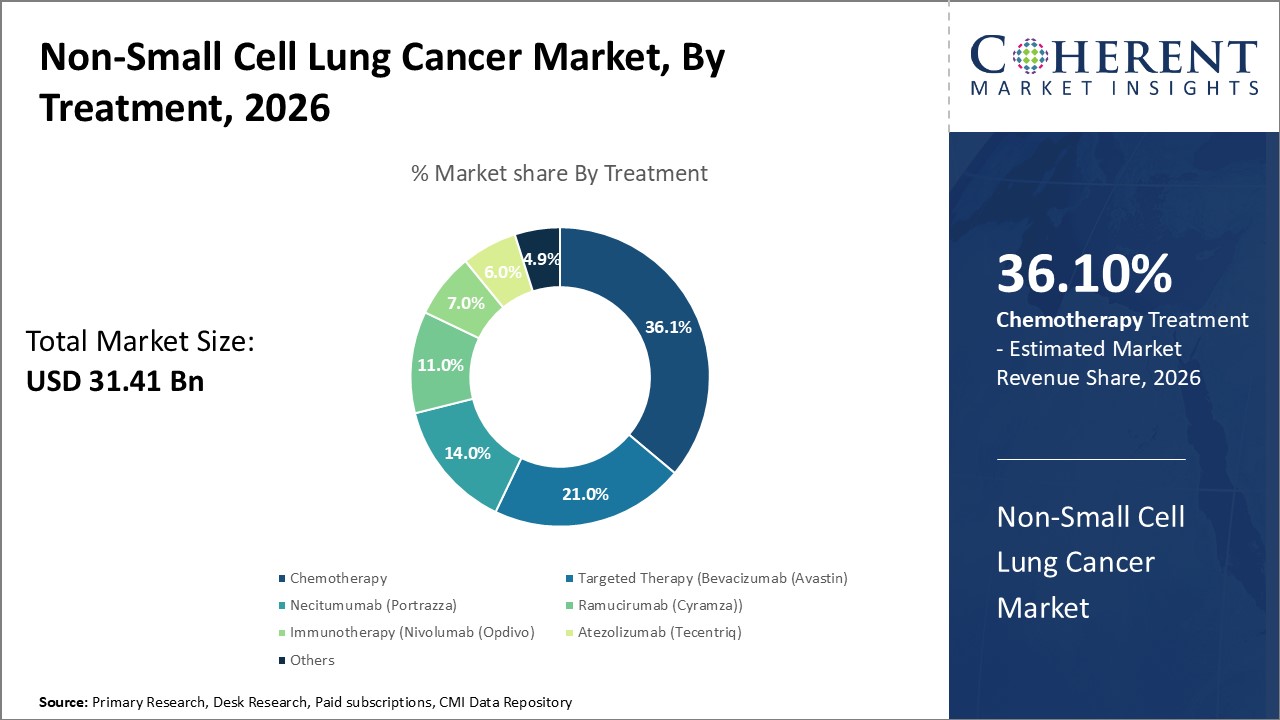
Global non-small cell lung cancer market is estimated to be valued at USD 31.41 Bn in 2026 and is expected to reach USD 60.44 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.8% from 2026 to 2033.
To learn more about this report, Request Free Sample
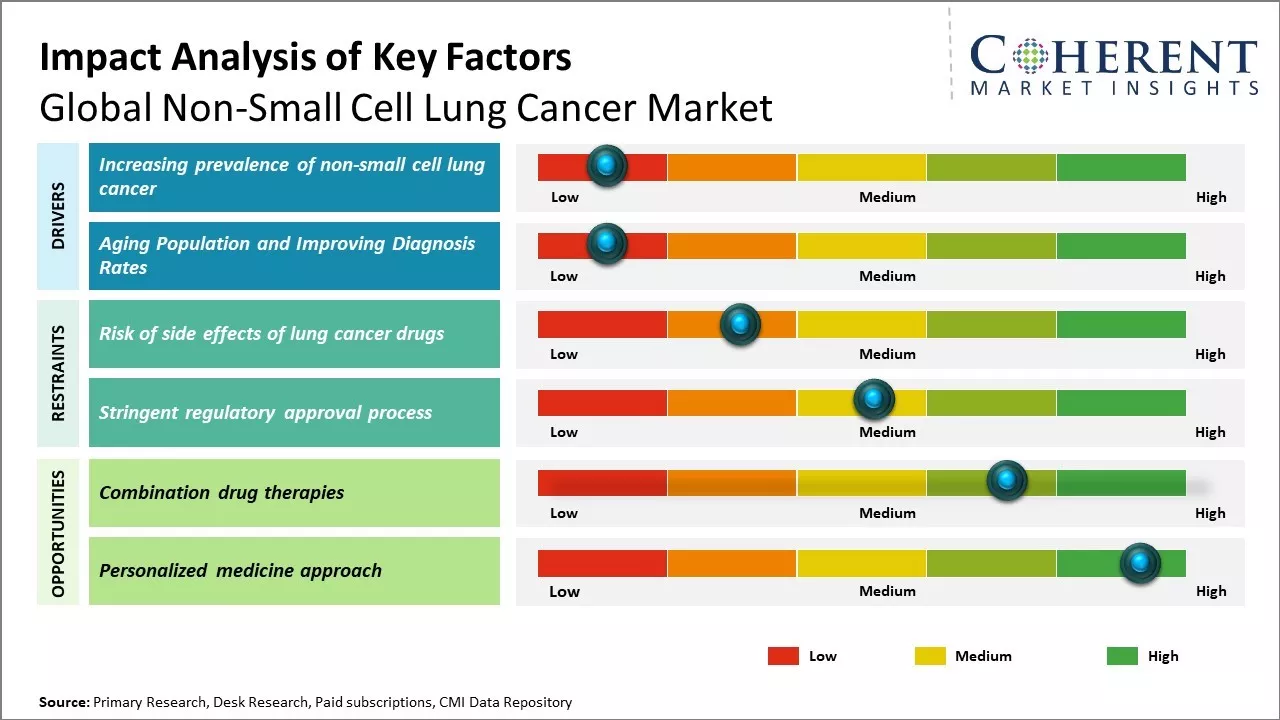
Increasing incidence of lung cancer worldwide due to growing tobacco consumption, rising pollution levels and aging population can drive the non-small cell lung cancer market growth. The market growth is driven by the increasing research and development activities for novel therapeutics, surging combination therapy and immunotherapy, and growing awareness about early diagnosis and treatment. Moreover, ongoing clinical trials evaluating new drug targets, innovative drug delivery methods and personalized medicine can offer opportunities for expansion. These factors collectively contribute to the rising non-small cell lung cancer market demand across global healthcare systems over the forecast period.
|
Current Event |
Description and its Impact |
|
Regulatory Approvals and Drug Launches |
|
|
Clinical Research and Trial Milestones |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Reimbursement policies for non-small cell lung cancer (NSCLC) treatments differ across healthcare systems but play a critical role in determining patient access to therapies. In the United States, Medicare and private insurers generally cover standard treatments such as chemotherapy, immunotherapies like pembrolizumab, and targeted therapies. However, high-cost oral drugs, including tyrosine kinase inhibitors (TKIs), often fall under Medicare Part D, where patients may face significant out-of-pocket expenses until they reach catastrophic coverage limits. In Canada, the process is more centralized and structured: after Health Canada approval, drugs undergo health technology assessments by CADTH, followed by pricing negotiations and provincial-level decisions. Meanwhile, countries like the UK and Israel tend to offer faster reimbursement for NSCLC drugs compared to regions like Australia, reflecting broader differences in health policy priorities and budgetary frameworks.
In terms of type, adenocarcinoma segment is estimated to contribute the highest market share of 41.6% in 2026. Adenocarcinoma is the most common type of non-small cell lung cancer (NSCLC), accounting for approximately 40% of all lung cancer cases. It develops in the glandular cells that line the alveoli (air sacs) and small air passageways in the lungs. Rising incidence rate of adenocarcinoma can be attributed to changing smoking patterns, as this cancer type is more commonly associated with cigarette smoking than other types. The availability of targeted therapies for adenocarcinoma can drive the segment growth. Multiple targeted drugs have been developed that have shown clinical benefit for patients with specific genetic mutations that commonly occur in adenocarcinoma, such as EGFR, ALK, ROS1, and BRAF inhibitors. This has provided patients and physicians effective non-chemotherapy options. Ongoing research in immunotherapy and combination therapies for adenocarcinoma has potential to further expand treatment capabilities.
In December 2024, Merus announced U.S. Food and Drug Administration approved zenocutuzumab‑zbco (Bizengri) for adults with advanced, unresectable or metastatic non–small cell lung cancer (NSCLC) and pancreatic adenocarcinoma harboring the NRG1 gene fusion, following disease progression on prior systemic therapy.
To learn more about this report, Request Free Sample
In terms of treatment, chemotherapy segment is estimated to contribute the highest market share of 36.12% in 2026 due to its widespread usage. Chemotherapy is not necessarily needed by all non-small cell lung cancer patients. However, it depends on the cancer’s stage and other factors. Chemotherapy treatment travels through the bloodstream and reaches the majority of the body parts. Combinations of 2 chemotherapy drugs are often used to treat early-stage lung cancer. Commonly used chemo drugs for non-small cell lung cancer are platinum-based chemo drugs, including carboplatin and cisplatin. Chemotherapy is usually given for one to three days, for several hours each time, every one to three weeks, which gives the body time to rest and recover.
In June 2025, At ASCO 2025, the phase 3 CheckMate 816 trial revealed that adding nivolumab (Opdivo) to platinum‑based chemotherapy before surgery significantly improves long‑term survival in resect able non‑small cell lung cancer (NSCLC).
In terms of end user, hospitals segment is estimated to contribute the highest market share of 51% in 2026. The hospitals segment is highly demanding due to its high incidence, complexity of treatment, and the need for multidisciplinary care. Non-small cell lung cancer has a high prevalence rate, accounting for about 80% of all lung cancer cases, making it one of the most common cancers treated in hospitals. Many patients present with advanced stages of the disease, requiring hospitalization for diagnosis, staging, and symptom management such as breathing difficulties, pain, or complications like pleural effusion. Hospitals provide advanced therapies for non-small cell lung cancer, such as D-L1 checkpoint inhibitors or EGFR/ALK-targeted drugs, which require hospitals to monitor side effects, administer infusions, and manage therapy-related complications. Hospitals also often participate in clinical trials, giving patients access to cutting-edge treatments.
In May 2025, the Oncology Department at Amrita Hospital, Kochi, in collaboration with the Indian Society of Medical and Pediatric Oncology, recently conducted a comprehensive two‑day workshop on lung cancer treatment. the event brought together leading oncologists, pulmonologists, and paramedical professionals from across India to explore the latest advancements in diagnosis, staging, and multimodal treatment strategies.
To learn more about this report, Request Free Sample
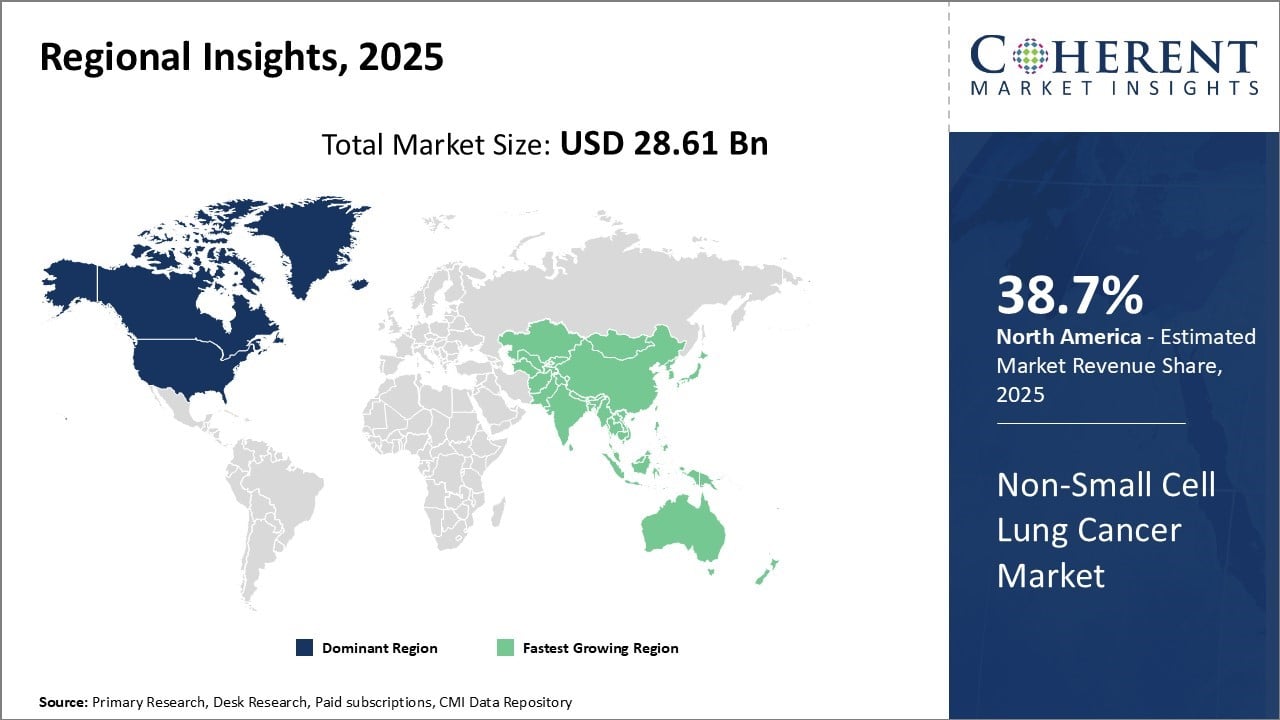
North America has established itself as the dominant region in the global non-small cell lung cancer market with an estimated market share of 38.7% in 2026. The U.S., in particular, accounts for the bulk of the market share due to strong industry presence and large patient population affected by the disease. Pharmaceutical companies have their headquarters and largest R&D centers based in the U.S., driving intense drug innovation and development activities in the region. High healthcare expenditure levels and availability of favorable reimbursement policies ensure faster adoption of newer treatments.
For example, in May 2026, the FDA granted accelerated approval to AbbVie’s Emrelis, an antibody-drug conjugate targeting c-Met–positive non-squamous NSCLC, after a Phase II trial showed a 35% response rate in progression-resistant patients. Such approvals are further supported by Medicare coverage for precise biomarker testing, reinforcing the region’s leadership in developing and accessing cutting-edge NSCLC treatments.
Asia Pacific region is emerging as the fastest growing market increasingly shaped by the region’s diverse economies like China and India have a huge patient population and rising healthcare investment capabilities. Growing awareness about early detection and treatment is resulting in improved survival rates. Local pharmaceutical industries are strengthening their non-small cell lung cancer drug portfolios by developing low-cost biosimilar and generic versions. This makes advanced therapeutics more affordable for patients. This trend underscored by recent approvals like China’s Taletrectinib for ROS1-positive NSCLC in January 2026.
Countries within Asia Pacific also differ significantly in their regulatory guidelines and healthcare infrastructure. For instance, Japan has well-established systems as comparable to the West while other Association of Southeast Asian Nations are still developing. This leads to varied growth trajectories across nations. Furthermore, Asia Pacific players are focused on treatments personalized to genetic patterns predominant in Oriental populations.
The United States non–small cell lung cancer market demand is surging due to a combination of rising incidence, advanced therapies, and strong diagnostic frameworks. Notably, recent initiatives have focused on expanding biomarker testing: Pfizer is actively promoting BRAF mutation screening for NSCLC patients to boost uptake of its Braftovi/Mektovi targeted therapy, stressing that “all patients with lung cancer should be tested for BRAF mutations, and that could be done with a simple blood test,” with insurance coverage already available for such tests.
Additionally, innovative liquid biopsies like AI-powered ctDNA screening developed at Johns Hopkins are enabling earlier detection and quicker treatment starts. A study revealed that ctDNA clearance by week 10 strongly predicted better progression-free and overall survival, reinforcing the clinical value of blood-based monitoring for TKIs. Medicare has been supportive, covering tests like Guardant360 since 2018, ensuring broader access to vital diagnostic tools. These developments highlight why the U.S. is experiencing high demand: advanced therapies and immunotherapies are driving widespread adoption of biomarker and liquid biopsy testing, supported by payer coverage and ongoing diagnostic innovation.
China’s non–small cell lung cancer market demand is intensifying, driven by its status as the country with the highest number of lung cancer cases worldwide and a steadily increasing burden of adenocarcinoma. This trend has spurred a rapid expansion of genomic testing and locally developed targeted therapies, particularly those addressing EGFR mutations.
In April 2026, Innovent Biologics received NMPA approval for limertinib, a third-generation EGFR tyrosine kinase inhibitor, as first-line treatment for NSCLC patients with EGFR exon 19 or 21 mutations, based on a Phase 3 trial demonstrating a doubling of progression-free survival. These developments underscore China’s commitment to integrating advanced genomic diagnostics with domestically developed targeted therapies, addressing its high adenocarcinoma prevalence and rapidly improving patient outcomes.
The increasing prevalence of non-small cell lung cancer (NSCLC) cases can drive the market growth. According to the American Cancer Society, the lung cancer data includes both small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC). Typically, about 13% of all lung cancers are SCLC, and about 87% are NSCLC. The lung cancer cases in the U.S. in 2026 include about 226,650 new cases of lung cancer and about 124,730 deaths from lung cancer. The high risk of mortality associated with NSCLC, with a 5-year survival rate of around 18-20%, has led to greater emphasis on early detection and effective treatment options. Advanced diagnostic techniques, along with improved understanding of NSCLC at the molecular level, have enabled targeted therapies. Pharmaceutical companies have correspondingly ramped up research and development of novel drug formulations over the past decade. Some of the newly approved drug regimens have shown promising efficacy outcomes with fewer side effects in clinical trials. For example, immune checkpoint inhibitors that help enhance the body's immune response against cancer cells have emerged as a breakthrough. Drugs like Pembrolizumab, Nivolumab, and Atezolizumab have notably improved progression-free and overall survival rates in specific patient groups as compared to standard chemotherapy, according to studies by the American Society of Clinical Oncology.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 31.41 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 60.44 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche Ltd, Honeywell International Inc., AstraZeneca PLC, Merck & Co., Inc., Pfizer Inc., Bristol-Myers Squibb Company, Novartis AG, Eli Lilly and Company, Boehringer Ingelheim International GmbH, Celgene Corporation, Amgen Inc., Johnson & Johnson, Gilead Sciences, Inc., AbbVie Inc., Astellas Pharma Inc., Takeda Pharmaceutical Company Limited, Sanofi, Teva Pharmaceutical Industries Ltd., Bayer AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
With overall enhancements in living standards and healthcare access, populations across the world are progressively aging. While this longevity is a positive outcome of development, it also contributes to higher risks of cancer. Aging is a significant risk factor for cancers like non-small cell lung cancer. The biological changes in cells and immune system with growing age weaken the body's ability to identify and eliminate malignant growths in the lungs. A large section of the current elderly population would have been exposed to unregulated tobacco consumption in the mid-20th century before awareness rose around its risks. This leads to higher incidence of lung cancer as residues of past addiction impacts their health today.
Combination drug therapies can offer opportunity for global non-small cell lung cancer market growth. Using multiple drugs that attack cancer through different mechanisms can help overcome treatment resistance and lead to better outcomes for patients. Existing therapies for non-small cell lung cancer mostly rely on single drugs. While these have helped increase survival rates to some extent, cancer often develops resistances over time. Combination therapies make it harder for cancer cells to mutate in a way that allows them to evade every drug. By using two or more drugs simultaneously, combination therapy aims to block multiple pathways that drive cancer growth and survival. This multi-pronged approach can keep the cancer from developing resistance for longer periods. Promising early studies suggest combination therapies may significantly improve survival.
The Non-Small Cell Lung Cancer market value is undergoing a profound transformation, driven not by incremental progress but by a sharp shift in clinical and molecular stratification that is redefining treatment standards. The traditional paradigm of cytotoxic chemotherapy is being rapidly displaced by biomarker-driven therapies and immune checkpoint inhibitors—an evolution that is not only clinically justified but commercially strategic.
One of the most defining changes is the stratification of NSCLC by actionable mutations, particularly EGFR, ALK, ROS1, BRAF, and more recently, MET exon 14 skipping and RET rearrangements. In the U.S., EGFR mutations occur in about 15% of NSCLC patients, but in East Asian populations, this proportion rises to as much as 50%, representing a high-value therapeutic segment. The approval and rapid uptake of osimertinib (Tagrisso, AstraZeneca) for EGFR-mutated NSCLC, for both first-line and adjuvant use, illustrates the market’s growing receptivity to precision oncology. Tagrisso’s expansion into early-stage resected NSCLC, based on the ADAURA trial showing an 80% reduction in disease recurrence or death, reflects a demand for deeper molecular intervention at earlier disease stages.
Anti-PD-1/PD-L1 agents such as pembrolizumab and nivolumab are no longer viewed as monoliths; their effectiveness is closely tied to PD-L1 expression thresholds and tumor mutational burden (TMB). Notably, the KEYNOTE-024 trial showed that pembrolizumab monotherapy in PD-L1 ≥50% NSCLC patients led to a median overall survival of 26.3 months, outperforming chemotherapy by a wide margin. This biomarker stratification is no longer optional—it is shaping access, pricing, and formulary decisions.
A concerning bottleneck, however, is the underutilization of molecular diagnostics. Despite the clinical imperative, global testing rates for EGFR and ALK mutations remain suboptimal, especially outside of tier-1 hospitals. In several European markets, less than 60% of patients undergo comprehensive genomic profiling at diagnosis, limiting the penetration of targeted therapies and resulting in a revenue gap that does not reflect therapeutic potential.
There is also an emerging commercial opportunity in KRAS G12C-mutated NSCLC, which represents approximately 13% of adenocarcinoma cases. The launch of sotorasib (Lumakras) and adagrasib marks the first viable entry into what was once deemed an “undruggable” mutation. However, early data suggests resistance mechanisms evolve rapidly, and combination regimens will likely define second-generation commercial viability in this space.
Equally important is the evolving competitive landscape in adjuvant and neoadjuvant settings. Trials such as CheckMate 816 have reshaped expectations by demonstrating that neoadjuvant nivolumab plus chemotherapy significantly improves event-free survival and pathological complete response in resectable NSCLC. This has extended the target population beyond metastatic disease, thereby expanding commercial runway for IO agents.
Payer scrutiny, particularly in high-income markets, is intensifying. The rising costs associated with combination therapies and extended treatment durations have prompted health technology assessment (HTA) agencies to demand demonstrable real-world value. This is likely to encourage outcome-based reimbursement models, especially as treatment durations with agents like pembrolizumab now often exceed two years.
*Definition: Non-small cell lung cancer is a group of lung cancers that behave similarly, such as squamous cell carcinoma and adenocarcinoma. Non-small cell lung cancer (NSCLC) treatment options include surgery, chemotherapy, radiation therapy, targeted therapy, and immunotherapy. Non–small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancers.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients