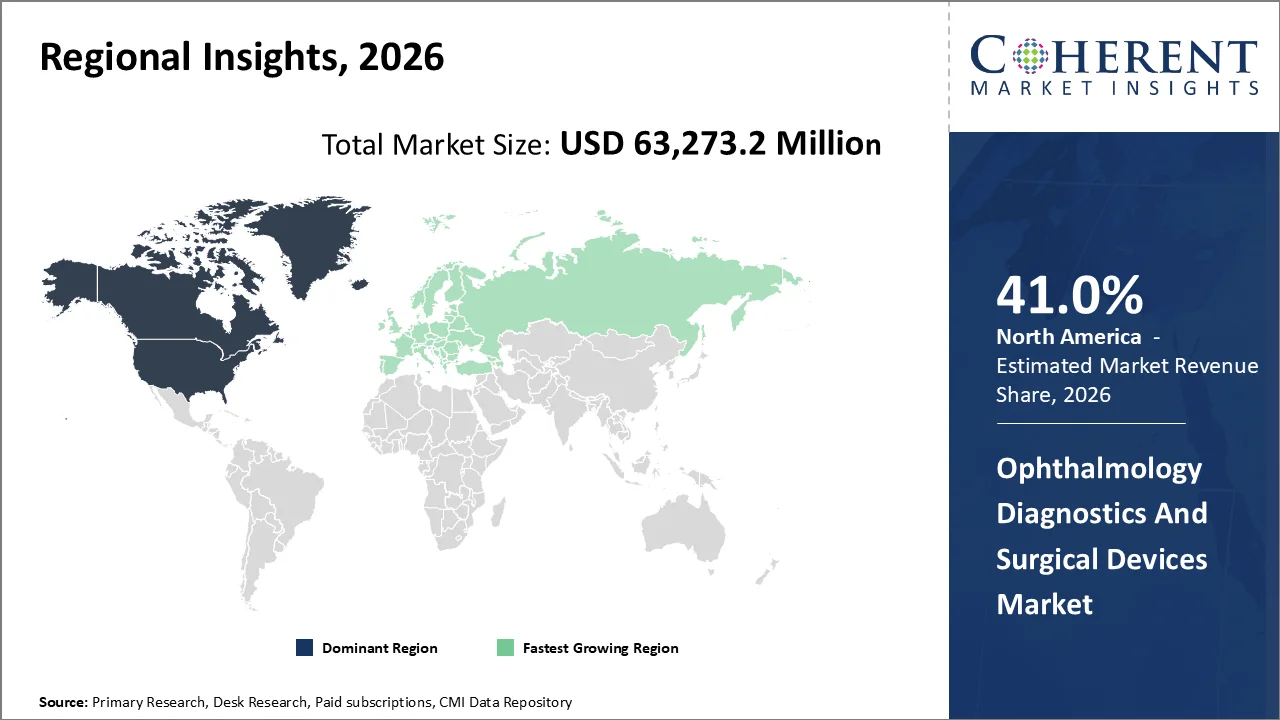
The Ophthalmology Diagnostics and Surgical Devices Market is anticipated to grow at a CAGR of 6.8% with USD 63,273.2 Mn in 2026 and is expected to reach USD 98,950.1 Mn in 2033. Demand for ophthalmology diagnostic and surgical devices is being sustained by a large untreated vision burden and procedure-linked diagnostic need. World Health Organization reported in February 2026 that at least 2.2 billion people have vision impairment, with cataract and refractive error remaining the leading causes globally. U.S. surveillance also shows 9.6 million people living with diabetic retinopathy, supporting demand for OCT, fundus imaging, and surgery.
Vision care is projected to account for the largest share of ophthalmology diagnostics and surgical devices market in 2026, representing approximately 52.0% of the total volume. Vision care is the dominant segment because it serves the largest and most recurring pool of ophthalmology patients, especially those needing refractive correction rather than one-time surgery. Its leadership is supported by the large burden of refractive errors, continuous demand for spectacles and contact lenses, repeat replacement cycles, rising myopia management needs in children, broader accessibility compared with surgery, and strong adoption through retail optical chains, clinics, and e-commerce. The segment also benefits from lower patient hesitation, faster product innovation in lenses and coatings, and regular follow-up requirements, all of which create steady and recurring demand.
For instance, on February 10, 2026, according to the data published by the WHO’s fact sheet states that at least 2.2 billion people globally have near or distance vision impairment, and identifies refractive errors and cataracts as the leading causes; WHO also notes that 2 out of 3 people in low-income countries who need eyeglasses lack access.
In addition, on September 25, 2025, the U.S. FDA authorized EssilorLuxottica’s Essilor Stellest lenses after clinical data showed a 71% reduction in myopia progression and 53% reduction in axial elongation at 24 months, highlighting strong innovation-led demand in spectacle lenses. CDC further reports that approximately 6 million Americans have vision loss and 1 million have blindness, sustaining long-term demand for corrective vision products and follow-up care.
An increasing geriatric population drives the Ophthalmology Diagnostics and Surgical Devices Market because the main vision disorders that require screening, imaging, and surgery rise sharply with age.
In February 2025, according to the data published by the World Health Organization states the number of people aged 60 and older worldwide is projected to grow from 1.1 billion in 2023 to 1.4 billion by 2030, while the U.S. Census Bureau reported the 65+ population rose to 61.2 million in 2024, up 3.1% from 2023. Older adults are at higher risk of cataract, glaucoma, and age-related macular degeneration; in January 2026, National Eye Institute stated that people over age 60 are at higher risk for glaucoma, and in February 2026 described AMD as a leading cause of vision loss in people over 50. This expands demand for OCT, fundus cameras, tonometers, and cataract and retinal surgery systems.
Increasing ophthalmic diseases drive the ophthalmology diagnostics and surgical devices market by expanding the number of patients who need screening, diagnosis, ongoing monitoring, and surgical intervention. In February 2026, World Health Organization, reported that at least 2.2 billion people globally have near or distance vision impairment, and in at least 1 billion cases the condition was preventable or remains unaddressed, indicating a very large pool requiring ophthalmic care. WHO also identifies cataract, refractive error, glaucoma, age-related macular degeneration, and diabetic retinopathy among the major causes of visual impairment, directly supporting demand for fundus cameras, slit lamps, tonometers, ophthalmoscopes, OCT systems, and cataract and retinal surgery devices. The same trend is visible in the U.S. disease burden. According to the Centers for Disease Control and Prevention estimates that 9.6 million people in the U.S. were living with diabetic retinopathy in 2021, including 1.84 million with vision-threatening diabetic retinopathy, which supports rising use of retinal imaging and treatment-linked diagnostics. CDC also estimates that 19.8 million Americans aged 40 and older were living with age-related macular degeneration in 2019, including 1.49 million with vision-threatening AMD.
Additionally, in February 2026, according to the World Health Organization reports uncorrected refractive error is the leading cause of vision impairment, while the National Eye Institute stated in January 2026 that as many as half of people with glaucoma do not know they have it. These trends directly support demand for OCT, fundus cameras, slit lamps, tonometers, and cataract and retinal surgery devices.
Technological advancement and innovation drive the ophthalmology diagnostic and surgical devices market by making eye care more accurate, faster, safer, and more outcome-oriented. When new devices improve disease detection, shorten procedure time, enhance visual outcomes, or widen the range of treatable patients, hospitals and eye-care centers are more willing to upgrade equipment and surgeons are more likely to adopt premium technologies. That directly supports replacement demand, procedure growth, and higher device ASPs across diagnostics and surgery.
The ophthalmology diagnostics and surgical devices market grow not only because of first-time adoption, but also because innovation pushes clinics to replace functioning equipment with better-performing systems. On April 6, 2026, Alcon, Ophthalmology company announced the U.S. launch of Clareon TruPlus, saying the new IOL is designed to increase depth of focus while maintaining high-quality distance vision. Alcon also stated that TruPlus demonstrated a 3.5-letter improvement in simulated visual acuity at -1.5 diopters (66 cm) versus Clareon Monofocal, and that the company would feature more than 60 scientific presentations at ASCRS 2026. Separately, in February 2026, Alcon reported that new product launches had helped accelerate sales. This is a direct example of how innovation converts into commercial growth.
Source: Alcon; https://investor.alcon.com
|
Current Event |
Description and its Impact |
|
Increasing Approval from the Regulatory Bodies |
|
|
Shifts in Global Data Privacy Regulations & Increasing Government Initiatives |
|
|
Government Policy Support / Public Healthcare Initiative |
|
|
Reimbursement and Healthcare Coding Policy–Driven Growth Factor |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Source: WHO
To learn more about this report, Request Free Sample
North America account 41.0% market share in 2026, supported by because the U.S. combines a large screened patient pool, a high chronic eye-disease burden, and strong reimbursement-backed procedure access. For instance, February 10, 2026, according to the data published by the World Health Organization, at least 2.2 billion people globally have near or distance vision impairment, and in at least 1 billion cases the impairment was preventable or remains unaddressed. This supports sustained demand for ophthalmic diagnostics and surgery worldwide, including high-device-adoption regions like North America. Furthermore, in January 2026, according to the data published by the National Eye Institute, as many as half of people with glaucoma do not know they have it. This supports regular screening demand for tonometers, ophthalmoscopes, OCT, and other diagnostic systems in North America. Moreover, in 2026, according to the data published by the Medicare, it has been stated that Part B covers cataract surgery, and its 2026 procedure price lookup says national averages are based on 2026 payments and copayments. This is a strong 2026 access-and-reimbursement instance supporting ophthalmic surgical device use in the U.S.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to very high unmet eye-care need with rapid healthcare expansion, aging, myopia growth, and stronger government eye-health programs. WHO reports that the Western Pacific Region has over 90 million people with visual impairment, including 10 million who are blind. WHO also notes that nearly 30% of the world’s 2.2 billion people with vision impairment live in the South-East Asia Region. This gives Asia-Pacific a much larger base for demand in diagnostics, vision correction, retinal screening, cataract devices, and surgical consumables.
Furthermore, population aging is a major reason Asia-Pacific is growing faster than more mature regions. Japan’s official statistics show that the population aged 65 years and over reached 36.243 million, or 29.3% of the total population, as of October 1, 2024, with the figure released officially in 2026. Older populations drive higher need for cataract surgery, glaucoma monitoring, OCT, fundus imaging, and vitreoretinal interventions.
Moreover, new development: stronger regional innovation and commercial activity in 2026. A notable recent development is the sharpening focus on myopia management and professional adoption in Asia-Pacific. EssilorLuxottica stated that APAO 2026 convened more than 11,000 eye care professionals from 111 countries, and used the event to highlight myopia-management pathways. The company also reported that Asia-Pacific revenue rose 9.0% at constant exchange rates in H1 2025, indicating strong recent commercial momentum in the region. This suggests Asia-Pacific is not only a high-need market, but also an active innovation and adoption hub.
The U.S. ophthalmology diagnostics and surgical devices market dominates North America given its driven by a large treatable patient pool, rapid device regulation, and reimbursement-backed procedure volumes. Census QuickFacts lists people aged 65+ at 18.0% of the U.S. population, while CDC’s 2026 diabetes update reports 40.1 million Americans with diabetes (12.0%), expanding risk for diabetic retinopathy, cataract, and glaucoma. CMS states cataract surgery with intraocular lens implantation remains a high-volume Medicare procedure, and Medicare continues annual glaucoma screening coverage for high-risk groups, including people with diabetes, African Americans aged 50+, and Hispanics aged 65+. On the regulatory side, U.S.FDA’s Division of Ophthalmic Devices continues active review pathways, with 2026 clearances such as Pentacam Cornea OCT and a Bausch + Lomb ophthalmic device, accelerating U.S. technology adoption.
India is one of the biggest players in the Asia Pacific ophthalmology diagnostics and surgical devices market in 2026. Increasing government initiatives are expanding screening and procedure volumes, which has driven the market growth significantly in the country. India is a strong example of policy-led growth in Asia-Pacific. Under the National Programme for Control of Blindness and Visual Impairment, the government reported that against a FY 2022–23 target of 7.5 million cataract surgeries, 8.344 million surgeries were performed. In January 2026, the Government of India state the programme continues to focus on cataract, refractive errors, glaucoma, diabetic retinopathy, childhood blindness, and corneal blindness, with a goal to reduce blindness prevalence to 0.20% by 2030. These initiatives support rising procurement of diagnostic and surgical ophthalmology equipment.
Additionally, on 23 March 2026, Press Information Bureau reported that 396 eye banks were functional across India, and cornea collection rose from 62,370 in 2022–23 to 69,848 in 2024–25. That points to strengthening transplant-linked ophthalmic infrastructure and equipment procurement. Wider public-health infrastructure improves access to diagnosis and referral drives the growth of the market. India also reported 5,457 urban primary health centres upgraded as Ayushman Arogya Mandirs, 8,636 urban sub-health centres operationalised, and 1,373 polyclinics functional as of late 2025. This expands the care network that can feed patients into ophthalmology diagnostics and surgery.
Source: Press Information Bureau; Government of India; PIB
Some of the major key players in Cognitive Systems Spending Marker are Abbott Medical Optics, Inc., Alcon Laboratories, Inc., Bausch & Lomb, Inc. (Valeant Pharmaceuticals), Carl Zeiss Meditec AG, CooperVision, Essilor International, Hoya Corporation, and Johnson & Johnson.
Source: Johnson & Johnson
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 63,273.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.8% | 2033 Value Projection: | USD 98,950.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Medical Optics, Inc., Alcon Laboratories, Inc., Bausch & Lomb, Inc. (Valeant Pharmaceuticals), Carl Zeiss Meditec AG, CooperVision, Essilor International, Hoya Corporation, and Johnson & Johnson. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Primary Research Interviews
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients