Mycoplasma Diagnostics Market Size and Forecast – 2026 – 2033
The Global Mycoplasma Diagnostics Market size is estimated to be valued at USD 1.75 billion in 2026 and is expected to reach USD 3.25 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 9.8% from 2026 to 2033.
Global Mycoplasma Diagnostics Market Overview
The mycoplasma diagnostics market offers a range of products designed for the detection and monitoring of Mycoplasma infections in humans, animals, and cell cultures. Key product types include molecular diagnostic kits, such as PCR-based assays, which provide rapid and highly sensitive detection. Serological tests detect antibodies against Mycoplasma species, while culture-based methods enable pathogen isolation and identification. Additionally, automated detection systems and point-of-care devices are gaining traction for quick, on-site testing. These products cater to clinical laboratories, research institutions, and pharmaceutical industries, supporting disease management, quality control in biologics, and improving overall diagnostic accuracy in healthcare and biotech applications.
Key Takeaways
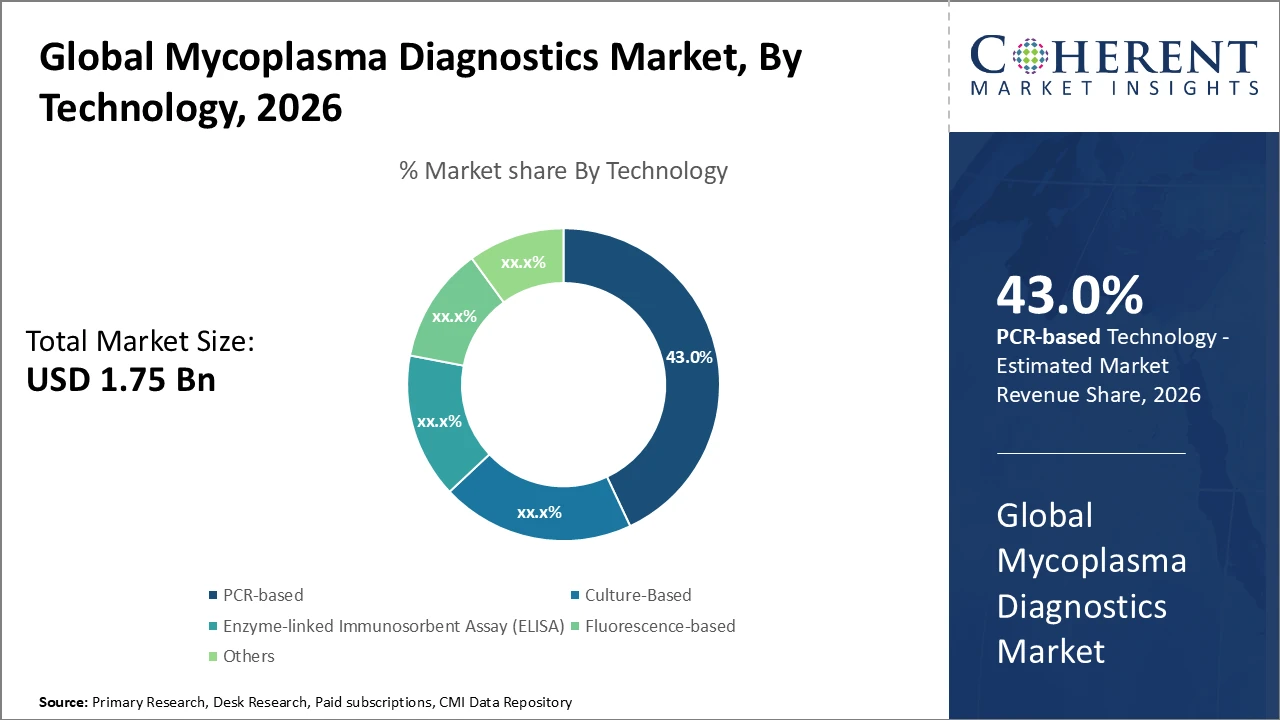
PCR-based technology remains the dominant segment, holding 43% of the market due to its high sensitivity and rapid testing capabilities, playing a key role in driving market growth.
In applications, clinical diagnostics leads in market size, driven by increasing respiratory infection screening campaigns. The biopharmaceutical industry represents the fastest-growing segment, fuelled by stringent contamination control requirements.
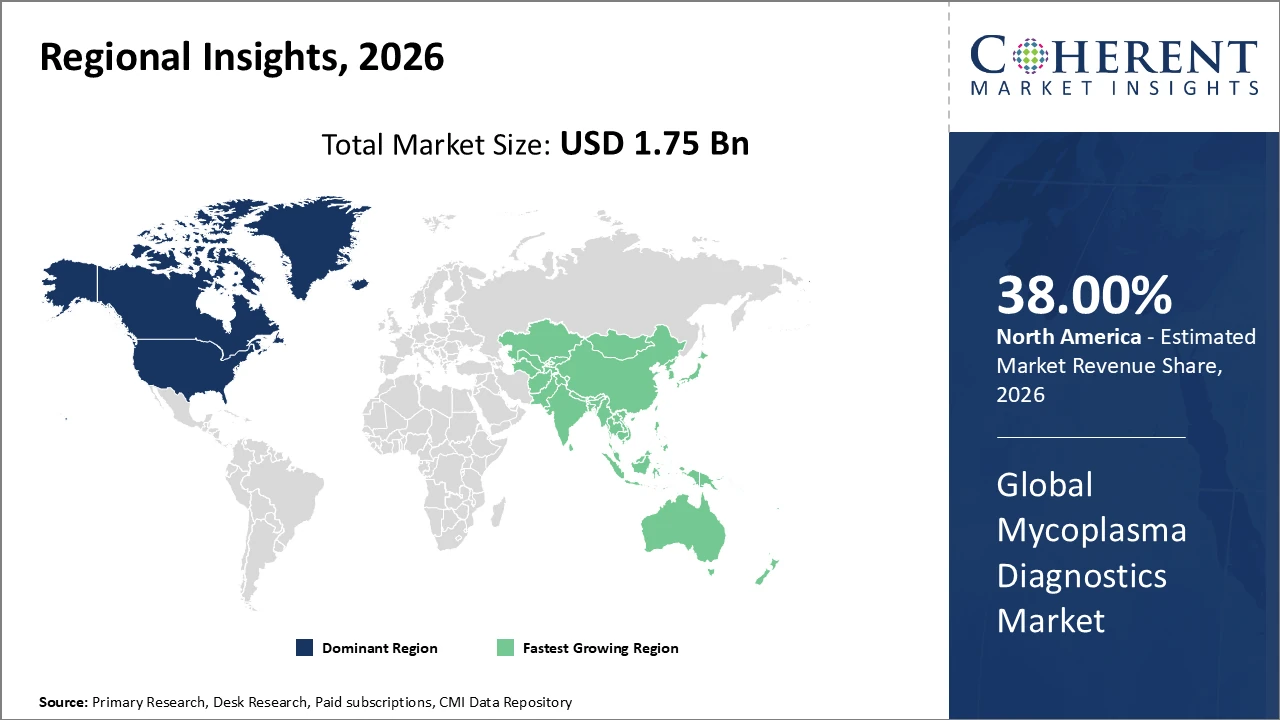
Regionally, North America holds the largest market share at over 38%, supported by advanced healthcare infrastructure and early adoption of cutting-edge diagnostic tools.
Asia Pacific is the fastest-growing region, with a CAGR above 11%, driven by improved healthcare accessibility and government funding initiatives.
Mycoplasma Diagnostics Market Segmentation Analysis
To learn more about this report, Request Free Sample
Mycoplasma Diagnostics Market Insights, By Technology
PCR-based technology leads the Mycoplasma Diagnostics market with a 43% share, driven by high sensitivity, specificity, and rapid turnaround compared to traditional culture methods. The integration of real-time and digital PCR platforms in clinical and biopharmaceutical testing enhances diagnostic accuracy, supporting widespread adoption. Fluorescence-based technology is the fastest-growing subsegment, favored for detecting low-level mycoplasma contamination in complex samples and enabling high-throughput automated workflows. Culture-based methods remain relevant for confirmatory testing and research due to cost-effectiveness. ELISA-based diagnostics provide immunoassay alternatives, while emerging approaches like biosensors and microfluidics offer future growth opportunities in the market.
Mycoplasma Diagnostics Market Insights, By End User
Hospitals and clinics dominate the Mycoplasma Diagnostics market, driven by expanding diagnostic screening programs and rising incidences of mycoplasma-related respiratory infections globally. Diagnostic laboratories represent the fastest-growing segment, supported by outsourcing trends and increased adoption of molecular diagnostics, with investments in high-throughput PCR platforms to serve both clinical and biopharmaceutical testing needs. Pharmaceutical and biotechnology companies hold a notable share due to regulatory requirements for stringent mycoplasma testing in drug development and manufacturing. Academic and research institutions generate niche demand through cell culture contamination studies and method development, while others, including government health agencies and contract research organizations (CROs), contribute moderately to overall market revenue.
Mycoplasma Diagnostics Market Insights, By Application
Clinical diagnostics dominates the mycoplasma diagnostics market, fuelled by large-scale screening for respiratory infections and growing awareness of antibiotic resistance, with hospitals generating significant testing demand and revenue. The biopharmaceutical industry is the fastest-growing application segment, driven by stringent regulatory requirements for mycoplasma contamination testing in biologics manufacturing, reinforced since 2026. The expansion of cell and gene therapies further increases the need for frequent, sophisticated testing to prevent severe contamination risks. Research laboratories maintain steady demand for detecting mycoplasma in cell culture models. Veterinary diagnostics is a smaller yet expanding segment, driven by rising awareness of infections in livestock and pets requiring rapid, cost-effective testing solutions.
Mycoplasma Diagnostics Market Trends
The mycoplasma diagnostics market is increasingly embracing digital transformation in testing workflows.
AI-enabled diagnostic platforms introduced in 2025 have reduced manual errors and improved testing throughput by up to 20%.
Multiplex diagnostics, allowing simultaneous testing for Mycoplasma and viruses such as RSV and Influenza, have gained wide adoption in clinical settings, especially following heightened infectious disease awareness post-pandemic.
Point-of-care (POC) testing devices with rapid, on-site diagnostic capabilities are growing in popularity, particularly in remote and emerging regions.
Asia Pacific saw a 28% increase in POC device deployment in 2026, reflecting expanding accessibility.
These trends indicate a shift toward faster, integrated, and more accessible diagnostics, supporting disease surveillance and patient management.
Mycoplasma Diagnostics Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Mycoplasma Diagnostics Market Analysis and Trends
In North America, the mycoplasma diagnostics market is dominated by a mature healthcare ecosystem featuring advanced diagnostic infrastructure, strong regulatory support, and significant R&D investments. The United States alone accounts for over 30% of the regional market share, driven by a robust biopharmaceutical industry and widespread clinical adoption of molecular diagnostic technologies. Key players, including Thermo Fisher Scientific and Roche Diagnostics, maintain headquarters and production facilities in the region, facilitating rapid deployment of innovations and streamlined supply chains. These factors collectively reinforce North America’s leadership in the global market, supporting sustained growth and technological advancement in mycoplasma diagnostics.
Asia Pacific Mycoplasma Diagnostics Market Analysis and Trends
Asia Pacific is the fastest-growing region in the mycoplasma diagnostics market, recording a CAGR above 11%. Growth is fuelled by rising healthcare expenditure, government initiatives targeting infectious disease control, and the expansion of biopharmaceutical manufacturing hubs in China and India. Improved access to advanced diagnostic technologies, including PCR and multiplex platforms, supports widespread adoption in both clinical and biopharmaceutical settings. Increasing awareness of mycoplasma-related infections among healthcare providers and patients further drives testing demand. These factors collectively enhance market revenue, positioning Asia Pacific as a key growth engine in the global mycoplasma diagnostics landscape over the coming years.
Mycoplasma Diagnostics Market Outlook for Key Countries
USA Mycoplasma Diagnostics Market Analysis and Trends
The U.S. Mycoplasma Diagnostics market holds a substantial share, driven by the rising prevalence of respiratory and urogenital mycoplasma infections reported by the CDC in 2025, which has increased demand for widespread diagnostic screening. Leading companies such as Thermo Fisher Scientific and Bio-Rad Laboratories have expanded their molecular testing portfolios, emphasizing rapid PCR diagnostics, resulting in a 14% revenue growth between 2024 and 2026. Furthermore, stringent FDA regulations for biologics manufacturing require rigorous mycoplasma testing, ensuring compliance and safety. These factors collectively sustain steady market growth, reinforcing the U.S. as a key contributor to the global mycoplasma diagnostics industry.
Germany Mycoplasma Diagnostics Market Analysis and Trends
Germany’s Mycoplasma Diagnostics market is expanding steadily, driven by stringent healthcare regulations and growing emphasis on infection control in clinical and biopharmaceutical settings. The adoption of advanced molecular diagnostics, particularly PCR and multiplex platforms, supports rapid and accurate detection of mycoplasma infections in respiratory, urogenital, and cell culture samples. Rising awareness of antibiotic resistance and the need for routine screening in hospitals and research laboratories further fuels demand. Biopharmaceutical companies in Germany increasingly invest in contamination testing for biologics, aligning with EU regulatory standards. Technological innovations, government support, and a strong healthcare infrastructure position Germany as a key contributor to Europe’s market growth.
Analyst Opinion
The surge in biopharmaceutical production has been a major quantitative driver for the Mycoplasma Diagnostics market. In 2025, over 65% of global bioprocessing facilities adopted advanced mycoplasma testing to ensure product safety, significantly boosting market revenue. Fast-turnaround molecular diagnostics increased testing efficiency by 20–25% compared to traditional culture-based methods.
Demand-side indicators show rising adoption of real-time PCR diagnostics, with a 30% improvement in diagnostic accuracy reported in 2024 across hospitals and research laboratories in North America. This precision supports early infection detection and containment, directly influencing market growth strategies.
Micro-indicators highlight stringent regulatory requirements worldwide; the FDA’s updated guidelines in 2026 for mycoplasma testing in cell-based therapies increased compliance-driven testing volumes by 18%, sustaining industrial demand.
Nano-size indicators reflect growing imports of high-sensitivity test kits in emerging markets such as India and Brazil, where import volumes doubled in 2025, indicating expanded market reach and increasing emphasis on quality diagnostics in developing healthcare infrastructures.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.75 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 3.25 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Thermo Fisher Scientific, Merck KGaA, Roche Diagnostics, Qiagen NV, Bio-Rad Laboratories, GenMark Diagnostics, Lonza Group AG, Hologic Inc., Abbott Laboratories, Seegene Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Mycoplasma Diagnostics Market Growth Factors
Key growth drivers in the Mycoplasma Diagnostics market include rising prevalence of pulmonary and urogenital infections, with CDC reports in 2025 highlighting increased clinical demand. Expansion of biopharmaceutical manufacturing has intensified contamination control requirements, boosting demand for rapid diagnostics, reflected by a 22% rise in bioprocess testing across Asia Pacific in 2026. Technological advancements, including real-time PCR and digital droplet PCR, have enhanced sensitivity and specificity, reducing false negatives and driving a 25% adoption increase in hospitals and research laboratories globally. Government initiatives to strengthen infectious disease diagnostic infrastructure in emerging economies, particularly India and Brazil, further expand market revenue and scope.
Mycoplasma Diagnostics Market Development
In 2025, Thermo Fisher Scientific launched an advanced real-time PCR assay for rapid mycoplasma detection, targeting both clinical and biopharmaceutical applications. The platform enabled high-throughput testing with improved sensitivity and turnaround time, reducing false negatives and supporting early infection management. Its adoption in hospitals and research laboratories across North America and Europe strengthened diagnostic workflows and contributed to increased market revenue.
Key Players
Leading Companies of the Market
Thermo Fisher Scientific
Merck KGaA
Roche Diagnostics
Qiagen NV
Bio-Rad Laboratories
Hologic Inc.
Lonza Group AG
Abbott Laboratories
GenMark Diagnostics
Seegene Inc.
Competitive strategies in the Mycoplasma Diagnostics market include Thermo Fisher Scientific’s acquisition of specialty assay developers in 2025, which expanded their North American market share by 12% and strengthened their molecular diagnostics portfolio. Similarly, Roche Diagnostics made significant investments in AI-driven diagnostic platforms targeting mycoplasma contamination control in Europe, resulting in a 15% increase in market revenue between 2024 and 2026. These strategic initiatives highlight the focus on portfolio expansion, technological innovation, and regional market penetration, enabling leading companies to consolidate their positions and drive growth in both clinical and biopharmaceutical testing segments.
Mycoplasma Diagnostics Market Future Outlook
The mycoplasma diagnostics market is poised for sustained growth, driven by rising prevalence of respiratory and urogenital infections, expanding biopharmaceutical production, and increasing demand for contamination-free biologics. Advances in molecular technologies, including real-time PCR, digital droplet PCR, and multiplex assays, are expected to enhance sensitivity, reduce false negatives, and accelerate diagnostic workflows. Growing adoption of AI-enabled and point-of-care testing platforms will improve accessibility and efficiency, particularly in emerging regions. Government initiatives to strengthen infectious disease diagnostic infrastructure, coupled with rising awareness of antibiotic resistance, will further expand market revenue. Overall, innovation, regulatory support, and global healthcare expansion will shape the market’s future trajectory.
Mycoplasma Diagnostics Market Historical Analysis
The Mycoplasma Diagnostics market has witnessed steady growth over the past decade, primarily driven by increasing awareness of mycoplasma-related respiratory and urogenital infections and the need for contamination control in biopharmaceutical production. Early market adoption focused on culture-based and ELISA methods, which, despite slower turnaround and lower sensitivity, were widely used in research laboratories and clinical settings. The introduction of PCR and molecular diagnostics in the mid-2010s marked a significant shift, offering rapid, accurate detection. Rising regulatory requirements, technological advancements, and expanding clinical and biopharmaceutical applications laid the foundation for consistent market expansion, setting the stage for future growth in global diagnostics.
Sources
Primary Research Interviews:
Executives and operations managers at mycoplasma diagnostics manufacturing and testing facilities
Clinical microbiologists and laboratory directors specializing in infectious disease testing
Technology providers for molecular diagnostics, PCR platforms, and point-of-care devices
Databases:
PubMed
Scopus
ClinicalTrials.gov
Embase
Others
Magazines:
Clinical Laboratory News – Diagnostic Innovations and Laboratory Trends
Molecular Diagnostics – PCR and Multiplex Assay Updates
BioProcess International – Biopharmaceutical Testing and Contamination Control
Laboratory Network News – Clinical and Research Diagnostics Developments
Journals:
Journal of Clinical Microbiology – Mycoplasma Detection and Diagnostic Methods
Journal of Molecular Diagnostics – PCR and Molecular Assay Research
Journal of Clinical Virology – Respiratory and Urogenital Mycoplasma Studies
Journal of Biotechnology – Biopharmaceutical Contamination Testing and Quality Control
Newspapers:
The Wall Street Journal – Diagnostics and Biotech Industry News
Financial Times – Healthcare, Biopharma, and Diagnostic Market Insights
The Guardian – Infectious Disease Awareness and Health Policy Updates
Science Daily – Clinical Diagnostics and Research Developments
Associations:
Clinical & Laboratory Standards Institute (CLSI) – Diagnostic Guidelines and Standards
International Society for Infectious Diseases (ISID) – Research and Laboratory Standards
American Society for Microbiology (ASM) – Microbiology and Diagnostic Research
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients