Cancer Diagnostics Market is estimated to be valued at USD 77.95 Bn in 2026 and is expected to reach USD 149.97 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 9.8% from 2026 to 2033.
Analysts’ Views on Global Cancer Diagnostics Market:
Increasing prevalence of Cancer disease, rising geriatric population, and increasing awareness about Cancer and new biomarker releases and strategic efforts by prominent market competitors are favorably impacting the industry's growth. Different healthcare professionals primarily focus on developing effective screening and treatment solutions to evaluate the prevalence levels. Several healthcare agencies and market players are launching different programs promoting different advanced diagnostics, thus accelerating market growth.
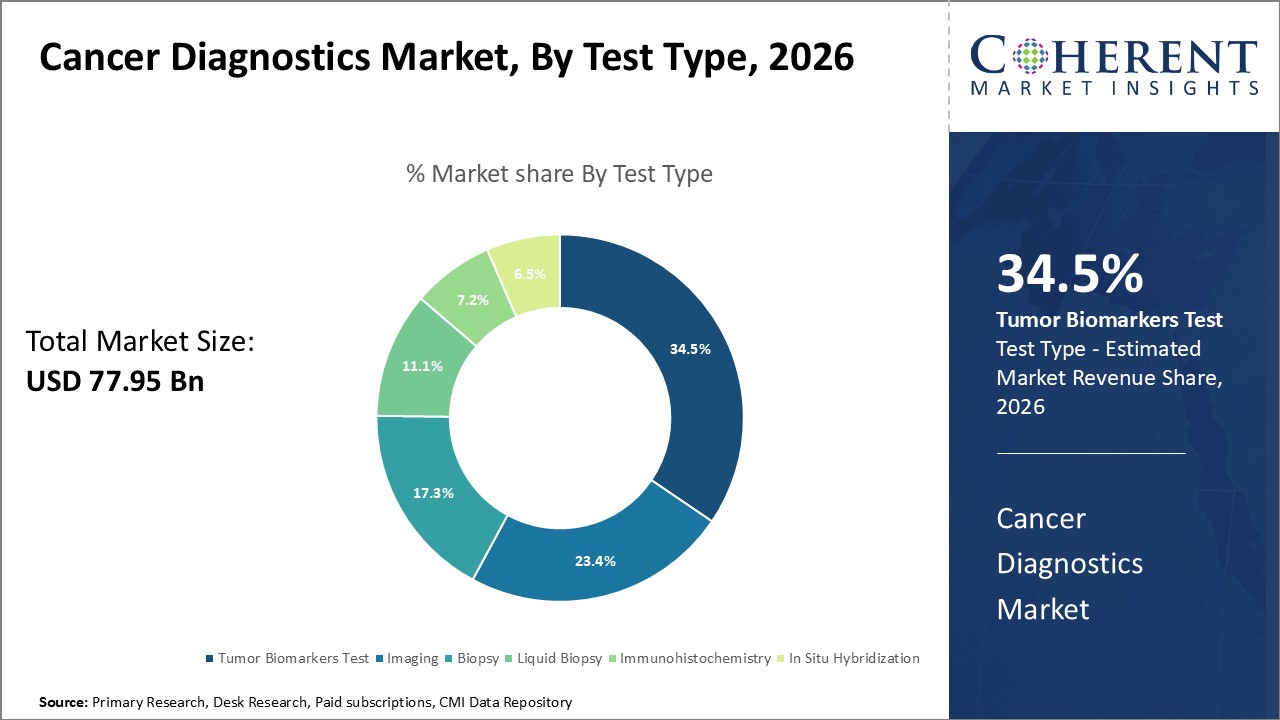
Figure 1. Global Cancer Diagnostics Market Share (%), By Test Type, 2026
To learn more about this report, Request Free Sample
Global Cancer Diagnostics Market – Driver
Increased Developments of Diagnostic Testing Techniques
Increased developments of diagnostic testing techniques is expected to propel the growth of the global cancer diagnostics market over the forecast period. Technological advancements in diagnostic testing are expected to boost market growth during the forecast period. For instance, in November 2021, Hologic, Inc., a medical technology company primarily focused on women’s health; it sells medical devices for diagnostics, surgery, and medical imaging, announced the commercial access of the Genius Digital Diagnostics System in Europe. The next-generation cervical cancer screening system includes deep learning-based AI and innovative volumetric imaging technology to help detect cervical cancer cells and pre-cancerous lesions in women. Thus, this is expected to boost market growth.
Increasing expenditure for the treatment and management of Cancer
Increasing expenditure for the treatment and management of cancer is also expected to aid in the growth of the market. For instance, the article published in the Journal of Lancet Regional Health in October 2026, reported catastrophic expenditure and treatment attrition in patients seeking comprehensive colorectal cancer treatment in India. 226 patients included, 20 died within six months of being offered a treatment plan and four were lost to follow-up. The median total cost of colorectal cancer treatment was US$ 5340 (407,508 Indian Rupees ), to which the biggest contributor was the patient's PARP (median INR 330,277 / US$ 4328 ).
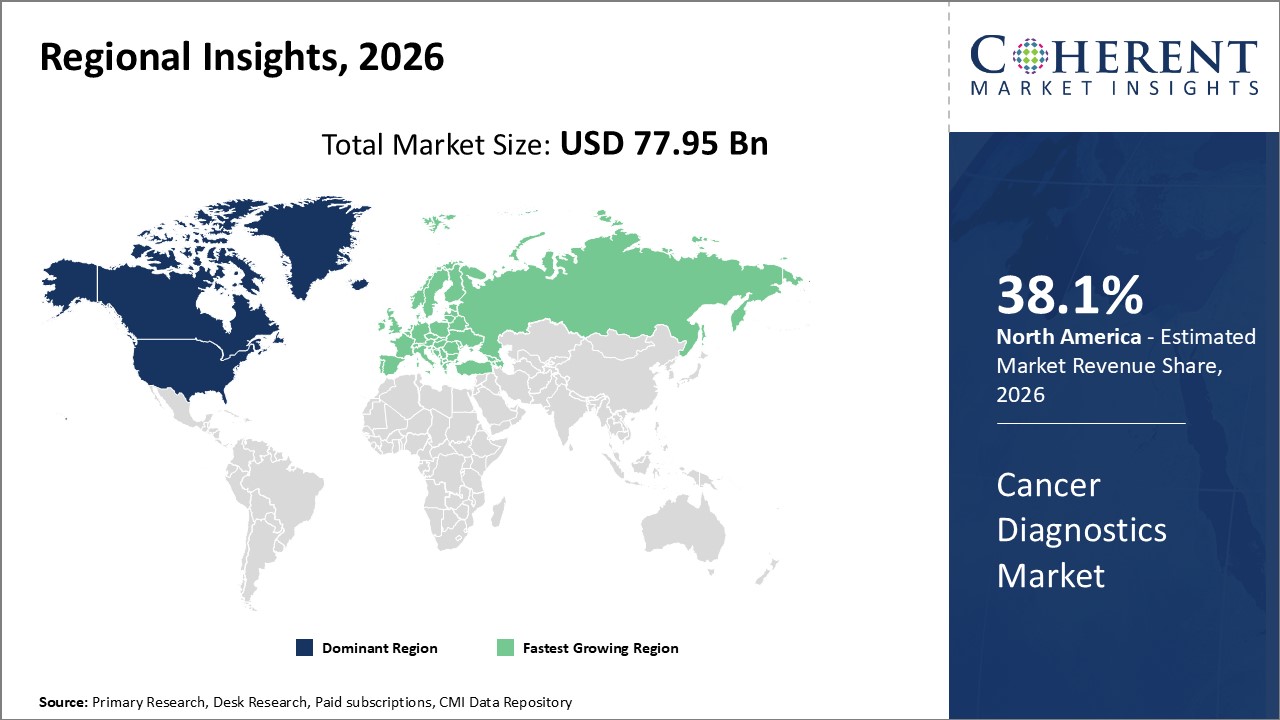
Figure 2. Global Cancer Diagnostics Market Value (US$ Billion), by Region, 2026
To learn more about this report, Request Free Sample
Global Cancer Diagnostics Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global cancer diagnostics market over the forecast period. North America is estimated to hold 38.1 % of the market share in 2026. The global cancer diagnostics market is expected to witness significant growth in the coming years, driven by the high prevalence of Cancer, favorable health reimbursement, and increased awareness of Cancer, North America to emerge as the leading region for the Cancer Diagnostics Market. For instance, according to a press release in February 2020, by imaware, which provides laboratory testing for wellness monitoring, informational, and educational use in North America, announced the launch of the At-Home Cancer Screening Test, designed by healthcare company Micro Drop. The imaware test for Cancer uses just a few drops of blood to detect unique biomarkers as well as two additional biomarkers to provide more comprehensive results. These kinds of developments in the healthcare system and rising health expenditures, as well as increased knowledge of advanced Cancer treatments, are fueling the Cancer market in North America.
Even though online consultations are advisable with patients who are having stable Cancer.
Cancer Diagnostics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 77.95 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 149.97 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Myriad Genetics, Inc., F. Hoffmann-La Roche AG, Invitae Corporation, NeoGenomics Laboratories, Inc., and BPS Bioscience, Inc. Antibodies Inc., Networks LLC, Beckman Coulter, Inc., Euro Diagnostica AB, F. Hoffmann-La Roche Ltd., Qiagen NV, Siemens Healthcare GmbH, Bio Rad Laboratories Inc., Exagen Inc. Genway Biotech, Inc., Microdrop LLC (imaware), Svar Life Science AB, Abbott Laboratories, Thermo Fisher Scientific Inc., Bio Rad Laboratories Inc., GE Healthcare, Illumina, Inc., Koninklijke Philips N.V., Toshiba Medical Systems Corporation, Agilent Technologies, Inc., Aurora Imaging Technologies, Inc., and Quest Diagnostics. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Cancer Diagnostics Market Segmentation:
The Global Cancer Diagnostics Market report is segmented into Test Type, Cancer Type, End User, and Region.
By Test Type, the market is segmented into Tumor biomarkers tests (PSA tests, CTC tests, AFP tests, CA-19 tests, HER2 tests, BRCA tests, ALK tests, CEA tests, EGFR mutation tests, KRAS Mutation Tests, Others), Imaging (ultrasound, mammography, MRI Scan, PET scan, CT scan, SPECT & Others), Biopsy (needle biopsy, endoscopic biopsy, bone marrow biopsy, others), Liquid biopsy, Immunohistochemistry, and In situ hybridization. Out of which, the Tumor biomarkers tests (PSA tests, CTC tests, AFP tests, CA-19 tests, HER2 tests, BRCA tests, ALK tests, CEA tests, EGFR mutation tests, KRAS Mutation Tests, Others) is expected to hold a dominant position in the global cancer diagnostics market during the forecast period and this is attributed to the awareness of cancer disease.
By Cancer Type, the market is segmented into Breast Cancer, Lung Cancer, Colorectal Cancer, Melanoma, Blood Cancers, Prostate Cancer, Ovarian Cancer, Stomach Cancer, Liver Cancer, Others. Out of which, Breast Cancer is expected to dominate the market over the forecast period and this is attributed to the breast cancer increases with age and peaks at menopause.
By End User, the market is segmented into Hospital associated labs, Independent diagnostic laboratories, Diagnostic imaging centers, Cancer research institutes, and others. Out of which, Hospital Segment is expected to dominate the market over the forecast period and this is attributed to the increasing expenditure of hospitals.
Among all the segmentations, the By End user associated labs segment expected to dominate the market over the forecast period, and this is attributed to the increasing obesity after menopause is which is expected to boost the growth of the cancer diagnostics market over the forecast period.
Global Cancer Diagnostics Market Cross Sectional Analysis:
Key players are making the cancer diagnostics market with advanced technology emerging economies is also expected to boost demand cancer diagnostics Market in North America region. For instance, in October 2020, Myriad Genetic Laboratories, Inc., a genetic testing and precision medicine company based in Salt Lake City, Utah, U.S., introduced Myriad's BRAC Analysis Treatment method from the Japanese Ministry of Health, Labor, and Welfare to be utilized as a partnered treatment with the cancer constraints.
Global Cancer Diagnostics Market: Key Developments
On June 1, 2023, Myriad Genetics, Inc., a company of genetic testing and precision medicine, launched new studies and expansion of its Precise Oncology Solutions portfolio at the 2023 American Society of Clinical Oncology (ASCO).
In May 2022, United Rheumatology, a rheumatology care management organization empowering rheumatologists to advance the standard of care and education, advocacy, and research organization for people living with Cancer disease, announced a partnership to develop the Cancer Wellness Center, a digital resource aimed at aiding those living with Cancer.
On March 22, 2023, F. Hoffmann-La Roche Ltd (Roche), a biotechnology company that develops drugs and diagnostics to treat major diseases, announced that it had entered into a collaboration with Eli Lilly and Company is pharmaceutical company in U.S., to support the development of Roche’s Elecsys Cancer Diagnostics Market The PARP is an innovative blood test that aims to facilitate the earlier diagnosis of Alzheimer’s disease.
In March 2022, Invitae, a medical genetics company, announced full access to its Personalized Cancer Monitoring (PCMTM) platform to help detect minimal or molecular residual disease (MRD) in patients with solid tumors. Invitae PCM uses a novel set of personalized assays based on a patient's tumor to detect circulating tumor DNA (ctDNA) in blood, offering the ability to perform risk stratification, response assessment to treatment, and detection of cancer recurrence, based on recent studies.
In January 2020, the American Cancer Society and Pfizer approved grants totaling more than USD 2.5 million in nine communities focused on reducing racial disparities and helping optimize outcomes for women facing a breast cancer diagnosis.
Global Cancer Diagnostics Market: Key Trends
Application of biomarker test for diagnosing Cancer
Application of biomarker tests for diagnosing Cancer is at an early stage of development. Major players in the market are focused on adopting partnership strategies to expand their product portfolio. Presently, there is no biochemical test available for the diagnosis of cancer. However, Rheumatoid Factor (RF) and anti–cyclic citrullinated peptide (anti–CCP) antibodies test require more refinement to improve their clinical utility. For instance, in March 2020, WorldCare Clinical, LLC, an independent contract research organization offering imaging in clinical trials, partnered with Navidea Biopharmaceuticals, Inc. is a biopharmaceutical company focused on the development of precision immunodiagnostic agents and immunotherapeutic, to offer imaging service following U.S. FDA approval of Navidea’s cancer diagnostic. Navidea’s strategy is to deliver novel products and advance the Company’s pipeline through global partnering with WorldCare Clinical, LLC.
Global Cancer Diagnostics Market: Restraint
High Cost of Diagnostic Tests
High Cost of Diagnostic Tests is expected to hamper the growth of the global Cancer diagnostics market. For instance, in May 2020, Myriad Genetics, Inc. is genetic testing and precision medicine company in U.S. its myChoice CDx became commercially available with a list price of US$ 4,590. The U.S. FDA has approved myChoice CDx for use as a companion diagnostic to identify patients with advanced ovarian cancer with positive homologous recombination deficiency (HRD) status by detecting BRCA1 and BRCA2.
Global Cancer Diagnostics Market - Key Players
Major players operating in the global Cancer Diagnostics Market include Myriad Genetics, Inc., F. Hoffmann-La Roche AG, Invitae Corporation, NeoGenomics Laboratories, Inc., and BPS Bioscience, Inc. Antibodies Inc., Networks LLC, Beckman Coulter, Inc., Euro Diagnostica AB, F. Hoffmann-La Roche Ltd., Qiagen NV, Siemens Healthcare GmbH, Bio Rad Laboratories Inc., Exagen Inc., Genway Biotech, Inc., Microdrop LLC (imaware), Svar Life Science AB, Abbott Laboratories, Thermo Fisher Scientific Inc., Bio Rad Laboratories Inc., GE Healthcare, Illumina, Inc., Koninklijke Philips N.V., Toshiba Medical Systems Corporation, Agilent Technologies, Inc., Aurora Imaging Technologies, Inc., and Quest Diagnostics.
*Definition: Cancer diagnostics is the process of identifying numerous proteins, biomarkers, and symptoms that lead to the presence of a cancerous tumor in patients. The detection of particular biomarkers and proteins that are common in cancer disorders causes the diagnosis process. Detecting cancer involves using particular techniques and devices used in its diagnosis. Imaging, laboratory tests, tumor biopsy, surgery, endoscopic examination, and genetic testing are all possible cancer diagnostic techniques.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients