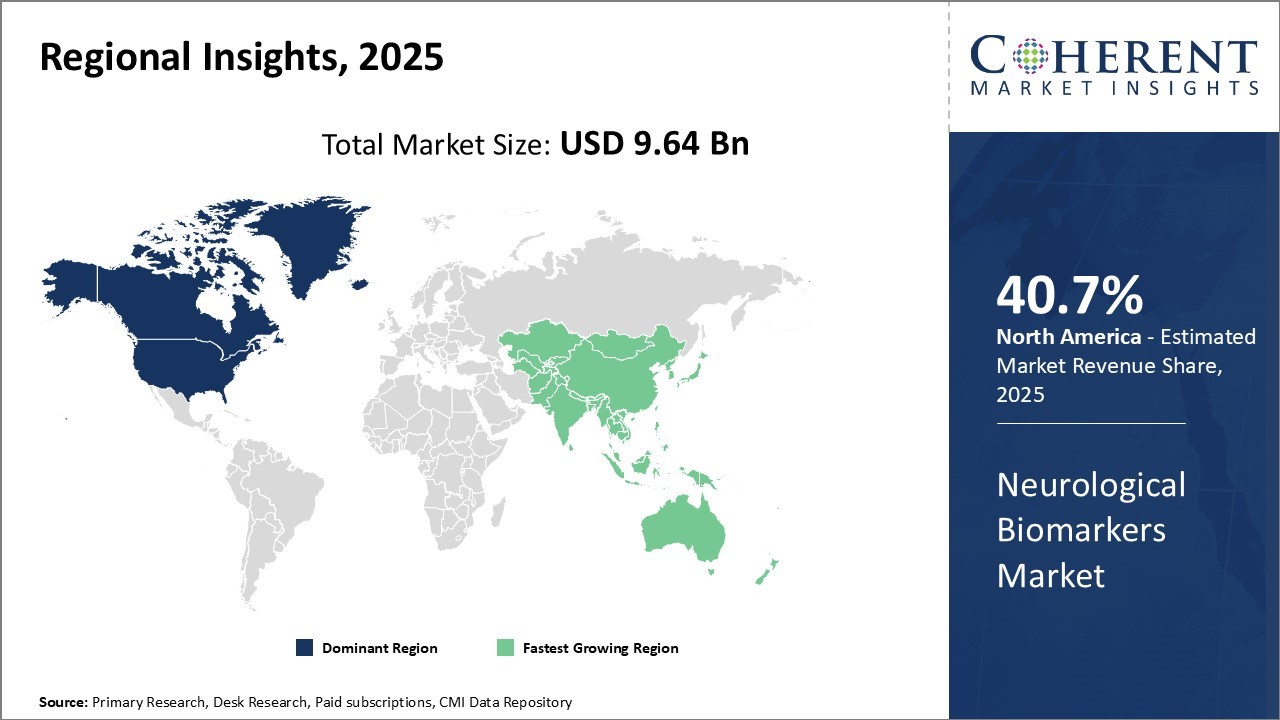
Global neurological biomarkers market is estimated to be valued at USD 9.64 Bn in 2025 and is expected to reach USD 23.12 Bn by 2032, exhibiting a compound annual growth rate (CAGR) 13.3% from 2025 to 2032.
To learn more about this report, Download Free Sample
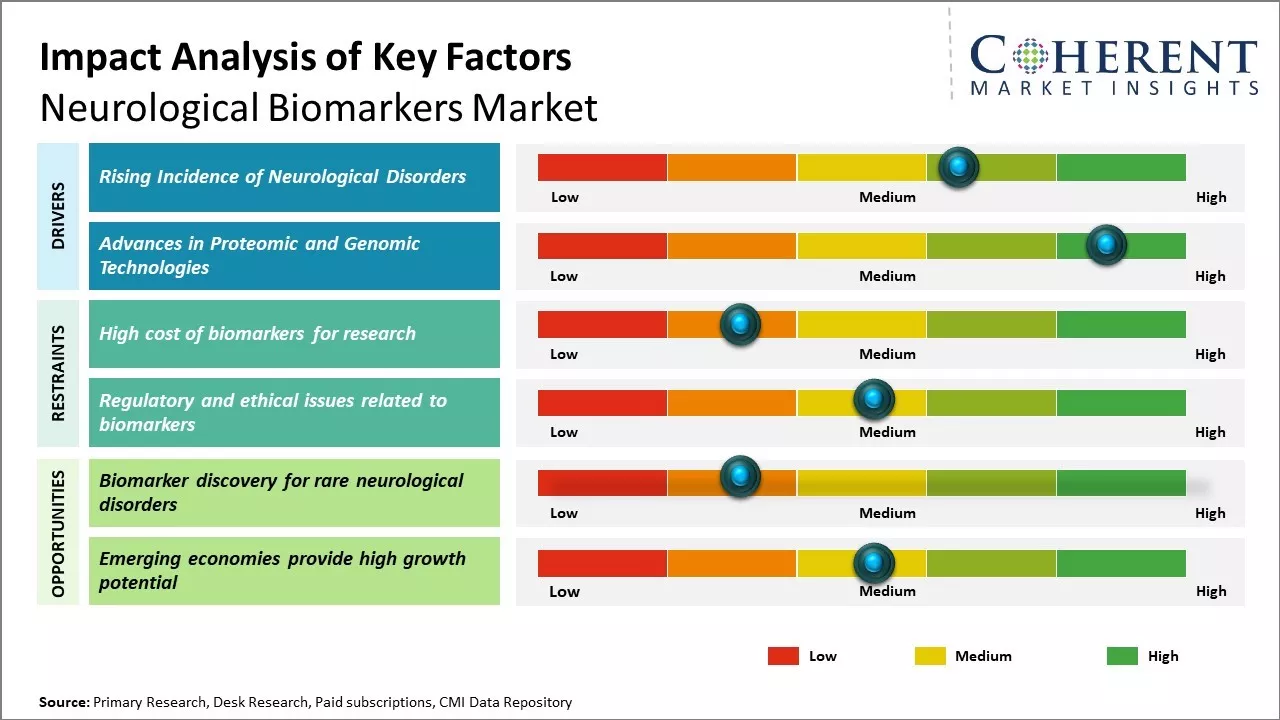
The global neurological biomarkers market is driven by rising prevalence of neurodegenerative diseases such as Alzheimer's, Parkinson's, and multiple sclerosis. The biomarkers are crucial for early diagnosis, disease monitoring, and improving treatment outcomes. Increasing demand for personalized medicine and neuroimaging and molecular diagnostics advancements are the primary drivers of growth in the market. Government funding of neurological research and the increasingly aged population, especially among the developed economies, also drive demand further.
|
Current Event |
Description and its impact |
|
Regulatory Support for Biomarker-Based Diagnostics |
|
|
Growing Integration of AI in Biomarker Research |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial Intelligence (AI) is playing an increasingly transformative role in the neurological biomarkers market by accelerating the discovery, validation, and application of biomarkers for neurological diseases. AI-powered algorithms can analyze vast datasets from medical imaging, genomics, proteomics, and clinical records with higher speed and precision than traditional methods.
Machine learning models also enhance biomarker validation by predicting disease progression and patient response to therapies, thereby improving personalized treatment strategies. Moreover, AI aids in integrating multi-modal data — including imaging, biochemical, and genetic information — providing a comprehensive understanding of disease mechanisms.
In May 2025, Renovaro Inc. launched Augusta, a Precision Neurology Platform designed for patient stratification, biomarker discovery, and drug development. Built on Renovaro’s Cube and Elion platforms, Augusta uses AI-driven multimodal data analytics and in silico screening to accelerate diagnosis and treatment in neurology, with recent validation in Parkinson’s disease and epilepsy.
To learn more about this report, Download Free Sample
The global market for neurological biomarkers is growing robustly, due to increasing incidence of neurological disorders globally. These are Alzheimer's disease, Parkinson's disease, epilepsy, multiple sclerosis, and traumatic brain injury, which are increasingly common and are now among the leading causes of death and disability.
According to a March 2025 WHO report citing The Lancet Neurology, over 3 billion people globally had neurological disorders in 2021, an 18% boost in disability and death attributable to the same since 1990. Over 35 million and 65 million people are affected by Alzheimer's disease and epilepsy, respectively.
Technological advances in genomics and proteomics are speeding biomarker identification and qualification. Technologies like mass spectrometry, next-generation sequencing, and bioinformatics facilitated the discovery of protein, genetic, and metabolic markers of neurodegenerative diseases. These technologies help identify biomarkers in cerebrospinal fluid and blood for Alzheimer's and discover genetic risk variants for Parkinson's. Imaging technologies further facilitate monitoring of brain structure and function.
The imaging biomarkers segment is likely to dominate the neurological biomarkers market in 2025. The growth is contributed primarily by advances in imaging technologies such as MRI, CT, PET scans, and sonography. These technologies offer precise, non-surgical visualization of structural and functional change in the brain and allow researchers and clinicians to monitor disease progression and treatment response more effectively. High-resolution imaging allows detection of neurological abnormalities early in the disease process at both the molecular and anatomical levels.
The Alzheimer’s disease segment is estimated to hold the highest market share in 2025. The rising global burden of Alzheimer's and the need for early, specific diagnosis are the primary reasons for this expansion. Alzheimer's is characterized by multifactorial, heterogeneous pathologies, and the diagnostic techniques available are not very specific. Despite this, studies on the discovery of valid biomarkers associated with distinct disease mechanisms such as amyloid beta deposition, tau protein deposition, and neuroinflammation are on the rise.
The hospital laboratories segment is projected to lead the neurological biomarkers market in 2025. Hospitals are the main diagnostic and treatment facilities for neurological disorders diagnosis , providing integrated services such as imaging, biochemical, and genetic testing. Such a centralized access within a single area simplifies patient care and enhances diagnostic efficiency.
To learn more about this report, Download Free Sample
North America is expected to lead the global neurological biomarkers market with maximum share in 2025. The leadership is a result of the advanced healthcare and research infrastructures available in this region. The United States and Canada are home to several major biotechnology and pharmaceutical firms, providing them with a leadership edge in the research and development of neurological biomarkers.
The Asia Pacific market is likely to be the fastest-growing market for neurological biomarkers during the forecast period. The growth is driven by development of healthcare infrastructure and increased spending on neurological treatment in emerging markets like China and India. A surge in economic growth in these nations has brought improved access to sophisticated diagnostic and treatment centers.
The United States has a dominant share in the North America neurological biomarkers market in 2025, the U.S. is at the forefront in the creation of innovative biomarker solutions for disease detection and treatment at an early stage of neurological disorders. Robust governmental funding by organizations such as the NIH drives neuroscience R&D in major universities and hospitals. Having sophisticated diagnostic labs, qualified experts, and high implementation of precision medicine guarantees large-scale application of neurological biomarkers in medical practice.
Germany is one of the dominant players in the neurological biomarkers industry in Europe because of its well-established healthcare system and emphasis on research and innovation. Research institutions in Germany are leading the field of neuroscience research, especially Alzheimer's and multiple sclerosis biomarker development. Public programs of funding and collaborative research with EU partners fuel biomarker research at the clinical and academic levels.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 9.64 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 13.3% | 2032 Value Projection: | USD 23.12 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
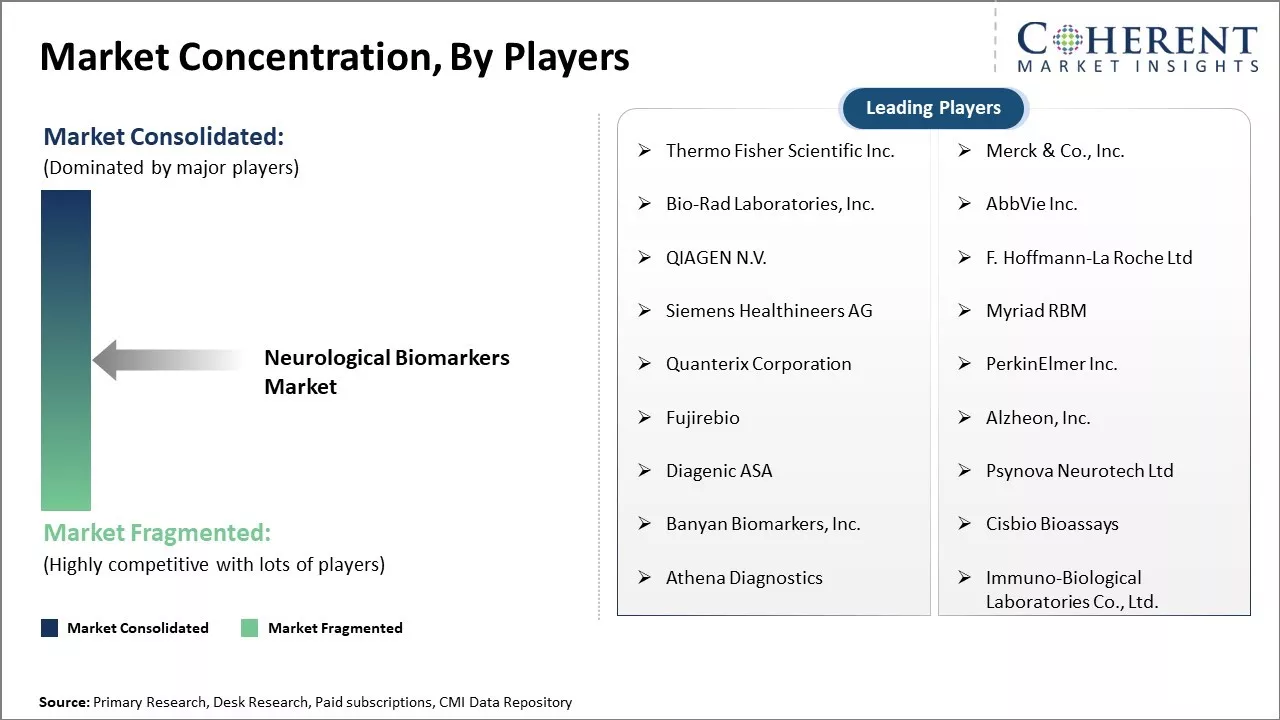
| Companies covered: |
Thermo Fisher Scientific Inc., Merck & Co., Inc., Bio-Rad Laboratories, Inc., AbbVie Inc., QIAGEN N.V., F. Hoffmann-La Roche Ltd, Siemens Healthineers AG, Myriad RBM, Quanterix Corporation, PerkinElmer Inc., Fujirebio, Alzheon, Inc., Diagenic ASA, Psynova Neurotech Ltd, Banyan Biomarkers, Inc., Cisbio Bioassays, Athena Diagnostics, Immuno-Biological Laboratories Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients