of FDY-5301 for the treatment of reperfusion injury following an ST-elevation myocardial infarction (STEMI) in the January 15, 2022 issue of the International Journal of Cardiology. Data from the study, known as Iocyte AMI, demonstrated the treatment was well-tolerated and provided encouraging signals of potential efficacy in minimizing cardiac damage. FDY-5301 is an elemental reducing agent containing sodium iodide, which works to catalytically destroy hydrogen peroxide, which is generated as a response to acute injury and contributes to loss of muscle mass and function.
Global Ischemia Reperfusion Injury Therapeutics Market: Restraint
Lack of approved drugs for treatment of ischemia reperfusion injury
No approved drugs available in the market for treatment of patients suffering from ischemia reperfusion injury is major hindrance in the market, on the other hand market players are adopting various growth strategies such as research & development activities and others, for development of drugs used in treatment of ischemia reperfusion injury is currently in clinical trial, which in future can get approved from the U.S FDA (Food and Drug Administration)
Global Ischemia Reperfusion Injury Therapeutics Market- Key Players
*Definition: Ischemia reperfusion injury (IRI), also known as reoxygenation injury, is tissue damage that arises when blood supply returns to the tissue (reperfusion) after a period of lack of oxygen (ischemia). The symptoms of an ischemia injury include bradycardia, headache, hypertension, and seizures.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients