The Global Non-PVC IV Bags Market is estimated to be valued at USD 2.71 Bn in 2026 and is expected to reach USD 5.58 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.9% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The growth of the global non-PVC IV bags market is attributed to increasing awareness regarding benefits of non-PVC IV bags among healthcare facilities. Non-PVC IV bags reduce exposure to toxic plasticizers like DEHP and support better clinical outcomes. They are latex-free and DEHP-free, making them safer for patients including newborns, pregnant women, and persons undergoing long-term infusion therapy. Market players are investing in enhancing production capabilities of non-PVC materials like polypropylene and copper to meet the growing demand. Adoption of non-PVC IV bags is expected to further propel supported by regulatory guidelines promoting use of safer medical devices.
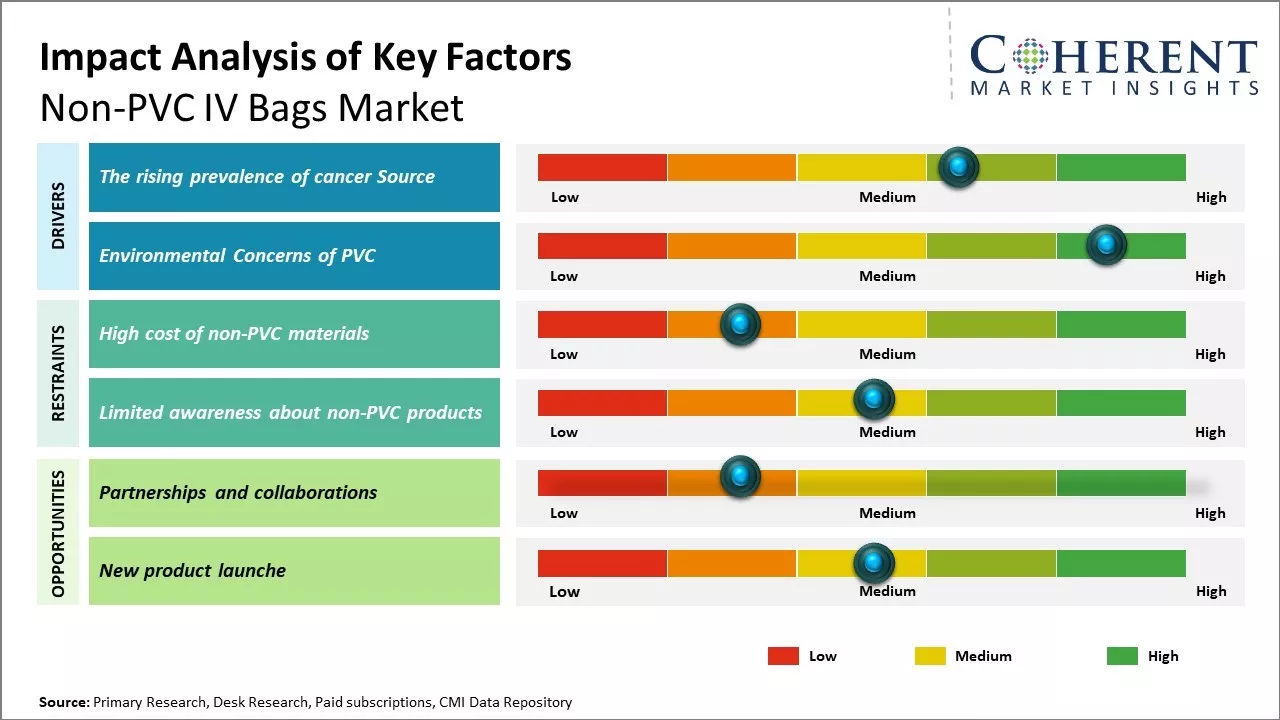
The Rising Prevalence of Cancer Source
The rising burden of cancer cases across the globe is a key factor driving the growth of the non-PVC IV bags market. Cancer being one of the leading causes of mortality worldwide has increased the demand for safer and better IV solutions for chemotherapy and other cancer treatments. Traditional PVC based IV bags can potentially leach toxic plasticizers like DEHP (diethylhexyl phthalate) into the fluids being administered to cancer patients. Such plasticizers have been found to act as endocrine disruptors and pose health risks especially to immunocompromised cancer patients. There is evidence that prolonged exposure to DEHP during chemotherapy can influence treatment outcomes negatively. This has increased awareness among healthcare professionals about the need for safer alternatives to PVC.
Non-PVC IV bags made of materials like polyethylene, polypropylene, and EVA are free from plasticizers and considered safer for cancer patients. They prevent potential toxic leaching and offer effective fluid administration. With rising cancer burden expected to continue in the coming years, the need for safer IV technologies throughout diagnosis, treatment and survivorship care is growing. For Instance, according to the January 2022 update by Breastcancer.org, around 287,850 new cases of invasive breast cancer are projected in the U.S., compared to 276,480 in 2020. India reported 18.3% of new cancer cases in 2020, with cervical cancer comprising 9.4% of all cases. Brazil recorded 300,114 new male cancer cases in 2020, with 97,278 cases being prostate cancer. Additionally, 292,098 new female cancer cases were reported in Brazil in 2020. This surge in cancer cases is expected to drive the demand for PVC-free IV bags due to the nutritional needs arising from cancer treatments, fostering market growth.
Get actionable strategies to beat competition: Request Free Sample
Environmental Concerns of PVC
The use of PVC in IV bags has raised significant concerns regarding its negative impact on the environment. PVC is considered as one of the most environmentally challenging plastics due to the chemicals required in its production and processing. It utilizes large amounts of chlorine in its manufacturing which leads to the release of dioxins - a group of toxic compounds. Dioxins are considered as persistent organic pollutants which can remain in the environment for very long periods. They are transferred between air, water and land and thus can travel long distances. Numerous studies have shown they can bioaccumulate in humans and wildlife. Due to this, they have been linked to various diseases and developmental and reproductive disorders in humans as well as wildlife. The disposal of PVC medical waste is also problematic. As PVC items do not degrade naturally, most of the used PVC items including IV bags ultimately end up in landfills. In landfills, the chlorine present in PVC can leach out into the soil and pollute the groundwater. Growing eco-consciousness among patients, healthcare professionals as well as stakeholders have created demand for safer alternatives to PVC that are free from such toxic elements. Many views non-PVC materials as a more sustainable option that can address both environmental and health risks associated with traditional PVC-based products.
Key Takeaways from Analyst
The global non-PVC IV bags market is expected to witness significant growth over the forecast period driven by the increasing demand for eco-friendly and biodegradable medical solutions. Non-PVC materials like copolymers provide a safer alternative to PVC as they do not emit toxic plasticizers like DEHP and phthalates during processing and administration. This will drive hospitals and healthcare centers across major markets to adopt non-PVC IV bags.
North America currently dominates the non-PVC IV bags market and is expected to remain the largest market over the next five years. Stringent regulations regarding the use of medical plastics along with the presence of many key market players in the region is driving the market growth. Asia Pacific is predicted to be the fastest growing regional market due to rising healthcare expenditure and increasing incidence of chronic diseases.
However, high manufacturing and transportation cost of non-PVC materials compared to PVC could hinder the market growth. Also, the lack of awareness about health impacts of PVC in developing regions poses a challenge. At the same time, growing preference for eco-friendly and safe healthcare solutions, especially in developing Asian markets, is anticipated to create new opportunities for non-PVC IV bag manufacturers. Furthermore, innovations in terms materials and bag designs are allowing players to capture more market share.
Market Challenges: High cost of non-PVC materials
The high cost of non-PVC materials is significantly restraining the growth potential of the global non-PVC IV bags market. Non-PVC materials like polypropylene, polyethylene, etc. that are used in manufacturing IV bags are generally more expensive than PVC resins. This price differential can be substantial at times. For example, according to data published by the Organisation for Economic Co-operation and Development (OECD) in 2022, the average price of polypropylene resins was around 30-40% higher than that of PVC resins. The healthcare industry operates on very low profit margins, where even small increases in input costs can affect overall profitability. Non-PVC materials push up the manufacturing price of IV bags which healthcare providers may find difficult to bear. This acts as a deterrent for them to switch from conventional and cheaper PVC-based IV bags to newer non-PVC options. Even though non-PVC materials help address environmental and health concerns associated with PVC, their premium price makes it challenging for healthcare facilities in price sensitive regions and lower income countries to adopt non-PVC bags.
Market Opportunities: Increased Partnerships and collaborations
Partnerships and collaborations could provide tremendous opportunities for growth in the global non-PVC IV bags market. As environmental and sustainability concerns rise globally, more hospitals, healthcare facilities, and regulatory agencies are seeking alternatives to traditional PVC materials. Non-PVC IV bags made from materials like polyolefins offer a greener solution and allow manufacturers to help address these important issues.
Through strategic partnerships along the supply chain, companies in this market can scale up production of non-PVC options in a more efficient and cost-effective manner. For example, bag manufacturers partnering with raw material producers gives both greater insight into challenges and improvements needed. This also ensures bag makers have a stable source of inputs to meet rising demand. Similarly, hospitals establishing long-term agreements with key suppliers ensures a reliable supply of non-PVC bags. This gives healthcare providers confidence in their ability to offer environmentally-friendly solutions without risk of shortages.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
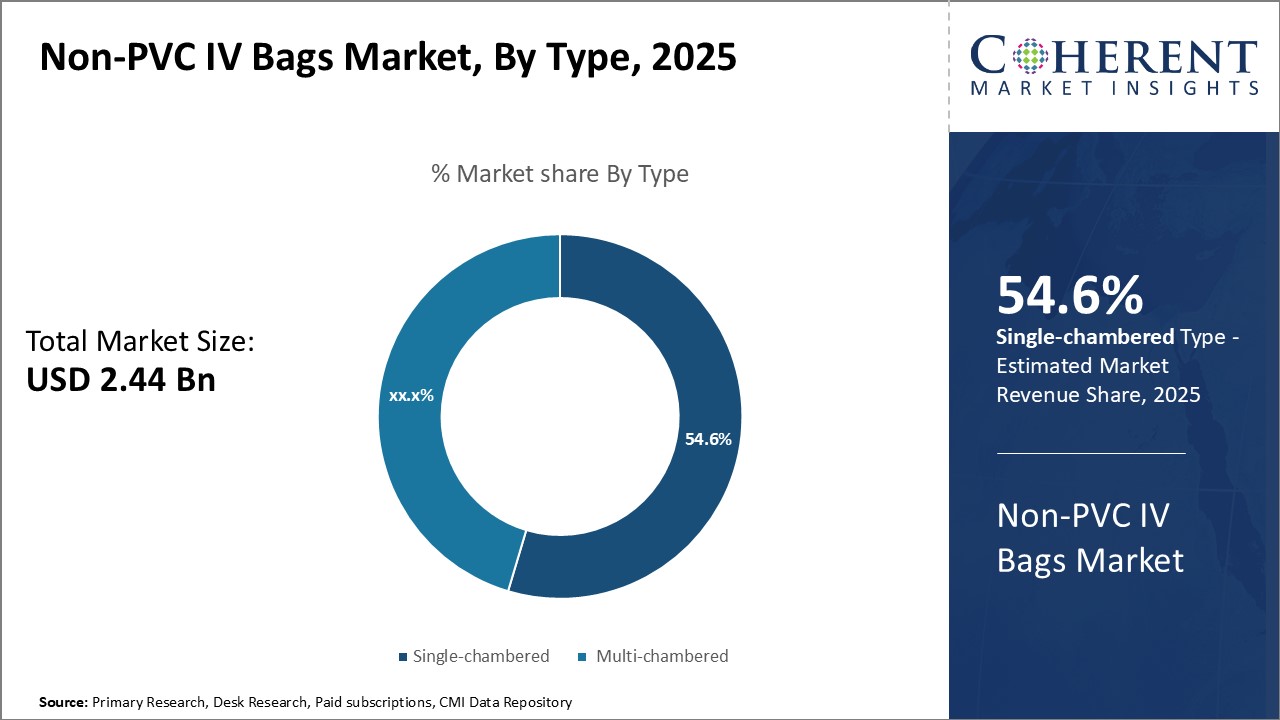
By Type, Focus on Convenience drives Single-chambered IV Bags adoption
By Type, the Single-chambered segment is estimated to hold the highest share of 55.6% in 2026, owing to its inherent convenience over multi-chambered bags. Single-chambered bags contain all diluted medication or fluid in a single compartment, making it effortless for medical staff to hang and administer without multiple connections or attachments. This simplified design reduces the preparation time at the point-of-care and streamlines the workflow in busy healthcare settings. Single-chambered bags are also preferred over multi-chambered for short term or emergency use where quick treatment is critical. Their unitary structure enables swift medicament administration without additional administration sets or complicated mixing steps. Furthermore, single-chambered bags minimize the risk of dosing errors or contamination that can potentially occur due to improper mixing of drugs stored in multiple chambers. Their user-friendly format is well-suited for acute care needs as well as home healthcare, driving greater uptake.
To learn more about this report, Request Free Sample
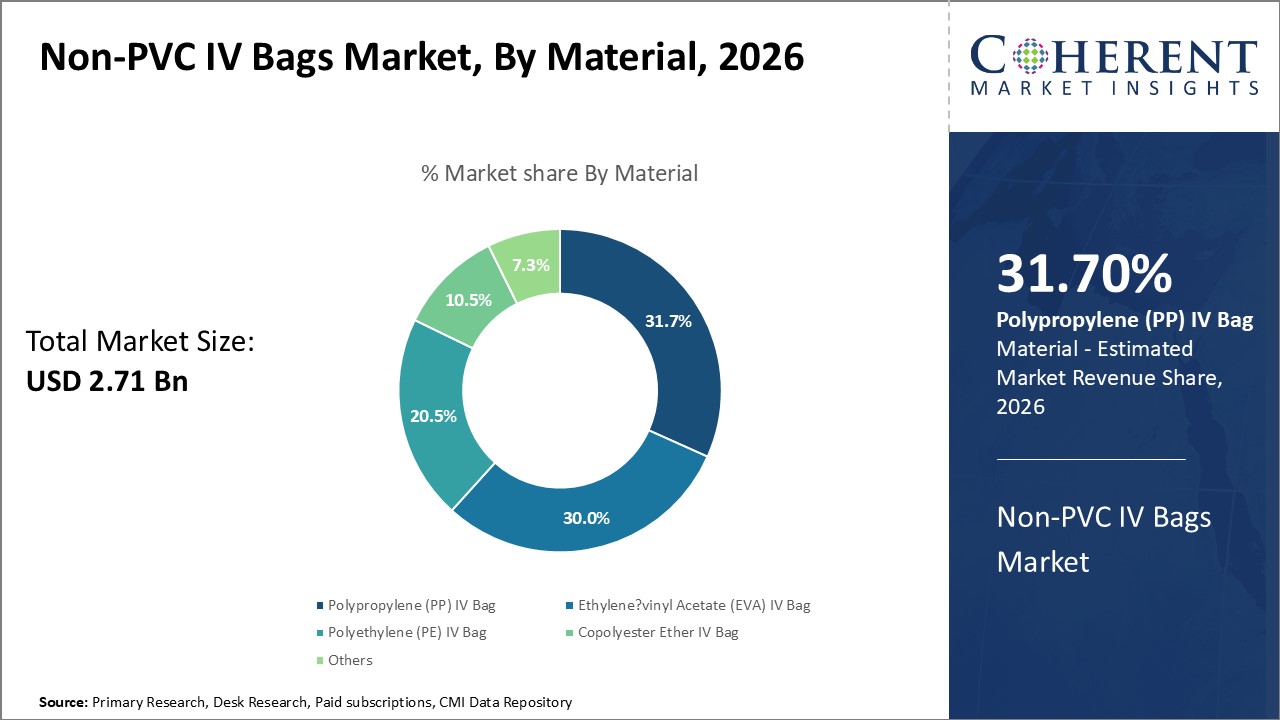
By Material, the Polypropylene (PP) IV Bag segment is Dominating the market
By Material, the Polypropylene (PP) IV Bag segment is estimated to hold the highest share of 31.7% in 2026, owing to its established safety profile and effectiveness. Polypropylene offers outstanding chemical resistance allowing it to be sterile-packed and gamma-ray sterilized for clinical applications. It does not interact with medicaments or infusion solutions, precluding the release of extractables. Moreover, polypropylene demonstrates high mechanical strength enabling it to withstand handling, hanging, and pressure-induced stresses during fluid flow without rupturing or leaching. Its glass-like clarity also facilitates easy visualization and inspection of contents. Most importantly, polypropylene has demonstrated biological inertness and is non-pyrogenic, translating to enhanced biocompatibility. This ensures safe administration over extended infusion periods without precipitation or interaction risks. Owing to its material properties, PP fully meets the purity and performance standards for medical-grade containers.
By Application, Chemotherapy dominates on prevalence of cancer & efficacy of treatment
By Application, the Chemotherapy segment is estimated to hold the highest share of 41.8% in 2024, owing to the high prevalence and incidence of cancer worldwide. According to the WHO, cancer burden has risen to over 10 million new cases in 2020 and is expected to further increase in the coming decades. Chemotherapy remains one of the principal modalities for treating curable as well as advanced-stage cancers owing to its effectiveness against a wide range of malignancies. It is extensively adopted for circulating anti-cancer drugs throughout the body via intravenous injection or infusion using non-PVC containers. Chemotherapy regimens often require the administration of multiple potent medications sequentially or concurrently in outpatient and inpatient hospital settings over long durations. Non-PVC IV bags are perfectly suited for the safe delivery of chemotherapeutics while avoiding material-mediated risks. This is driving their widespread uptake among oncology centers and cancer care hospitals.
Need a Different Region or Segment? Request Free Sample
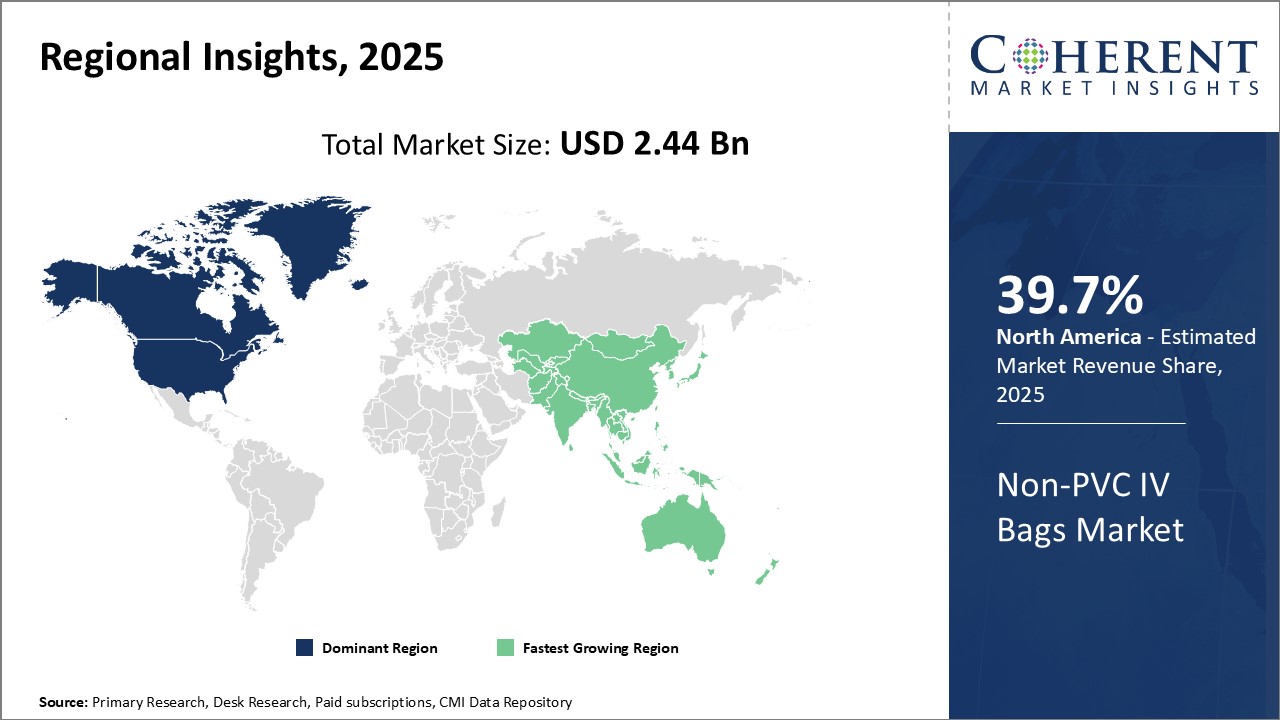
North America has been the dominant region in the global non-PVC IV bags market and is estimate to hold a market share of 39.7% in 2026. This can mainly be attributed to stringent regulations pertaining to the use of plasticizer DEHP in medical devices. In recent years, the U.S. Food and Drug Administration (FDA) has taken a strong stance against the use of DEHP due to potential adverse health effects. As a result, major manufacturers have accelerated their production capabilities of non-PVC alternatives while hospitals and healthcare providers are replacing existing plasticized inventory. Moreover, North America is home to several prominent multinational companies that have established manufacturing facilities and supply chain infrastructure catering to the demands of local hospitals. This helps ensure product and material availability along with supply security.
Over the past five years, the Asia Pacific region has emerged as the fastest growing market for non-PVC IV bags globally. This growth can be linked to rising healthcare expenditures, growing patient volumes, and increasing awareness regarding product safety among medical facilities in developing Asian countries. China, in particular, has built robust local production capacities as it aims to reduce import dependence for medical supplies. Local companies have gained valuable expertise through technical collaborations with international leaders. Additionally, regional trade agreements have strengthened intra-Asia commerce of non-PVC bags. Overall market conditions in the region such as evolving regulations, healthcare reforms, and rising disposable incomes will continue propelling the non-PVC IV bags segment at a rapid pace in Asia Pacific.
Non-PVC IV Bags Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.71 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.9% | 2033 Value Projection: | USD 5.58 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
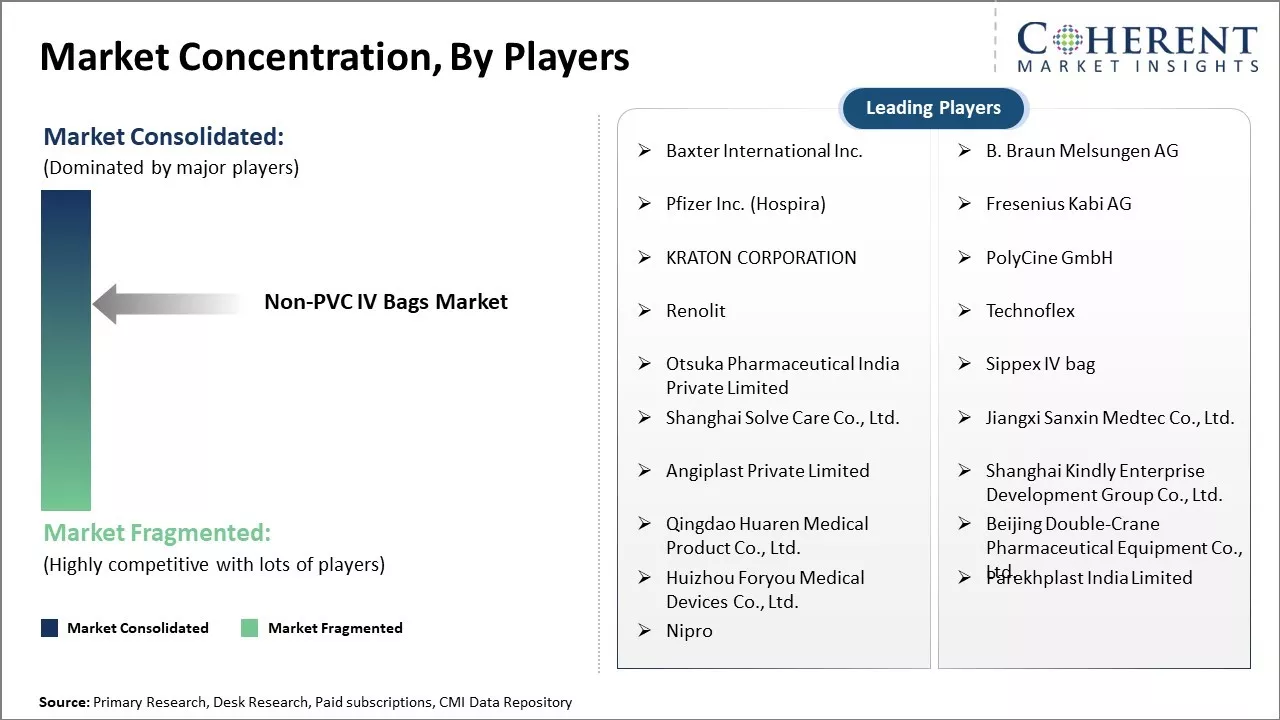
Baxter International Inc., B. Braun Melsungen AG, Pfizer Inc. (Hospira), Fresenius Kabi AG, KRATON CORPORATION, PolyCine GmbH, Renolit, Technoflex, Otsuka Pharmaceutical India Private Limited, Sippex IV bag, Shanghai Solve Care Co., Ltd., Jiangxi Sanxin Medtec Co., Ltd., Angiplast Private Limited, Shanghai Kindly Enterprise Development Group Co., Ltd., Qingdao Huaren Medical Product Co., Ltd., Beijing Double-Crane Pharmaceutical Equipment Co., Ltd., Huizhou Foryou Medical Devices Co., Ltd., Parekhplast India Limited, and Nipro |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients