Oral Biologics Market is estimated to be valued at USD 6.52 Bn in 2026 and is expected to reach USD 54.98 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 35.6% from 2026 to 2033.
Analysts’ Views on Global Oral Biologics Market:
The rising number of patients with diabetes, hyperoxaluria, acromegaly, irritable bowel syndrome (IBS), and chronic idiopathic constipation for whom oral biologics are used for treatment. Thus, it offers the opportunity for various manufacturers to offer various oral biologics to treat these diseases.
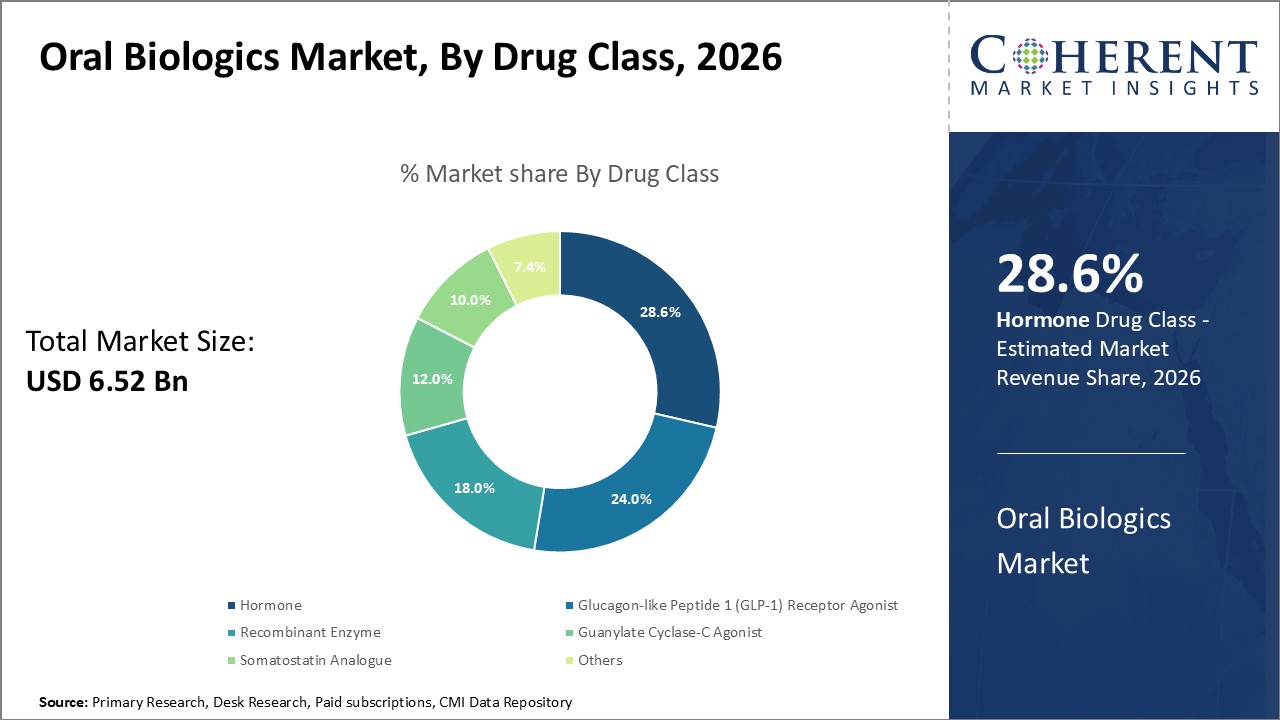
Figure 1. Global Oral Biologics Market Share (%), By Drug Type, 2026
To learn more about this report, Request Free Sample
Global Oral Biologics Market– Drivers
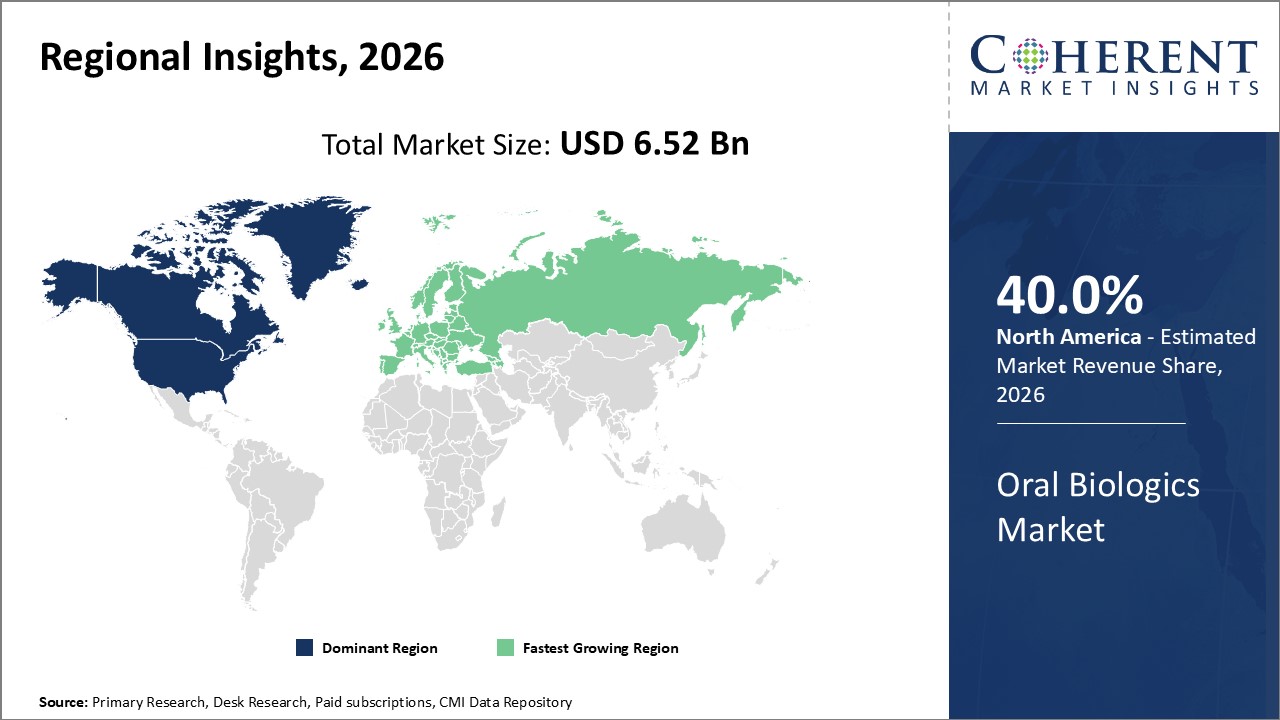
Figure 2. Global Oral Biologics Market Share(%), By Region, 2026
To learn more about this report, Request Free Sample
Global Oral Biologics Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global oral biologics market over the forecast period. North America is estimated to hold 40.0% of the market share in 2026. The global oral biologics market is expected to witness significant growth in the coming years, driven by the high prevalence of Irritable Bowel Syndrome, favorable health reimbursement, and increased awareness. The increasing prevalence of irritable bowel syndrome (IBS) is contributing to the growth of the surgical equipment market in the North America region. For instance, according to the American College of Gastroenterology, 2020, about 10% to 15% of people in the U.S. suffer from IBS symptoms annually.
This includes older individuals, immunocompromised people, patients with underlying illnesses like diabetes,hypertension, and obesity, as well as unvaccinated patients.
Oral Biologics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.52 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 35.6% | 2033 Value Projection: | USD 54.98 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novo Nordisk A/S, Biocon Limited, Oramed Pharmaceuticals, Inc., Rani Therapeutics, Entera Bio Ltd., AbbVie Inc., Enteris BioPharma, Inc., Chiasma, Inc. and Allena Pharmaceuticals, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Oral Biologics Market Segmentation:
The global oral biologics market report is segmented by drug class, by disease indication, by distribution channel, and by region.
Among all the segmentation, the disease indication segment is expected to dominate the market over the forecast period, and this is attributed to the increasing prevalence of different diseases.
Global Oral Biologics Market- Cross Sectional Analysis:
In drug class, hormone segment is expected to dominate the market in the North America region, and this is attributable to the increasing product approvals from regulatory authorities. For instance, in December 2020, Myovant Sciences, a biopharmaceutical company, announced that the U.S. FDA approved Orgovyx (relugolix), the first oral hormone therapy for treating advanced prostate cancer.
Global Oral Biologics Market: Key Developments
In January 2020, the European Medicines Agency recommended granting a marketing authorization in the European Union (EU) for oral Rybelsus (semaglutide) for the treatment of adults with insufficiently controlled type 2 diabetes to improve glycaemic control as an adjunct to diet and exercise. Rybelsus is the first glucagon-like peptide (GLP-1) receptor agonist treatment, a class of non-insulin medicines for people with type 2 diabetes.
In June 2020, the U.S. FDA approved MYCAPSSA, the first and only oral somatostatin analogue (SSA), for the long-term maintenance treatment of acromegaly patients who had responded to and tolerated treatment with octreotide or lanreotide. The Mycapssa is manufactured by Chiasma, Inc., a commercial-stage biopharmaceutical company.
In February 2022, Rani Therapeutics Holdings, Inc., a clinical-stage biotherapeutics company, announced the development of RaniPill HC, a high-capacity oral biologics device. The newly launched RaniPill HC is capable of delivering up to a 500%-plus higher drug payload than Rani’s existing oral biologic capsule.
Global Oral Biologics Market: Key Trends
Increasing number of partnerships between the key market players
An increasing number of partnerships between the key market players is expected to boost market growth over the forecast period. For instance, in June 2023, Rani Therapeutics Holdings, Inc. announced that the company had extended its existing partnership with Celltrion, a biopharmaceutical company, by entering into an agreement for the development of RT-105, an orally administered adalimumab biosimilar.
Global Oral Biologics Market: Restraint
Product recalls by the manufacturers
Voluntary recalls of oral biologics such as hormones are expected to hinder market growth over the forecast period. For instance, in May 2020, Pfizer, a U.S.-based biotechnology company, announced a voluntary recall of Duavee, an estrogen-based menopause hormone therapy, to ensure product stability throughout its shelf life.
The manufacturing companies are expected to standardize the manufacturing and production processes of the drugs to ensure product quality and stability throughout their shelf life.
Global Oral Biologics Market - Key Players
Major players operating in the global oral biologics market Novo Nordisk A/S, Biocon Limited, Oramed Pharmaceuticals, Inc., Rani Therapeutics, Entera Bio Ltd., AbbVie Inc., Enteris BioPharma, Inc., Chiasma, Inc. and Allena Pharmaceuticals, Inc.
*Definition: Biologics drugs can be defined as molecules derived from living cells such as microorganism, plant, or animals and used for the treatment, diagnosis, or prevention of various diseases. Biologic therapeutics has proven to be a highly effective treatment for various chronic ailments, such as diabetes, plaque psoriasis, arthritis, Crohn’s disease, and ulcerative colitis.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients