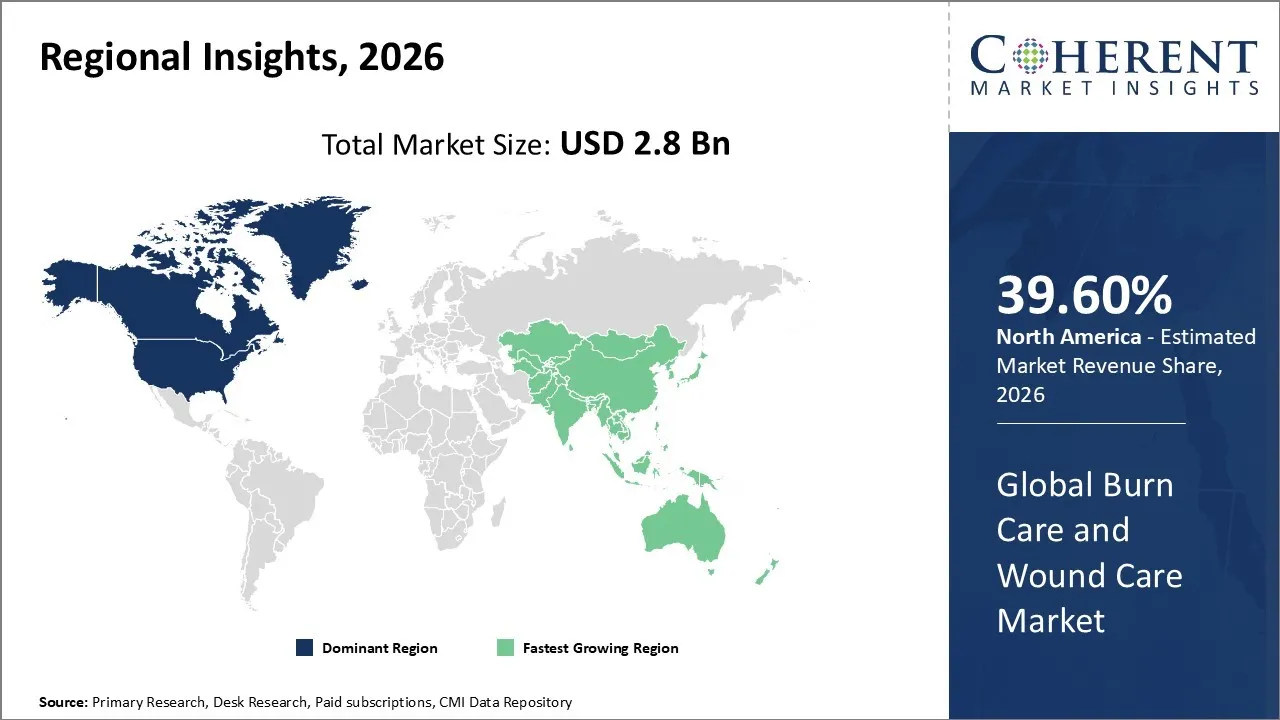
The Burn Care and Wound Care Market is estimated to be valued at USD 2.8 Bn in 2026 and is expected to reach USD 4.5 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.1% from 2026 to 2033.
The increasing incidence of burn injuries, chronic wounds, and an aging population drive the Burn Care and Wound Care Market. Technological advancements in wound care products, such as advanced dressings, skin substitutes, and antimicrobial agents, are transforming treatment options. A growing emphasis on infection prevention, patient education, and specialized care in hospitals, clinics, and home settings further boosts market growth. Additionally, rising awareness of chronic wound management and cosmetic concerns fuels the demand for innovative wound care solutions.
|
Current Events |
Description and its impact |
|
Geopolitical and Conflict-Driven Events |
|
|
Economic and Healthcare Policy Trends |
|
|
Regulatory and Standards Developments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospitals and Clinics acquired the prominent market share of 43.3% in 2026. Hospitals and clinics drive the growth of the Burn Care and Wound Care Market as the number of burn injuries and chronic wound cases rises. The demand for specialized care, including advanced wound healing technologies, has led these institutions to adopt cutting-edge treatments. Improved healthcare infrastructure, better access to care, and a focus on infection prevention are also boosting wound care services. Furthermore, rising patient awareness and the growing need for timely, effective treatments are contributing to the market's expansion. For instance, in December 2025, Holland Hospital expanded its wound healing and hyperbaric medicine program, providing West Michigan patients with access to advanced treatments for chronic and complex wounds.
Topical Agents hold the largest market share of 35.7% in 2026. The demand for topical agents in the Burn Care and Wound Care Market is rising as they promote healing and prevent infection. Innovations in formulations, including antimicrobial, anti-inflammatory, and growth factor-based agents, are enhancing treatment outcomes for both acute burns and chronic wounds. Their convenience, effectiveness, and non-invasive nature make topical treatments ideal for patient care. Additionally, increasing awareness of scar prevention, infection control, and self-care is driving the growing preference for topical wound management solutions. For instance, in April 2025, SKNV, a leader in dermatologic innovation, has launched Kefunova Cream (Fluorouracil 5% / Calcipotriene 0.005%), a prescription-only topical therapy designed to meet dermatologists’ needs in treating actinic keratosis (AK) and superficial basal cell carcinoma (sBCC).
Wound Type expected to hold largest market share of 41.1% in 2026. The growing prevalence of conditions like burns, diabetic ulcers, pressure ulcers, and surgical wounds is driving the wound type segment in the Burn Care and Wound Care Market. Each wound type demands specific treatments, which increases the need for specialized care solutions. The rise in chronic diseases, aging populations, and traumatic injuries further fuels the demand for targeted wound care. Additionally, advanced wound care technologies and increasing healthcare awareness are enhancing the management and outcomes of various wound types. For instance, in November 2025, the Department of Atomic Energy (DAE) unveiled two key scientific breakthroughs in healthcare and rare-earth research. In partnership with Cologenesis Pvt. Ltd., DAE introduced ColoNoX, India’s first nitric-oxide-releasing wound dressing for diabetic foot ulcers (DFU), developed by Bhabha Atomic Research Centre (BARC) and approved by DCGI after Phase II and III trials.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 39.60% in 2026. Technological advancements in treatments like bioactive dressings, skin substitutes, and negative pressure wound therapy are driving significant growth in the North America Burn Care and Wound Care Market. Healthcare providers are adopting more specialized care due to the rising prevalence of chronic wounds, burns, and traumatic injuries. Growing awareness of scar prevention and infection control, along with improved healthcare infrastructure and insurance coverage, is fueling demand for advanced wound care treatments in hospitals, clinics, and home care settings. For instance, in August 2025, Imbed Biosciences Inc., a leader in advanced wound care technology, launched SAM™ PainGuard™ with Lidocaine, a new addition to its acute care portfolio of fully synthetic, antimicrobial, and biocompatible wound matrices.
The Asia Pacific Burn Care and Wound Care Market is growing rapidly as healthcare access improves and the incidence of burns, chronic wounds, trauma, and an aging population increases. Healthcare providers are adopting advances in wound care technology, such as hydrocolloid dressings, biological skin substitutes, and antimicrobial agents. Growing awareness of wound care management, better patient education, and government efforts to improve healthcare infrastructure are all driving the region's rising demand for advanced burn and wound care treatments. For instance, in June 2025, Cipla Health launched its campaign ‘Cipladine – Aapka Fast Aid Expert’, featuring actress Neena Gupta. The campaign positions Cipladine as India’s trusted solution for cuts, wounds, burns, and infections, providing fast and effective healing when it matters most.
The United States Burn Care and Wound Care Market is growing rapidly as burn injuries, chronic wounds, and trauma-related cases rise. Healthcare providers are increasingly adopting advanced wound care technologies, including collagen-based dressings, negative pressure wound therapy, and biologic treatments, which are transforming patient care. A greater focus on infection prevention, scar management, and patient-centered care is driving further demand. Additionally, improved healthcare infrastructure, expanded insurance coverage, and heightened awareness of wound care are all contributing to the market's growth. For instance, in September 2024, Solventum launched the V.A.C. Peel and Place Dressing, an integrated dressing and drape that applies in under two minutes and can stay on for up to seven days. Traditional negative pressure wound therapy dressings take longer to apply, require multiple steps, and often need changing several times a week.
The India Burn Care and Wound Care Market is growing as burn injuries, chronic wounds, and traumatic accidents increase. Healthcare providers are adopting advanced wound care solutions like antimicrobial dressings, hydrocolloid bandages, and bioengineered skin grafts, which are enhancing patient outcomes. Improved healthcare access, particularly in rural areas, is boosting the demand for more effective treatments. Government efforts to strengthen healthcare infrastructure, coupled with rising awareness of proper wound care practices, are further driving the market’s growth across the country.
The Burn Care and Wound Care Market is seeing a shift towards advanced wound care products such as bioactive dressings, hydrocolloids, and negative pressure wound therapy (NPWT). These technologies are revolutionizing wound healing by promoting faster recovery and reducing the risk of infections. Their increasing adoption across hospitals, clinics, and home care settings reflects a broader trend of prioritizing effective, non-invasive treatment methods that provide better outcomes for burn and chronic wound patients.
Personalization in wound care is gaining momentum as healthcare providers recognize the importance of tailored treatments. With advancements in bioengineering and genetic research, clinicians are increasingly offering solutions specific to a patient’s wound type, underlying conditions, and overall health. This trend is helping improve outcomes for complex burns and chronic wounds, particularly in diabetic and geriatric patients, who require more specialized and effective wound management protocols for better healing and reduced complications.
The expansion of healthcare infrastructure in emerging markets and rural areas presents a vast opportunity for the Burn Care and Wound Care Market. As these regions experience better access to healthcare facilities and wound care treatments, the demand for affordable, high-quality solutions is increasing. Companies can capitalize on this growth by introducing cost-effective, innovative products tailored to local needs, including basic wound care kits and antimicrobial dressings, to cater to underserved populations.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.1% | 2033 Value Projection: | USD 4.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Smith & Nephew Plc, Johnson & Johnson, ConvaTec Inc., Medtronic Plc, Medline Industries, Inc., 3M, Baxter International Inc., Cardinal Health, Mölnlycke Health Care AB, Integra LifeSciences, B. Braun Melsungen AG, DeRoyal Industries, Inc., Coloplast A/S, Advancis Medical, Essity Aktiebolag (publ), Schweitzer-Mauduit International, Inc., Medtrade Products Limited, Advanced Medical Solutions Group plc., Beiersdorf, and L&R Medical UK Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients