Medical Purity Analyzer Market is estimated to be valued at USD 478.2 Mn in 2026 and is expected to reach USD 719.0 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.0% from 2026 to 2033.
Analysts’ Views on the Global Medical Purity Analyzer Market:
Increasing number of diseases, rising patient hospitalization, and rising geriatrics population, along with the growing healthcare industry are anticipated to propel the market growth over the forecast period. For instance, in July 2022, according to an article published in British Medical Global Health Journal, globally an estimated 5.5 million all-cause hospital admissions occurred annually between 2000 and 2020 in 5-19 year olds, causing 87,900 in-hospital deaths annually.
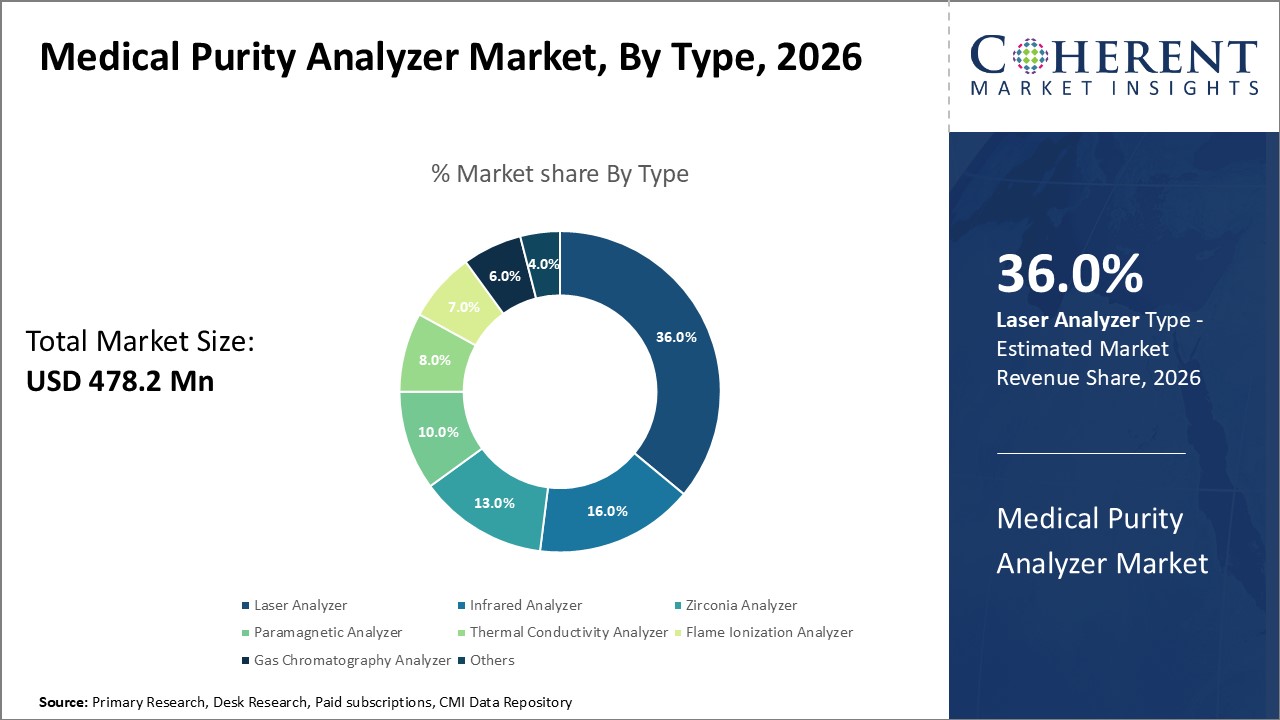
Figure 1. Global Medical Purity Analyzer Market Share (%), By Type, 2026
To learn more about this report, Request Free Sample
Global Medical Purity Analyzer Market - Drivers
Increasing adoption of inorganic strategies by key players
Increasing adoption of inorganic strategies by key players, such as partnerships as well as acquisitions, are expected to drive the market growth over the forecast period. For instance, in September 2020, Teledyne Technologies Incorporated, a provider of sophisticated electronic components, instruments, and communications products, announced the successful acquisition of 3M, a technology company, which manufactures industrial, safety, and consumer products for US$ 230 million in cash. The gas and flame detection business includes Oldham, Simtronics, Gas Measurement Instruments (GMI), Detcon, and select Scott Safety products.
Rising Geriatric Population
Rising geriatric population which led to the increased hospitalization rates for the treatment of various diseases is expected to drive the market growth over the forecast period. For instance, in October 2022, according to the World Health Organization (WHO), in 2020, the number of people aged 60 years and older was 1.2 billion and this number will increase to 1.4 billion by 2030 and 2.1 billion by 2050.
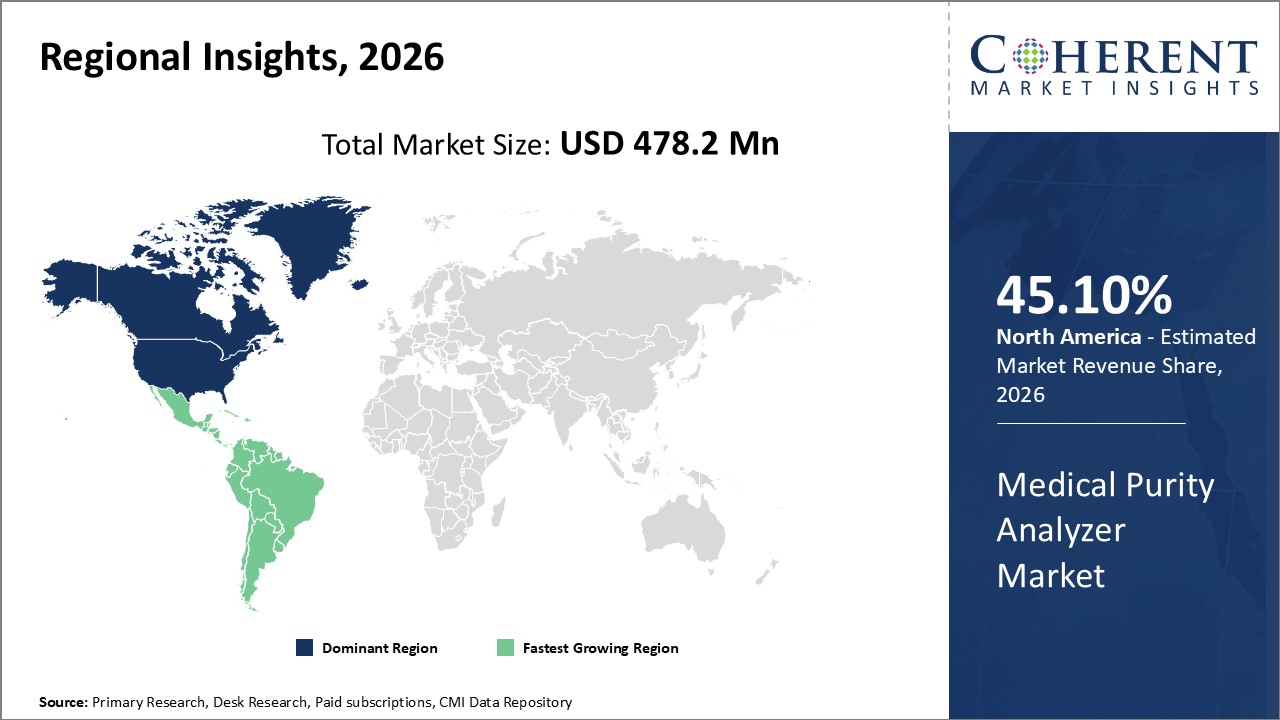
Figure 2. Global Medical Purity Analyzer Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Medical Purity Analyzer Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global medical purity analyzer market over the forecast period owing to the increasing prevalence of chronic diseases and increasing healthcare facilities. For instance, in September 2026, according to Centers for Diseases Control and Prevention (CDC), in 2020, 51.8% of the U.S. adults had at least one chronic condition, and 27.2% had multiple chronic conditions.
oxygendemand.
Global Medical Purity Analyzer Market- Segmentation
The global medical purity analyzer market report is segmented into type, application, end user, and region.
Among type, the global medical purity analyzer market is segmented into laser analyzer, infrared analyzer, zirconia analyzer, paramagnetic analyzer, thermal conductivity analyzer, flame ionization analyzer, gas chromatography analyzer, and others. Out of which, the laser analyzer segment is to dominate the market over the forecast period and this growth is attributed due to the use of medical gases in medical procedures such as surgeries, intensive care, and emergency response, as well as the high prevalence of medical procedures performed worldwide.
Among application, the global medical purity analyzer market is segmented into medical gas production, medical gas verification, and others. Out of which, the medical gas production is expected to dominate the market over the forecast period and this is attributed to the increasing demand for medical gas as increase in the prevalence of chronic diseases.
Among end user, the global medical purity analyzer market is segmented into healthcare, mining, military, manufacturing construction, and others. Out of which, the military segment is expected to grow in the global medical purity analyzer market over the forecast period, and this growth is attributed to the use of medical gases in medical procedures, such as surgeries, intensive care, and emergency response, as well as the high prevalence of medical procedures performed worldwide.
Among region, the global medical purity analyzer market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, North America is expected to dominate the market over the forecast period and this is attributed to the increasing adoption of new technologies and the availability of advanced healthcare infrastructure and also the increasing prevalence of geriatric population which led to the increasing demand for gas analyzers.
Among all segmentation, the end user segment has the highest potential due to the increasing prevalence of chronic diseases such as cancer, strokes, and respiratory diseases over the forecast period. For instance, in June 2020, according to an article published in Journal of The Lancet Respiratory Medicine, an estimated 544.9 million individuals worldwide had a chronic respiratory disease, equivalent to a 39.8% increase compared with the number of individuals. The global prevalence in 2020 was around 7.1%.
Medical Purity Analyzer Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 478.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.0% | 2033 Value Projection: | USD 719.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AMETEK, Inc., California Analytical Instruments, Inc., Drägerwerk AG & Co. KGaA, Eaton, Emerson Electric Co., Honeywell International Inc., Spectris, Teledyne Technologies Incorporated, Vasthi Instruments, NOVAIR MEDICAL, Roscid Technologies, Linde plc, Praxair S.T. Technology, Inc., Fluke Process Instruments, MEECO1, and MATHESON TRI-GAS, INC. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Medical Purity Analyzer Market- Cross Sectional Analysis
Among type, the laser analyzer segment held a dominant position in North America over the forecast period owing to the increasing adoption of new technology and the availability of advanced healthcare infrastructure. In the North America region, the U.S. held the largest market share due to factors, such as increasing count of geriatrics, along with the growing healthcare industry, which is anticipated to stimulate the demand in this region. For instance, in July 2021, according to Administration for Community Living, in 2020, the population aged 65 was 54.1 million, among which 30 million were women and 24.1 million men.
Global Medical Purity Analyzer Market- Key Developments
In April 2022, Emerson Electric Co., a global technology and engineering company providing innovative solutions, expanded its ASCO Series 090 line of miniature solenoid valves, adding a new three-way Series 090 valve configuration to enable lighter, more space-efficient solutions for gas control in oxygen therapy, compression therapy, and gas analyzer devices.
In August 2022, QED ENVIRONMENTAL SYSTEMS, a provider of environmental technology solutions, announced the new LANDTEC BIOGAS 3000 fixed gas analyzer, a next-generation system offering continuous monitoring of the complete gas production process. Building on field-proven gas analysis technology, the compact, self-contained BIOGAS 3000 system offers cost-effective online monitoring that is ideal for anaerobic digestion, biogas monitoring and landfill gas monitoring applications. The new BIOGAS 3000 fixed gas analyzer can utilize up to four sample ports to monitor methane (CH4), carbon dioxide (CO2), and oxygen (O2), with optional monitoring of hydrogen sulfide (H2S), hydrogen (H2) and carbon monoxide (CO) levels. Operators can choose up to five gases to monitor.
In December 2022, Emerson Electric Co., a global technology and engineering company providing innovative solutions has announced the release of two new-to-the-world technologies for the Crosby J-Series Pressure Relief Valve (PRV) product line. The first is a Balanced Diaphragm, which eliminates the need for bellows in PRVs, while providing balanced operation against backpressure to lower cost of ownership and enhance performance. The second technology is Bellows Leak Detection, enabling remote detection of bellows ruptures in PRVs, with the capability to reduce and accurately calculate volumetric emissions in real-time.
On April 18, 2023, Teledyne FLIR, a part of Teledyne Technologies Incorporated, a provider of sophisticated electronic components, instruments, and communication products, debuted the new G-Series, a family of high-tech, cooled-core Optical Gas Imaging (OGI) cameras that can help Leak Detection and Repair (LDAR) professionals seamlessly locate and document harmful gas emissions. The G-Series is designed to empower everyday users in the oil and gas, manufacturing, steel, and utility industries to spend more time prioritizing leak repairs, and less time documenting them while gaining better insight into the severity of the emission.
Global Medical Purity Analyzer Market- Key Trends
Partnership by the key market players
Partnerships among the key market players to expand the product portfolio is expected to drive the market over the forecast period. For instance, in July 2021, Picarro, Inc., a provider of gas concentration and stable isotope analyzers and systems for scientific and industrial applications, and Sonoma Technology, Inc. (STI), a global provider of innovative, science- and technology-based solutions for environmental applications, announced that they have signed a channel partnership agreement. Through this agreement, Sonoma Technology, Inc., (STI) will combine Picarro, Inc., gas concentration monitoring technology with its expert services to provide comprehensive systems and solutions for measurement, monitoring and analysis of ethylene oxide, hydrogen sulfide, ammonia, and formaldehyde. Utilizing Picarro, Inc. analyzers to produce data with exceptional accuracy on the concentration of these hazardous chemicals in emissions gas streams and the environment, Sonoma Technology, Inc., (STI) clients will be better able to address industrial air quality compliance, environmental justice, community monitoring, and other challenges.
Global Medical Purity Analyzer Market: Restraints
Poor stability and greater environmental impact of medical analyzers
Factors such as poor stability and greater environmental impact of medical analyzers is expected to hamper the growth of the market over the forecast period. For instance, in January 2020, according to an article published by Avcray, a manufacturer of sensor for measuring oxygen concentration, gas medical analyzers provides poor stability and greater environmental impact on the analysis of the gases. In order to avoid this, poor stability and greater environmental impact, limiting current type gas analyzers to be used.
Global Medical Purity Analyzer Market- Key Players
The major players operating in the global medical purity analyzer market include AMETEK, Inc., California Analytical Instruments, Inc., Drägerwerk AG & Co. KGaA, Eaton, Emerson Electric Co., Honeywell International Inc., Spectris, Teledyne Technologies Incorporated, Vasthi Instruments, NOVAIR MEDICAL, Roscid Technologies, Linde plc, Praxair S.T. Technology, Inc., Fluke Process Instruments, MEECO1, and MATHESON TRI-GAS, INC.
*Definition: Medical purity analyzers are used in hospitals and healthcare facilities to evaluate, detect, and quantify pure or mixed medical gases. Instruments known as medical purity analyzers are used to identify and quantify the qualitative and quantitative content of pure medical gases or mixes of medical gases.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients