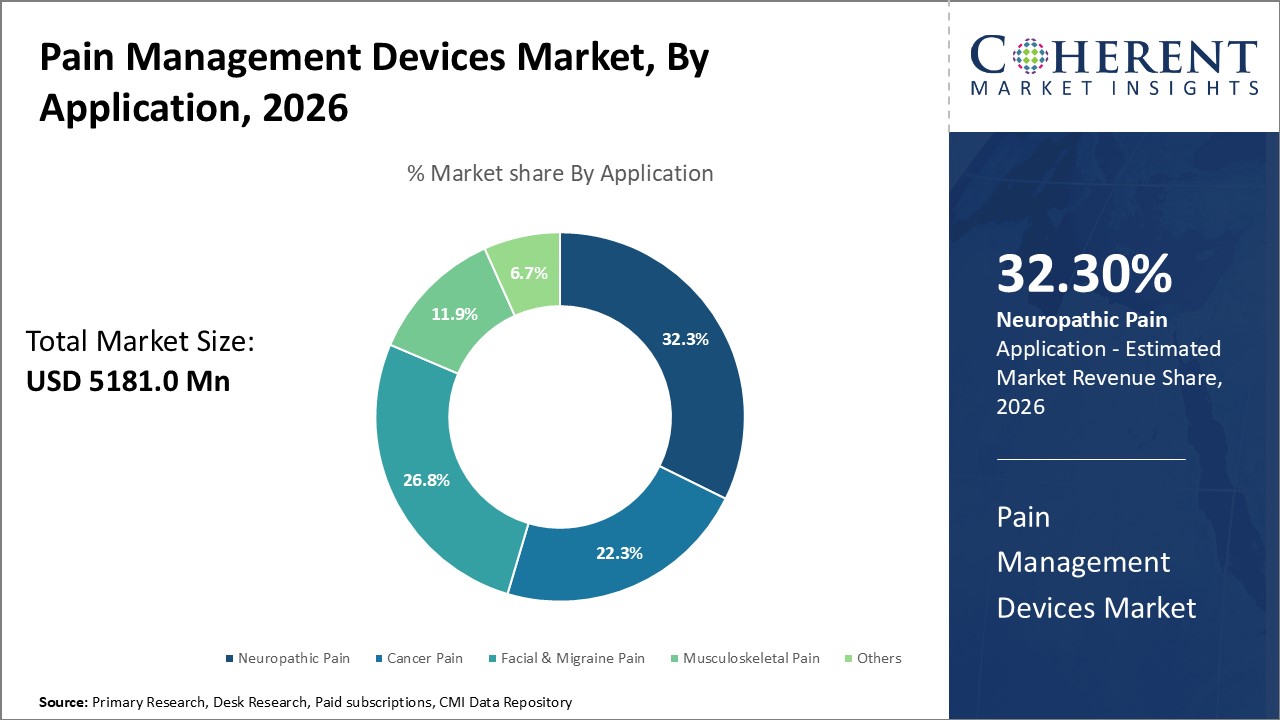
Pain Management Devices Market is estimated to be valued at USD 5,181.0 Mn in 2026 and is expected to reach USD 8,157.7 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.7% from 2026 to 2033.
Technology advancement in the pain device management is the key driver of progress and innovation in modern society, and also trigger the approval of pain device management. These advancements have revolutionized the way pain is managed, providing more effective and personalized treatment options. Additionally, the approval of pain device management has opened up new avenues for patients to regain control over their pain and improve their quality of life.
For instance, on April 18, 2023, Neuspera Medical, a medical device company, announced that the company has received U.S. Food and Drug Administration (FDA) clearance for the next-generation Neuspera ultra-miniaturized system. The system is comprised of a micro-implant that delivers neurostimulation therapy through a wireless platform including a wearable transmitter and iPad-based clinician programmer.
|
Event |
Description and Impact |
|
Technological Advancement in Neuromodulation |
|
|
Aging Demographics and Chronic Disease Prevalence |
|
|
Geopolitical Tensions and Trade Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The Pain Management Devices Market operates within a diverse and evolving reimbursement landscape that significantly impacts market accessibility and adoption. In the United States, reimbursement is primarily governed by CPT and HCPCS codes—including codes for spinal cord stimulator implantation (63650-63688), neurostimulator electrodes (64553-64595), and TENS units (E0720-E0749). Medicare typically covers about 80% of approved pain management procedures, with spinal cord stimulator implantations reimbursed between $15,000 and $25,000. Medicaid coverage varies widely by state, often requiring prior authorization.
In Europe, reimbursement policies depend on country-specific health technology assessments (HTAs) aligned with the Medical Device Regulation (MDR) 2017/745. Countries like Germany and France use Diagnosis Related Group (DRG) systems to determine coverage levels for neuromodulation and infusion devices.
Emerging markets in the Asia-Pacific region are seeing gradual improvements in reimbursement frameworks, with Japan’s PMDA facilitating accelerated approvals and national insurance schemes expanding coverage for pain devices.
Overall, reimbursement complexities—ranging from coding variability to coverage restrictions—pose challenges, especially for innovative devices and in emerging economies. Enhanced payer engagement and robust clinical evidence will be crucial for improving reimbursement and accelerating patient access worldwide.
The Neuromodulation & Neurostimulation Devices sub-segment is projected to dominate the global pain management device market in 2026, accounting for 48% of the total market share by value. This leadership is attributed to the introduction of advanced products and the increasing prevalence of chronic pain conditions worldwide.
These devices, which deliver targeted electrical stimulation to modulate nerve activity, are becoming a preferred choice among healthcare providers due to their minimally invasive nature and proven long-term efficacy in pain reduction.
Following Neuromodulation & Neurostimulation Devices, the Analgesic Infusion Pumps and Ablation Devices sub-segments are anticipated to hold the remaining market share in 2025. While these devices continue to play a crucial role in specific pain management applications, their growth is expected to be comparatively moderate. Nevertheless, ongoing innovation and broader clinical adoption of ablation techniques for chronic pain are likely to support steady expansion of these categories over the forecast period.
To learn more about this report, Request Free Sample
The Neuropathic Pain sub-segment is expected to lead the market throughout the forecast period, driven by the increasing prevalence of neuropathic conditions, which affect approximately 3.4% of the general population. The growing demand for effective solutions to manage nerve-related pain disorders has propelled the adoption of advanced pain management devices, particularly neuromodulation-based technologies.
Other key application areas include Cancer Pain, Facial & Migraine Pain, Musculoskeletal Pain, and Others. These segments collectively represent a significant portion of the market, as pain management devices are increasingly being integrated into treatment protocols to improve quality of life and reduce dependency on pharmacological interventions.
The Hospital sub-segment is anticipated to dominate the market over the forecast period, as hospitals continue to adopt advanced pain management technologies to enhance patient outcomes. Their ability to integrate comprehensive treatment approaches, combined with rising investments in technology-driven care solutions, positions hospitals as the primary users of pain management devices.
The Clinics and Others segments also play a critical role in device adoption, particularly as outpatient care and home-based treatments gain popularity. These end users are expected to witness gradual market share growth, supported by the increasing availability of portable and user-friendly devices.
To learn more about this report, Request Free Sample
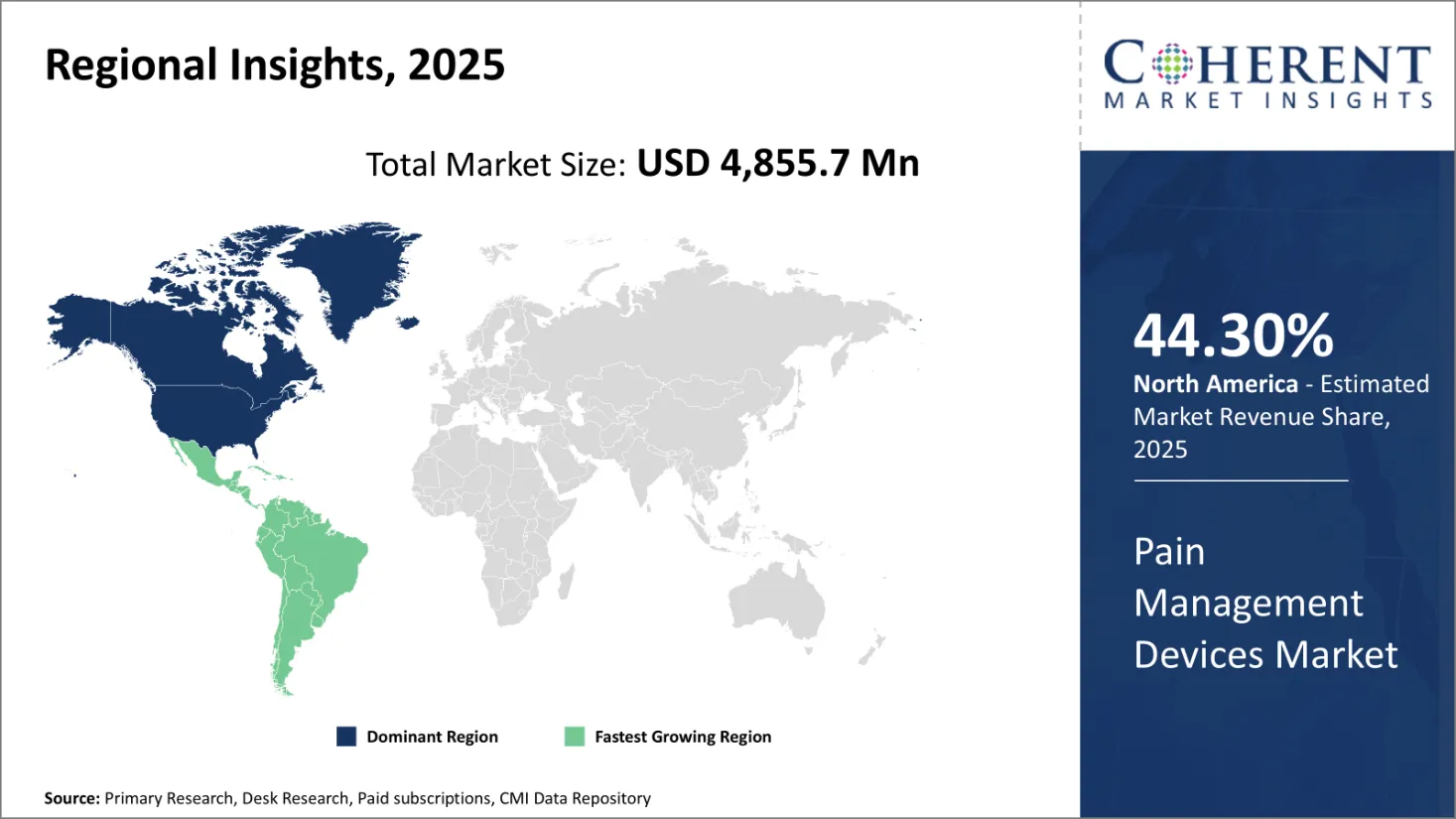
North America is the largest region in the global pain management devices market, holding 44.30% share of the global market in 2026. The United States remains the primary contributor, driven by the high prevalence of chronic pain, advanced healthcare infrastructure, and significant presence of major industry players. According to the U.S. Centers for Disease Control and Prevention (CDC), an estimated 20.9% of U.S. adults experienced chronic pain in 2021, with 6.9% reporting high-impact chronic pain—affecting 51.6 million and 17.1 million individuals, respectively.
Growing adoption of neuromodulation and neurostimulation technologies, robust investments in healthcare, and favorable reimbursement policies continue to fuel market growth. Additionally, populations such as non-Hispanic American Indian and Alaska Native adults, who experience a higher prevalence of chronic pain, are further driving the demand for advanced pain management devices in the region.
Europe maintains a strong position in the global pain management devices market, supported by its advanced healthcare systems, increasing aging population, and continuous adoption of innovative technologies. Countries like Germany, the United Kingdom, and France lead in market contribution due to high awareness of pain management therapies and robust public healthcare funding.
Stringent regulations on opioid use are also driving greater reliance on device-based pain therapies, particularly neuromodulation and infusion pumps. The region’s commitment to improving quality of life for chronic pain patients, coupled with the presence of established medical device manufacturers, ensures steady market growth through the forecast period.
Asia Pacific (APAC) is emerging as one of the fastest-growing regions in the pain management devices market, fueled by a large patient base, rising healthcare investments, and growing awareness about non-invasive pain therapies. China, India, Japan, and South Korea are key contributors, with expanding healthcare infrastructures and a rising prevalence of chronic diseases driving market adoption.
Favorable government initiatives to improve access to advanced healthcare technologies, along with increasing private sector investments, are positioning APAC as a critical growth engine. As more healthcare professionals and patients seek alternatives to traditional pain medications, device-based pain management solutions are expected to gain significant traction in this region.
The U.S. leads the North American market, supported by a high burden of chronic pain, rapid adoption of advanced medical technologies, and strong presence of market leaders. A supportive reimbursement landscape and ongoing R&D activities further bolster growth in this market.
China dominates the Asia Pacific market, driven by a large patient population, increasing healthcare expenditure, and government initiatives to expand access to advanced medical devices. Domestic manufacturers are also investing in cost-effective solutions to meet rising local demand.
India is emerging as a key growth market in APAC, supported by its rapidly growing healthcare infrastructure, increasing awareness about chronic pain management, and government programs like “Make in India” encouraging domestic production of medical devices.
Germany leads the European market due to its advanced healthcare system, high diagnosis rates for chronic pain conditions, and strong support for innovative medical technologies. Its focus on reducing opioid dependence continues to drive demand for device-based therapies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,181.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.7% | 2033 Value Projection: | USD 8,157.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Stryker Corporation, Pfizer Inc. (Hospira Inc.), Medtronic Plc, Boston Scientific Corporation, Baxter International Inc., Braun Melsungen AG, Abbott Laboratories, Colfax Corporation (Djo Global LLC), Kimberly-Clark Corporation (Baylis Medical), Johnson & Johnson (Codman and Shurtleff, Inc.), Neuspera Medical, ZetrOZ Systems, Therabody, BioElectronics Corporation, Nevro Corp, Stevanato Group S.p.A., Bexson Biomedical, Inc., SPR Therapeutics, Lumaflex, Saluda Medical Pty Limited, DyAnsys Inc., Pacira BioSciences, Inc., InterSystems, Nalu Medical, Inc., and StimaWELL North America Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence rate of chronic pain is the main factor for the market expansion. For instance, according to the Pain and Therapy Journal 2021, stated that musculoskeletal pain is a challenging condition for both patients and physicians.
Many adults have experienced one or more episodes of musculoskeletal pain at some time of their lives, regardless of age, gender, or economic status. It affects approximately 47% of the general population. Of those, about 39–45% have long-lasting problems that require medical consultation. Inadequately managed musculoskeletal pain can adversely affect quality of life and impose significant socioeconomic problems.
The integration of Artificial Intelligence can boost the market growth and also provides the opportunity to the researchers and the patient. Researchers can utilize AI to analyze large amounts of data and identify patterns or trends that may have otherwise been missed. Additionally, patients can benefit from AI-powered technologies that enable personalized treatment plans and improve healthcare outcomes.
For instance, in March 2023, Nevro Corp., a global medical device company that is delivering comprehensive, life-changing solutions for the treatment of chronic pain, announced that the company has now initiated the U.S. full market launch of its HFX iQ spinal cord stimulation (SCS) system, following the completion of a successful limited market release.
One of the main issues impeding market expansion is the strict regulatory environment for pain management devices. Due to the significant expenses associated with the development and approval processes, several makers of pain management devices are delaying the release of its products on the market. A significant barrier to the widespread use of pain management devices among patients and healthcare professionals in many nations is its expensive cost.
To overcome the challenge, certain awareness programs should be done to aware people about such devices, and also to treat by using pain management devices.
The effectiveness of pain device management is the key to market expansion. The pain device management market relies heavily on the effectiveness of pain management devices in providing relief to patients. As more individuals seek alternative pain management solutions, the market has significant potential for growth.
For instance, on January 26, 2023, ZetrOZ Systems, a leading healing innovations in sports medicine, developing wearable bioelectronic devices for the delivery of sustained acoustic medicine (sam), demonstrated the effectiveness of its samx1 and sam2.0 sustained acoustic medicine devices for soft tissue injury therapies. The samx1 is the first U.S. FDA-cleared pain management device of its kind cleared for every day, at-home use.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients