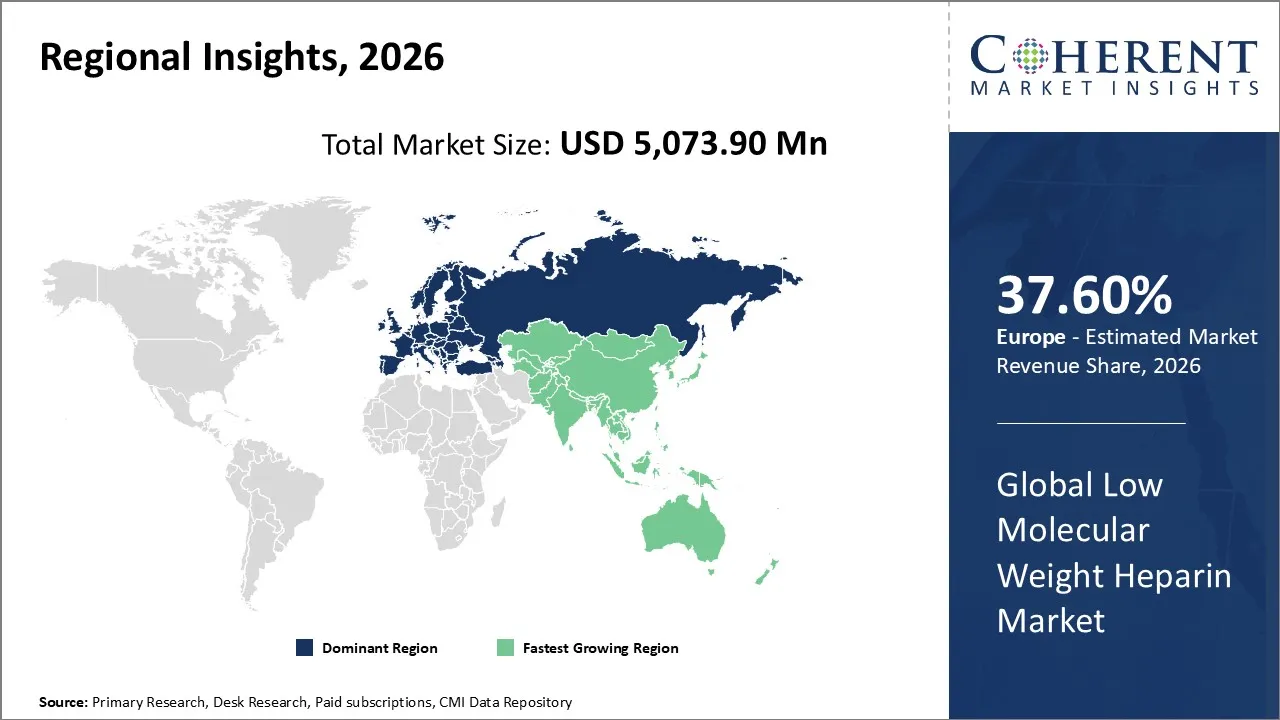
The global low molecular weight heparin market was valued at USD 5,073.90 Mn in 2026 and is forecast to reach a value of USD 8,008.0 Mn by 2033 at a CAGR of 6.7% between 2026 and 2033.
Low molecular weight heparin (LMWH) has several advantages over other heparin such as longer and more predictable activity than unfractionated heparin (UFH) and can be self-administrated at home via subcutaneous injection, reducing or eliminating hospital stays, thereby no regular blood monitoring required. Furthermore, heparins do not cross the placenta or harm the fetus, so they are the preferred anticoagulants for pregnant women who experience or who are at heightened risk of blood clots. These advantages make LMWH as the most preferred heparin in inhibiting clotting factors.
|
Current Event |
Description and its Impact |
|
Regulatory Harmonization and Biosimilar Market Expansion |
|
|
Technological Innovation and Direct Oral Anticoagulants (DOACs) Competition |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug type, the enoxaparin segment is expected to hold 47.3% share in 2026, as it has been shown to work well in preventing and treating venous thromboembolism. It is the best choice since it is widely used in clinics, has a good safety record, and is available in many areas. Physicians rely on it for both inpatient and outpatient care, ensuring its dominance over other LMWH variants.
For instance, in January 2026, Amphastar and Watson launched with a biosimilar version of Enoxaparin sodium injection, which is a popular low molecular weight heparin that is used to stop and treat conditions related to blood clots, like deep vein thrombosis and pulmonary embolism. This change makes things more competitive, easier to get to, and cheaper to treat.
In terms of packaging, the prefilled syringes segment is expected to lead the market with 58% share of the market in 2026, because they lower the risk of dosing mistakes, make patients safer, and make things easier. For ease of use and infection control, hospitals and home-care providers prefer them. Their growing use indicates that healthcare is moving toward solutions that are better for patients, which makes them the most trusted format compared to traditional multi-vial packaging.
For instance, in November 2025, the WAVe clinical trial (NCT03297359) examined weight-adjusted Dalteparin in cancer patients who had venous thromboembolism. Treatment was given with prefilled syringes of different strengths based on the patient's weight.
In terms of application, the deep vein thrombosis segment is projected to account for 42% share of the market in 2026, as more elderly individuals, those who have had surgery, and those with sedentary lifestyles. LMWH is the best way to prevent blood clots from forming, lower the risk of complications, and speed up recovery. Since it is so important for treating DVT, this segment will still be the most important one in 2026.
For instance, in January 2026, Health Canada provided approval to Valeo Pharma's Redesca and Redesca HP, which are low molecular weight heparin biosimilars. These are the first LMWH biosimilars to be approved in Canada. These blood thinners are used to treat and prevent deep vein thrombosis and pulmonary embolism.
In terms of end user, the hospitals segment is projected to capture 63.5% share in 2026. Most prescriptions and administrations of LMWH still visit hospitals, which are the main end users. They are the best as they perform high volume of surgeries, need emergency care, and have patients requiring anticoagulation. Both private and public hospitals depend on LMWH a lot, which makes them the largest group of users compared to clinics or home care.
To learn more about this report, Request Free Sample
Europe is expected to dominate the low molecular weight heparin market with 37.60% share in 2026, due to its aging population, high prevalence of cardiovascular and thromboembolic disorders, and advanced healthcare infrastructure. Policies that make it easy to get paid back and hospitals that use the service widely also help growth. Innovation in pharmaceuticals and the availability of biosimilars also make them easier to get, which keeps Europe the largest regional market.
For instance, in September 2025, The EDQM published the establishment of Heparin Low Molecular Mass Calibration CRS Batch 6, a new reference standard for laboratory and regulatory testing. This calibration ensures consistent quality, accuracy, and safety in Low Molecular Weight Heparin products. It supports harmonized pharmaceutical practices across Europe, reinforcing LMWH’s reliability in clinical applications.
Asia Pacific is expected to be the fastest growing region, because healthcare infrastructure is being built quickly, more people are learning about anticoagulant therapies, and cardiovascular and clotting disorders is rising. Affordable biosimilars are making it easier for people to get them, and more surgeries and cancer-related thrombosis cases are driving adoption even more. This makes APAC the fastest-growing region.
Germany's market for low molecular weight heparin is growing due to it has a large population of older people, a significant number of heart and blood clotting disorders, and a well-developed healthcare system. Strong hospital use, good reimbursement policies, and the presence of top pharmaceutical companies all add to demand. These things make sure that Germany remains a major market hub in Europe.
Low Molecular Weight Heparin from China Market demand is going up because the population is getting older quickly, cardiovascular and thromboembolic disorders are becoming more prevalent, and healthcare infrastructure is growing. More surgeries and cancer-related blood clots are also driving adoption. Affordable LMWH biosimilars make it easier for people to get them, which makes China a major driver of growth in the Asia-Pacific market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,073.90 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.7% | 2033 Value Projection: | USD 8,008.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., LEO Pharma A/S, Sanofi S.A., Teva Pharmaceutical Industries Ltd., Amphastar Pharmaceuticals Inc., Abbott Laboratories, Aspen Pharmacare Holdings, Laboratorios Farmaceuticos ROVI SA, Changzhou Qianhong Biopharma, and Intrapharm Laboratories |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of thromboembolic conditions such as deep vein thrombosis (DVT) and pulmonary embolism (PE) is a major driver of the Low Molecular Weight Heparin Market growth. These disorders pose serious health risks, often requiring immediate and effective anticoagulant therapy. LMWH is a popular choice because its pharmacological profile is easy to predict, it has a lower risk of complications, and it is easy to give. As global healthcare systems face rising cases of clot-related diseases, the demand for LMWH continues to accelerate steadily.
The growing number of older people around the world is a big reason why the Low Molecular Weight Heparin Market demand. Anticoagulant therapy is needed for older adults who are more likely to get heart disease, clotting disorders, and problems after surgery. LMWH is often used in geriatric care because it is safer, works better, and doesn't need to be checked as often as regular heparin. As life expectancy rises globally, the aging population is anticipated to continue as a significant driver for the adoption of LMWH in hospitals and outpatient care facilities.
The Low Molecular Weight Heparin Market forecast shows that the growing demand for homecare solutions is opening up significant opportunities. Patients can give themselves anticoagulant therapy outside of hospitals with prefilled syringes and easier-to-use formulations, reducing clinical visits. This trend is especially beneficial for older people who need long-term care and like things that are easy to use. As healthcare systems focus more and more on patient-centered care, the use of LMWH in homecare settings is expected to grow, which will lead to steady market growth over the next few years.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients