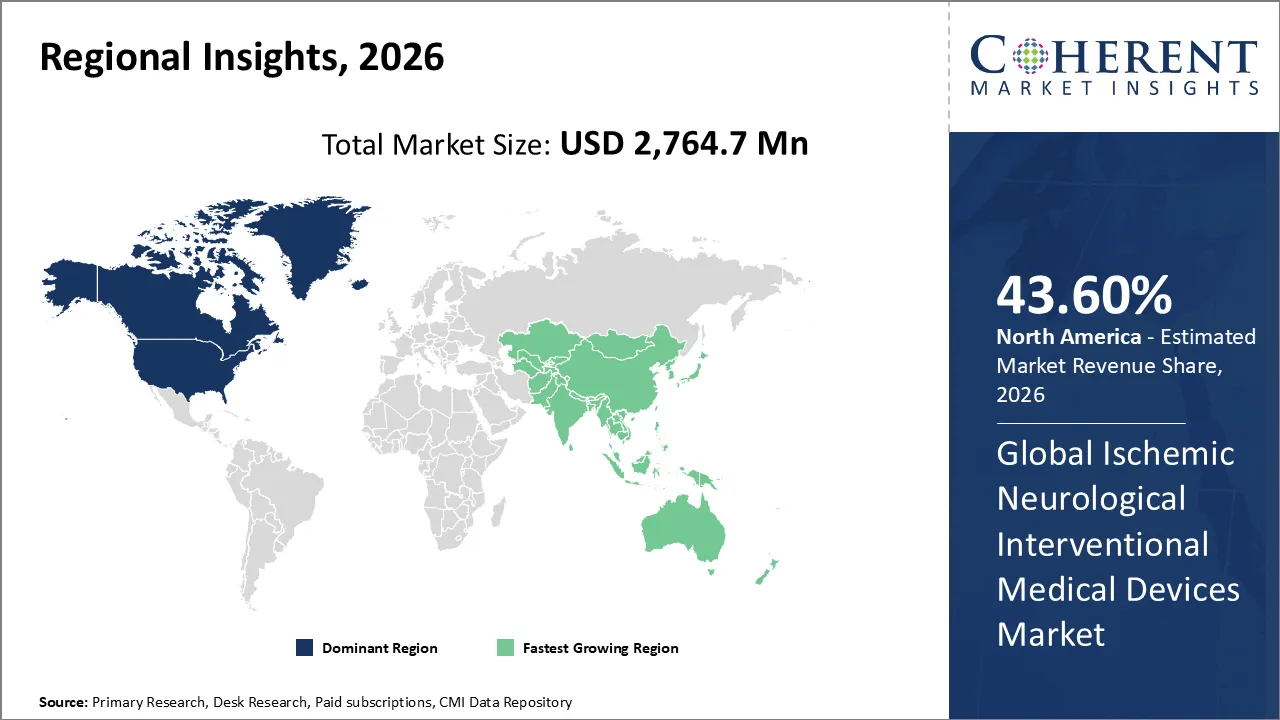
The global ischemic neurological interventional medical devices market is valued at USD 2,764.7 Mn in 2026 and is forecast to reach a value of USD 4,050.2 Mn by 2033 at a CAGR of 5.6% between 2026 and 2033.
Medical devices are the electronic products which have medical use and make medical claim. The medical devices includes ultrasound products, x-ray machines, medical lasers and neurological interventional kits. Neuro-interventional devices are catheters-based, minimally-invasive approaches which are used to treat specific diseases of the blood vessels of spine, neck, head and which includes strokes, brain aneurysms, vascular malformation in brain, spine and artery diseases.
The several types of Neuro-interventional devices such as flow diverters, stents, and aspiration devices are mainly used for ischemic stroke. There are mainly two types of strokes ischemic and hemorrhagic. Ischemic stroke is common type, and usually cause by blood clotting that blocks blood vessels of brain. Different Neuro-interventional devices like Aspiration device, stenting systems, clot retriever, lasers are used for diagnosis and treatment of ischemic stroke.
|
Current Event |
Description and its Impact |
|
AI Integration and Next-Generation Device Innovation |
|
|
Emerging Market Healthcare Infrastructure Development |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Treatment |
Cost Impact |
Hospital Stay |
Rehab Costs |
Notes |
|
Mechanical Thrombectomy |
Saves USD 20k–30k per patient |
~3 days shorter |
Much lower due to independence |
High device cost offset by reimbursement. |
|
IV Thrombolysis (tPA) |
Drug cost USD 6k–8k |
Slightly shorter |
Moderate; limited large vessel use |
Effective only within 4.5-hour window. |
|
Medical Therapy Alone |
Low upfront (USD 2k–5k) |
Longer (7–10 days) |
High; disability common |
Appears cheap but costly long-term. |
|
Carotid Stenting/CEA |
USD 15k–20k |
2–4 days |
Preventive savings |
More preventive than acute intervention. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of procedure type, the mechanical thrombectomy segment is expected to lead the market with 55% share in 2026. Evidence shows it improves functional independence in nearly half of treated patients, which is much better than standard care. It is the most common procedure because it works well, has a high success rate, and is well-known in international stroke guidelines.
For instance, in February 2025, Johnson & Johnson MedTech released the Cereglide 92 Catheter System to treat acute ischemic stroke. The device makes it easier to get to the neurovascular system and get rid of clots, which directly helps with mechanical thrombectomy procedures. This new idea makes their role in ischemic neurological interventional medical devices stronger, which leads to better results and more advanced stroke care options around the world.
In terms of device type, the neurothrombectomy devices segment is expected to hold 40% share of the market in 2026, because they have been shown to restore blood flow in strokes that block large blood vessels. Clinical trials consistently show that patients reflect better outcomes when they get both medical therapy and clinical trials. Endorsements from guidelines have made them even more popular, and by 2026, they will be the most important part of treating ischemic stroke.
For instance, in May 2025, S3V Vascular Technologies, with help from the Department of Science and Technology, announced India's first indigenous mechanical thrombectomy kit for acute ischemic stroke. The goal of the initiative is to make the product less reliant on imports, more affordable, more widely available through Ayushman Bharat, and to get CE and USFDA approvals for global distribution.
In terms of end user, the hospitals segment is projected to account for 60% shares of the market win 2026, because they have advanced imaging systems, specialized neurointerventional teams, and full stroke units. Their infrastructure allows for quick diagnosis and treatment, which is crucial for managing acute ischemic stroke. This readiness makes sure that hospitals are still the main place to get thrombectomy and stenting procedures in 2026.
For instance, in March 2026, Johnson & Johnson launched CEREGLIDE 42 and CEREGLIDE 57 aspiration catheters, as well as INNERGLIDE 7, to improve the treatment of acute ischemic stroke. These devices make it easier to get to distal clots, make aspiration more effective, and boost procedural confidence. They also strengthen hospital-based neurointerventional workflows and add to their portfolio of ischemic neurological interventional medical devices.
To learn more about this report, Request Free Sample
North America is expected to dominate the ischemic neurological interventional medical devices market with 43.60% share in 2026, because there is large incidence of strokes, the healthcare system is advanced, there are good reimbursement policies, and new devices are quickly adopted. The area's aging population and the presence of major manufacturers make the market even stronger.
For instance, in January 2026, the new AHA/ASA guideline for acute ischemic stroke, which released in the US, adds to the advice for mechanical thrombectomy and thrombolysis. It emphasizes the importance of hospital-based stroke systems, direct transport to centers that can do thrombectomy, and longer treatment windows.
Asia Pacific is expected to exhibit the fastest growth, due to the rise in strokes, the rapid growth of healthcare infrastructure, and government efforts to make it easier for people to access care. The market is growing the fastest in this region because more people are moving to cities, lifestyles are changing, and more people are using advanced neurointerventional technologies.
For instance, in July 2025, Researchers at CUHK created a microcatheter with a magnetic tip that can be turned for use in treating acute ischemic stroke. The device accurately moves through complicated blood vessels in the brain to deliver drugs, break up clots, and pick up debris. It was tested on both animals and humans and showed that it was safe and effective, which was a big step forward for neurointerventional medical devices.
In 2026, the U.S. has high demand for ischemic neurological interventional medical devices due to high prevalence of strokes, the healthcare system is advanced, the reimbursement systems are exceptionally good, and new technologies are being adopted quickly. The country's market dominance is even stronger because its population is getting older and there are many leading manufacturers there.
For instance, in March 2026, Medtronic plan on purchasing Scientia Vascular, which is based in Salt Lake City, Utah, for $550 million, plus extra payments for reaching certain goals. The deal, which was announced from Galway, Ireland, combines Scientia's guidewires and microcatheters with Medtronic's neurovascular therapies. This will improve global ischemic neurological interventional medical device innovation and expand advanced stroke care capabilities around the world.
India's need for ischemic neurological interventional medical devices will grow in 2026 because strokes are becoming more common, cities are growing quickly, and people are living unhealthy lives. India is a key growth market in Asia-Pacific due to its healthcare infrastructure is getting better, the government is working to make it easier for people to get care, and more people are using advanced neurointerventional technologies.
For instance, in March 2026, XCath made history in India by performing the world's first remote robotic mechanical thrombectomy for acute ischemic stroke. Using the Iris Surgical Robotic System, doctors were able to remove a clot that was more than 200 kilometers away. This restored blood flow and showed how robotics could help people around the world get better stroke care.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,764.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.6% | 2033 Value Projection: | USD 4,050.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Stryker, Johnson & Johnson Private Limited., Penumbra, Inc., Medtronic, VESALIO, LLC., Sense Neuro, 880 Medical, LLC., Terumo Corporation., Imperative Care., W. L. Gore & Associate, Inc., MicroPort Scientific Corporation., KANEKA COPORATION., Integer Holdings Corporation., Wallaby Medical. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing focus on research and development by key players in order to strengthen their market presence in the global market is expected to drive growth of the global ischemic neurological interventional medical devices market over the forecast period. For instance, in March 2026, Researchers examined multidirectional diffusion-weighted imaging (DWI) utilizing portable low-field MRI to enhance the detection of acute ischemic stroke. In a study involving 95 patients, multidirectional diffusion-weighted imaging (DWI) demonstrated superior sensitivity compared to single-direction DWI, successfully identifying minute lesions and improving emergency triage, indicative of continuous advancements in ischemic neurological interventional medical devices.
The increasing product launch and product approval by key players in order to strengthen their market presence in the global market is expected to drive growth of the global ischemic neurological interventional medical devices market over the forecast period. For instance, in February 2026, The CE Mark approvals for NeVa VS, which treats cerebral vasospasm, and NeVa 3.0 mm, which is used for mechanical thrombectomy in smaller vessels, added to Vesalio's neurovascular portfolio. The company also got FDA 510(k) approval for aspiration catheters. This is a big step forward for ischemic neurological interventional medical devices and stroke care.
The ischemic neurological interventional medical device market forecast shows that aspiration and suction catheters are essential because they work well with thrombectomy procedures. They do a great job of getting rid of thrombus fragments, which lowers the risk of distal embolization and makes patients risk-free. Hospitals and neurosurgery centers all over the world are becoming more interested in them because they are minimally invasive and work with existing thrombectomy systems. This is especially true where cost-effective solutions are important. Aspiration catheters are an enormous expansion opportunity in the global market because more people want better stroke care.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients