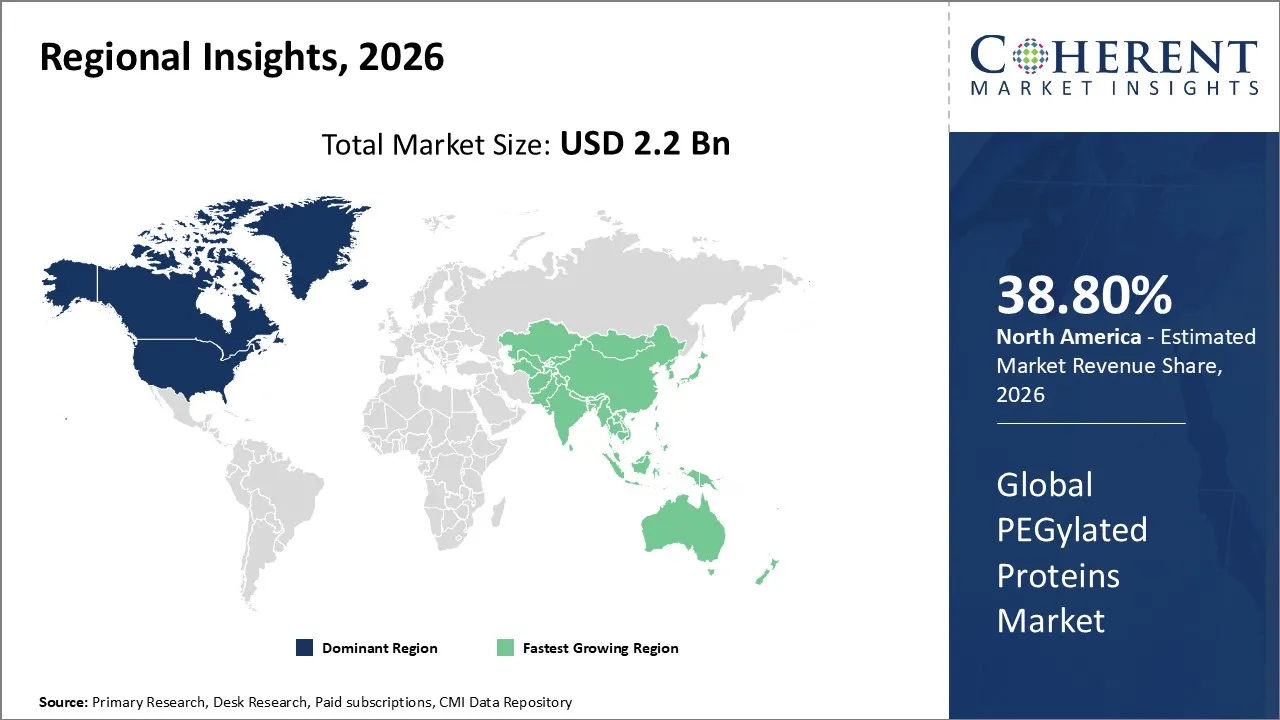
The PEGylated Proteins Market is estimated to be valued at USD 2.2 Bn in 2026 and is expected to reach USD 4.7 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 11.6% from 2026 to 2033.
Rising demand for advanced biologics to treat chronic diseases such as cancer and autoimmune disorders drives the PEGylated proteins market. PEGylation improves protein stability, solubility, and half-life, thereby enhancing therapeutic efficacy and patient compliance. Expanding biopharmaceutical research, growing adoption of long-acting drugs, and a strong clinical pipeline further support market growth. North America leads the market with its advanced healthcare infrastructure, while Asia-Pacific continues to emerge due to increasing investments and improving healthcare access.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments |
|
|
Technological Advancements in PEGylation and Delivery |
|
|
Geopolitical and Supply Chain Factors |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Consumables hold the largest market share of 53.7% in 2026. Routine laboratory workflows and large-scale biologics production drive the demand for consumables in the PEGylated proteins market. Researchers and manufacturers repeatedly use PEG reagents, linkers, and kits for conjugation processes, creating consistent demand. The expansion of protein-based drug development, especially for diseases like cancer, increases consumption across both research and manufacturing stages. Continuous innovation in PEGylation chemistries and rising outsourcing activities further boost the need for high-quality, ready-to-use consumable solutions in biopharmaceutical operations.
Colony-stimulating factor expected to hold largest market share of 36.6% in 2026. The essential role of colony-stimulating factors in managing chemotherapy-induced neutropenia in patients with cancer drives growth in the PEGylated proteins market. Healthcare providers increasingly administer PEGylated CSFs, which extend circulation time and reduce injection frequency, enhancing patient convenience and adherence. Expanding oncology treatment protocols and growing use in stem cell mobilization boost demand further. Ongoing product development and broader clinical applications encourage hospitals and specialty care centers to adopt PEGylated CSFs more widely. For instance, The U.S. FDA approved FILKRI™ (filgrastim-laha), Accord BioPharma’s biosimilar to NEUPOGEN®, to treat various conditions including chemotherapy-induced neutropenia, leukemia, bone marrow transplantation, severe chronic neutropenia, and radiation exposure.
Cancer acquired the prominent market share of 37.7% in 2026. The cancer segment drives growth in the PEGylated proteins market as PEGylation improves the effectiveness of protein-based oncology therapies. PEGylated proteins increase stability, solubility, and circulation time, allowing longer-lasting treatments with fewer doses, which benefits patients undergoing intensive chemotherapy. Rising cancer prevalence and expanding biologics pipelines boost demand further. By reducing toxicity and enabling targeted drug delivery, PEGylation encourages healthcare providers to adopt these therapies and supports ongoing development of innovative cancer treatments across research, clinical trials, and commercial production.
Pharmaceutical and Biotechnology Companies captures the largest market share of 42.8% in 2026. Pharmaceutical and biotechnology companies propel the PEGylated proteins market by applying PEGylation in drug development to boost protein stability, extend half-life, and enhance therapeutic performance. They actively use PEGylated technologies to develop long-acting biologics, increase patient adherence, and differentiate products in competitive pipelines. Growing research and clinical trials in oncology, autoimmune, and chronic diseases further expand their use of PEGylation. By partnering with contract research and manufacturing organizations and driving innovation in PEGylation chemistries, these companies maintain their position as the market’s primary end-users. For instance, in May 2025, the U.S. FDA approved Bayer’s Jivi for treating hemophilia A in patients aged 7 and older, with contraindications for those allergic to its components.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.8% in 2026. North America dominates the PEGylated proteins market, supported by its advanced healthcare infrastructure and robust biopharmaceutical ecosystem. Pharmaceutical and biotechnology companies in the region actively invest in research, development, and commercialization of PEGylated biologics. Rising prevalence of chronic diseases, especially cancer and autoimmune disorders, drives demand for safer, long-acting protein therapies. Healthcare providers rapidly adopt innovative PEGylation technologies, while strong clinical pipelines and supportive regulatory frameworks reinforce the market. Expanding collaborations and outsourcing further increase production capacity and accessibility across North America.
The Asia Pacific PEGylated proteins market grows rapidly as improving healthcare infrastructure and increasing biopharmaceutical investments drive adoption. Rising cases of chronic diseases, including cancer and autoimmune disorders, push demand for advanced protein therapies with longer half-lives and enhanced efficacy. Local pharmaceutical and biotechnology companies actively develop and commercialize PEGylated biologics, often collaborating with global partners. Expanding clinical research, increasing awareness among healthcare providers, and government initiatives to enhance healthcare access further accelerate the region’s adoption of PEGylated protein therapies.
The United States leads the PEGylated proteins market, supported by its advanced healthcare system and strong pharmaceutical and biotechnology sectors. Rising cases of chronic diseases, including cancer and autoimmune disorders, drive demand for safer, long-acting protein therapies. U.S. companies actively develop, research, and commercialize PEGylated biologics, while healthcare providers adopt innovative PEGylation technologies to enhance patient outcomes. Expanding clinical trials, supportive regulatory policies, and collaborations with contract research and manufacturing organizations further reinforce the country’s dominance in the PEGylated proteins market. For instance, Bio-Rad Laboratories launched two new chromatography media—CHT Ceramic Hydroxyapatite XT Media and Nuvia HP-Q Resin—for protein purification.
China drives growth in the PEGylated proteins market as improvements in healthcare infrastructure and increasing government support encourage adoption of advanced biologics. Rising cases of chronic diseases, including cancer and autoimmune disorders, push demand for long-acting, effective protein therapies. Domestic pharmaceutical and biotechnology companies actively develop and commercialize PEGylated biologics, frequently partnering with global firms to access innovative technologies. Expanding clinical research, increasing awareness among healthcare providers, and investments in manufacturing capabilities further accelerate China’s adoption of PEGylated protein therapies, strengthening its regional and global market presence.
The market is shifting toward long-acting PEGylated proteins as healthcare providers prioritize patient convenience and adherence. By extending half-life and reducing dosing frequency, PEGylated biologics improve therapeutic outcomes for chronic diseases like cancer, autoimmune disorders, and hematological conditions. Pharmaceutical and biotechnology companies increasingly integrate PEGylation in protein therapeutics to create competitive, patient-friendly products. This trend also encourages ongoing research into novel PEGylation methods that maintain efficacy while improving pharmacokinetics and minimizing side effects.
Cancer treatments dominate the PEGylated proteins market, as PEGylation enhances stability and targeted delivery of oncology biologics. Researchers and clinicians increasingly rely on PEGylated proteins for chemotherapy support, growth factor therapies, and immuno-oncology applications. Rising prevalence of various cancers and demand for safer, long-acting treatments drive adoption. Companies are developing PEGylated protein therapeutics that reduce toxicity, improve tumor-specific accumulation, and offer consistent dosing schedules, reflecting a sustained trend toward integrating PEGylation in oncology pipelines.
Pharmaceutical companies have the opportunity to develop long-acting PEGylated proteins that reduce dosing frequency and improve patient adherence. Chronic diseases such as cancer, autoimmune disorders, and blood disorders require sustained therapy, creating demand for extended half-life proteins. Companies can leverage novel PEGylation techniques to enhance protein stability and solubility, creating differentiated products that offer competitive advantages in crowded therapeutic areas while meeting unmet patient needs and improving overall treatment outcomes.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.2 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.6% | 2033 Value Projection: | USD 4.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Biogen Inc, Pfizer Inc, UCB S.A., Leadiant Biosciences, Inc., Amgen Inc., Thermo Fisher Scientific Inc., Horizon Therapeutics Plc., Novo Nordisk A/S, F. Hoffmann-La Roche AG, Bayer AG, BioMarin, RedHill Biopharma Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients