North America Scar Treatment Market is estimated to be valued at USD 4,943.9 Mn in 2026 and is expected to reach USD 6,728.0 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.5% from 2026 to 2033.
Analysts’ Views on North America Scar Treatment Market:
Rising demand for scar treatment options to treat burn injuries is expected to drive market growth over the forecast period. For instance, in February 2020, a report published by the National Centre for Biotechnology Information (NCBI), stated that according to The American Burn Association (ABA) National Burn Repository, 41% of injuries in the U.S. are burn injuries.
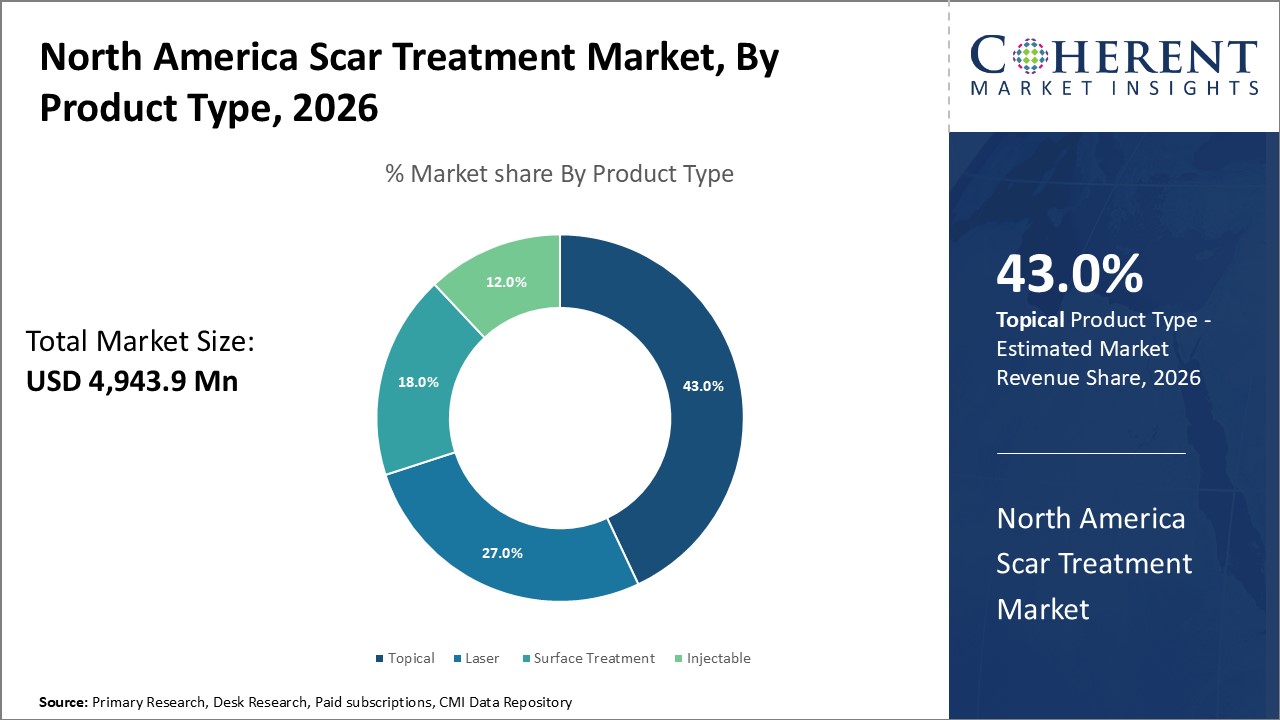
Figure 1. North America Scar Treatment Market Share (%), By Product Type, 2026
To learn more about this report, Request Free Sample
North America Scar Treatment Market– Drivers
Increasing incidence of surgeries
Scar treatment is mostly used to treat scars due to surgeries. Thus, the increasing rate of surgical operations is expected to drive the market growth over the forecast period. For instance, in March 2022, according to a report published by John Wiley & Sons, Inc., a research and education company, in a survey conducted on people living in the U.S., 35% of women and 28% of men indicated that scarring was caused due to general or orthopedic surgery.
Increasing incidence of burn injuries
Scar treatment is used for the treatment of scars caused due to burn injuries, and thus, the increasing incidence of burn injuries is expected to drive the market growth over the forecast period. For instance, in December 2022, according to an article published in Springer Nature, a scientific journal, 67% of the scars were caused due to burn injuries in the U.S.
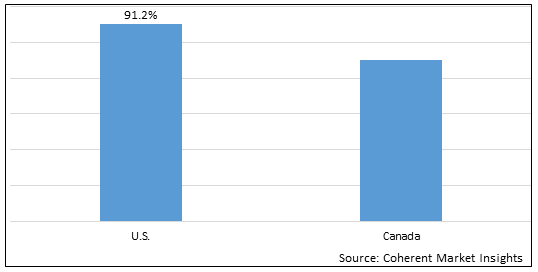
Figure 2. North America Scar Treatment Market Value (US$ Million), By Country, 2026
To learn more about this report, Request Free Sample
North America Scar Treatment Market- Country Analysis
Among countries, U.S. is estimated to hold a dominant position in the North America scar treatment market over the forecast period, due to increasing adoption of organic growth strategies such as product launches by key players in the region to expand their product portfolio. For instance, in March 2022, Cocoon Medical, a leading global developer and provider of energy-based aesthetic systems for practitioners, announced the launch of its Primelase diode laser platform in the U.S. Primelase guarantees a 4.800W maximum power, providing peak performance and highest profitability for the market.
For instance, in June 2022, a report published by National Center for Biotechnology Information (NCBI), stated that a decrease of 22.7% and 11.7% was observed in the total number of primary and secondary surgeries.
North America Scar Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,943.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.5% | 2033 Value Projection: | USD 6,728.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
ScarAway, Thrive Aesthetics MN, HRA Pharma, MARLO WHITNEY, Smith & Nephew Inc, Merz Therapeutics, Enaltus LLC, Oculus Innovative Sciences, Inc, CCA Industries, Bausch Health Companies Inc., Candela Medical, Perrigo Company plc, Sonoma Pharmaceuticals, Inc., Hologic, Inc., Johnson & Johnson Surface Treatment, Inc., Quantum Health, AVITA Medical, Revitol Corp, Scarguard labs, Lumenis Be Ltd, Cynosure, GC Aesthetics, Sientra, Inc, SkinCeuticals, Cimeosil.com, Honeydew Products, 3V Sigma USA Inc., Syneron Medical Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
North America Scar Treatment Market- Segmentation
North America scar treatment market is segmented into product type, scar type, distribution channel, and country.
Based on Product Type, the market is segmented into topical scar product (gels, creams, oils, and sheets), laser (co2 laser, excimer laser, and pulsed dye laser), surface treatment, and injectable. Out of which, the topical scar product segment is expected to dominate the North America scar treatment market during the forecast period and this is attributed to an increase in product launches by key players in the market.
Based on Scar Type, the North America scar treatment market is segmented into atrophic scars (acne, trauma, and surgery), hypertrophic scars and keloids, contractures, and stretch marks. The atrophic scars segment is expected to dominate the market over the forecast period and this is attributed to the increased incidence of surgical operations.
Based on Distribution Channel, the North America scar treatment market is segmented into hospitals, clinics, pharmacies and drug stores, and e-commerce. Out of which, the e-commerce segment is expected to dominate the market over the forecast period and this is due to the increased use of scar treatment technology for treatment of scars.
Based on Country, the market is segmented into U.S. and Canada. Out of which, the U.S. is expected to dominate the market over the forecast period, owing to increasing organic growth strategies such as product launches by key players in the country.
Among all segmentation, product type segment has the highest potential due to the increasing incidence of scars due to burn injuries. For instance, in November 2022, an article published on UpToDate, Inc., which is a point-of-care medical resource, stated that at an estimate, there are between 7 and 12 million people (up to 33,000 each day) who sustain burn injuries every year in the U.S.
North America Scar Treatment Market- Cross Sectional Analysis
Under the product type segment, topical scar product segment held a dominant position in North America region due to the increasing incidence of accidents that require surgical operations and thus result in the development of scars. For instance, on January 17, 2023, the Center for Disease Control and Prevention (CDC), stated that there were 24.8 million physician office visits for accidental injuries.
North America Scar Treatment Market- Key Developments
On June 12, 2022, Syneron Medical Ltd, a cosmetic surgery device manufacturer, acquired Candela Corporation, a laser manufacturing company, under the terms of the merger agreement, Candela shareholders will receive 0.2911 ordinary shares of Syneron for each share of Candela common stock they own. Syneron has issued approximately 6.7 million shares to acquire Candela.
On April 28, 2022, Lumenis Be, Ltd., a global leader in the field of minimally-invasive clinical solutions for the ophthalmology and aesthetic market, announced the launch of triLift, a facial stimulation device targeting three aging elements—structure, contour, and texture—by treating the three tissue layers. The device affects the muscles, the dermal layer, and the epidermal layer.
On August 17, 2021, Merz Therapeutics, a manufacturer of pharmaceutical products, came into a partnership with VENSICA, a medical device company for the needleless delivery of botox to treat scars.
On September 28, 2022, Johnson & Johnson Services, a leading healthcare company, announced an independent new consumer health company, Kenvue, which will be a standalone leading global consumer health company in 2023, subject to legal requirements including consultation with works councils and employee representatives, as required.
North America Scar Treatment Market- Key Trends
Increasing research and development activities by key market players
Increasing research and development activities for scar treatment by key market players, in order to expand their product portfolio is expected to drive the market growth over the forecast period. For instance, in April 2021, Pharmaxis Ltd, a pharmaceutical research company, collaborated with the University Of Western Australia (UWA), to start a world-first clinical trial to investigate a cream to stop scars forming after trauma, particularly following burn injuries.
North America Scar Treatment Market- Restraint
High cost of laser treatment
The major factors that can hamper the growth of the North America scar treatment market over the forecast period include the high cost of laser treatment for scar removal. For instance, on January 27, 2023, PRISTYN CARE, a health-tech company that deals in minimally invasive medical and surgical interventions, stated that the cost of acne or chicken pox-related scar removal is US$ 72.50 to US$ 241.66.
North America Scar Treatment Market- Key Players
Major players operating in the North America scar treatment market include ScarAway, Thrive Aesthetics MN, HRA Pharma, MARLO WHITNEY, Smith & Nephew Inc, Merz Therapeutics, Enaltus LLC, Oculus Innovative Sciences, Inc, CCA Industries, Bausch Health Companies Inc., Candela Medical, Perrigo Company plc, Sonoma Pharmaceuticals, Inc., Hologic, Inc., Johnson & Johnson Surface Treatment, Inc., Quantum Health, AVITA Medical, Revitol Corp, Scarguard labs, Lumenis Be Ltd, Cynosure, GC Aesthetics, Sientra, Inc, SkinCeuticals, Cimeosil.com, Honeydew Products, 3V Sigma USA Inc., Syneron Medical Ltd.
*Definition: Scar is a fibrous tissue that forms when normal tissue is destroyed by disease, injury, or surgery. For example, scar tissue forms when a wound heals after a cut, sore, burn, or other skin condition, or when an incision (cut) is made into the skin during surgery. Not all scars require treatment and many fade over time. Treatment can help if scars are bothersome or cause pain.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients