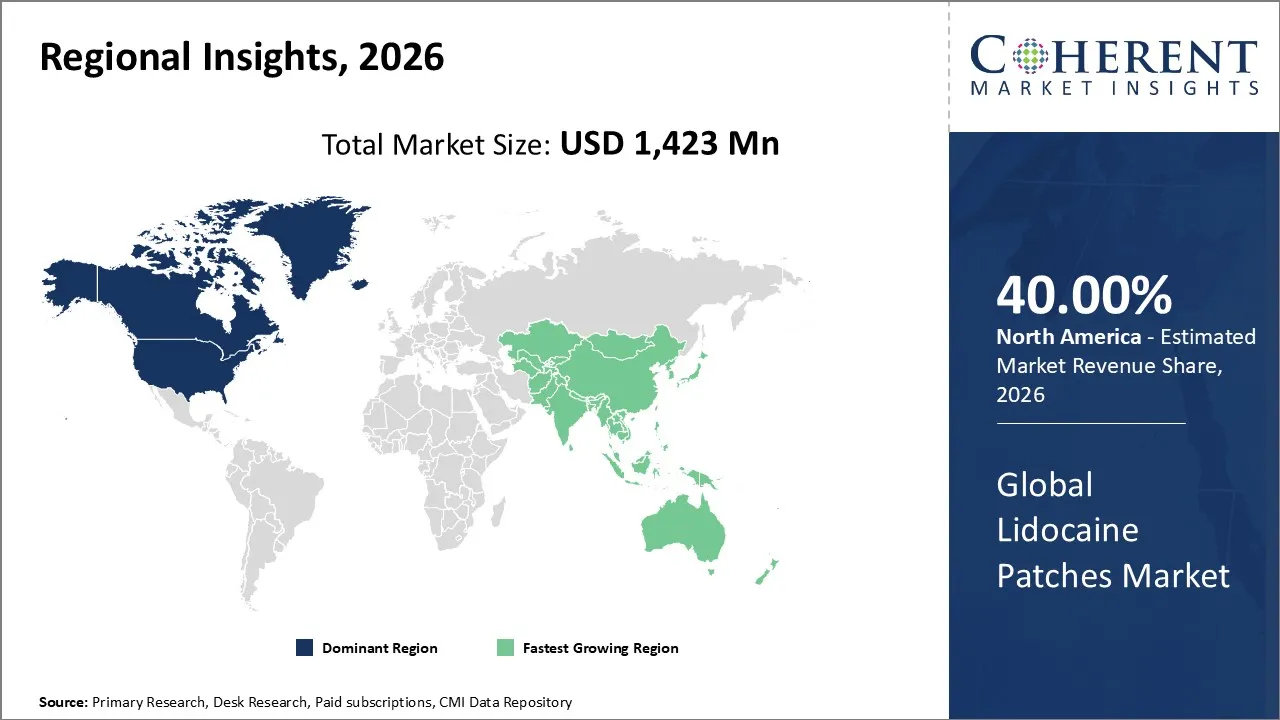
Lidocaine patches market is estimated to be valued at USD 1,423 Mn in 2026 and is expected to reach USD 2,403 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.8% from 2026 to 2033.
Increasing incidence rate of chronic pain-related diseases like post-herpetic neuralgia, arthritis, and musculoskeletal disorders is fostering the demand for lidocaine patches across the globe. Lidocaine patches are currently attaining significant attention as a pain-relieving measure for chronic diseases as they offer an invasive-free method for patients, especially the elderly, and reduce the chances of side effects associated with other analgesic drugs. Increasing awareness about topical anesthetic pain-relieving drugs and the need for opioid-free pain management is also spurring the demand for lidocaine patches.
|
Current Events |
and its impact |
|
Regulatory and Healthcare Policy Changes |
|
|
Supply Chain and Raw Material Disruptions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on strength, the 5% segment is estimated to contribute the highest market share of around 55% in 2026, largely due to its effectiveness in managing moderate to severe localized pain conditions. In particular, the 5% lidocaine patch is widely used in managing post-herpetic neuralgia, aside from its general applications in chronic pain.
For instance, in May 2024, Teikoku Pharma USA announced that Lidoderm® 5% lidocaine patch was approved by the NMPA in China for the treatment of post-herpetic neuralgia
By adhesive material, the aqueous segment is projected to account for the largest market share of approximately 60% in 2026. This is due to its higher tolerance and easy application nature. The aqueous nature of lidocaine patches provides comfort with reduced irritation to the skin and enhanced breathability, especially for elderly patients.
For instance, in September 2025, the U.S. Food and Drug Administration (FDA) has approved Bondlido, a novel topical lidocaine system being developed by MEDRx Group and D. Western Therapeutics Institute, to treat post herpetic neuralgia (PHN).
Application-wise, the PHN segment is expected to dominate the share with around 60% by 2026, as lidocaine patches are widely recommended as a first-line topical treatment for neuropathic pain associated with PHN. Increasing incidence of herpes zoster infections among aging populations, together with long-term pain complications, has maintained demand for lidocaine patches in this segment.
For instance, in March 2025, Aveva Drug Delivery Systems, a company of DifGen, announced that the United States Food and Drug Administration (FDA) approved the company's patch formulation for its generic lidocaine 1.8% for the treatment of localized nerve pain, such as postherpetic neuralgia (PHN).
Based on distribution channel, retail pharmacies are expected to contribute nearly 45% of the global market share in 2026, owing to their increased accessibility, higher prescription fill rates, and increasing availability of both prescription and OTC formulation. Retail pharmacies act as a means of purchase for many customers suffering from chronic pain since they need immediate alleviation of the symptoms.
For instance, in July 2025, Biofreeze expanded retail pain relief portfolio with new flexible lidocaine patch offerings, Biofreeze introduced the Ultraflex Patch, a 4% lidocaine patch designed for over‑the‑counter pain relief, featuring enhanced flexibility and comfort.
To learn more about this report, Request Free Sample
North America is likely to become the major revenue contributor to the lidocaine patches market in 2026, accounting for a market share of around 40%. The factors contributing to this include the high prevalence of chronic pain conditions, strong healthcare infrastructure, a very high adoption of topical analgesics, and the presence of leading pharmaceutical manufacturers. Supportive insurance coverage and well-established retail and hospital pharmacy networks add to better accessibility for patients.
For instance, in May 2024, China's National Medical Products Administration (NMPA) gave Teikoku Pharma USA the go-ahead for its Lidoderm® 5% patch. This shows how North American companies are expanding around the world to take advantage of their leading position and meet the growing demand for PHN and other chronic pain treatments.
The Asia Pacific market is expected to be the fastest-growing market for lidocaine patches due to the increasing awareness about the management of chronic pain, higher healthcare spending, and rising insurance coverage in the region, as well as the increased availability of prescriptions and OTC topical analgesics. New pharmaceutical companies in the market are launching novel formulations for lidocaine patches to serve the market in China, India, Japan, and Southeast Asia.
For instance, in May 2025, Jiudian Pharmaceuticals launched a lidocaine gel patch for PHN treatment in China, which was recognized as a first-line topical therapy under national insurance coverage.
The U.S. lidocaine patches market is witnessing significant growth due to the high rate of occurrence of post-herpetic neuralgia (PHN) and chronic pain, coupled with the established health care system and high health insurance coverage in the country. Prescription and OTC lidocaine patches are increasingly being used in the hospital and retail pharmacy chains.
China’s focus on healthcare expansion and chronic pain treatment is accelerating lidocaine patch adoption. Government support for accessible pain management and coverage of topical therapies under insurance programs encourages use among the aging population.
For instance, in May 2025, Jiudian Pharmaceuticals launched a lidocaine gel patch for PHN treatment in China, marking the product as a first-line topical therapy under national insurance coverage.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,423 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.8% | 2033 Value Projection: | USD 2,403 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Endo International plc, Teva Pharmaceutical Industries Ltd, Viatris Inc. (Mylan N.V.), Teikoku Pharma USA, Inc., Hisamitsu Pharmaceuticals Co. Inc., Galen Limited, Sorrento Therapeutics, Inc., Amneal Pharmaceuticals, Inc., Rhodes Pharmaceuticals L.P., 6iPain Healthcare Private Limited, and Sanofi S.A. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
An increasing rate of chronic pain syndromes, including post-herpetic neuralgia, diabetic neuropathy, and musculoskeletal discomfort, is fueling the demand for lidocaine patches. Patients, as well as clinicians, prefer topical agents that avoid the side effects of systemic agents like tablets or opioids. Expanding availability through more hospital, retail, and internet sales is also supporting the product’s rising popularity.
The market offers scope for expansion and has good growth prospects through its entry into emerging markets that include Asia Pacific, Latin America, and the Middle East. Moreover, the development of new therapeutic applications such as diabetic neuropathy and post-surgical pain, and the development of new and different forms of patches such as hydrogel or extended-patch formulation, also provide scope.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients