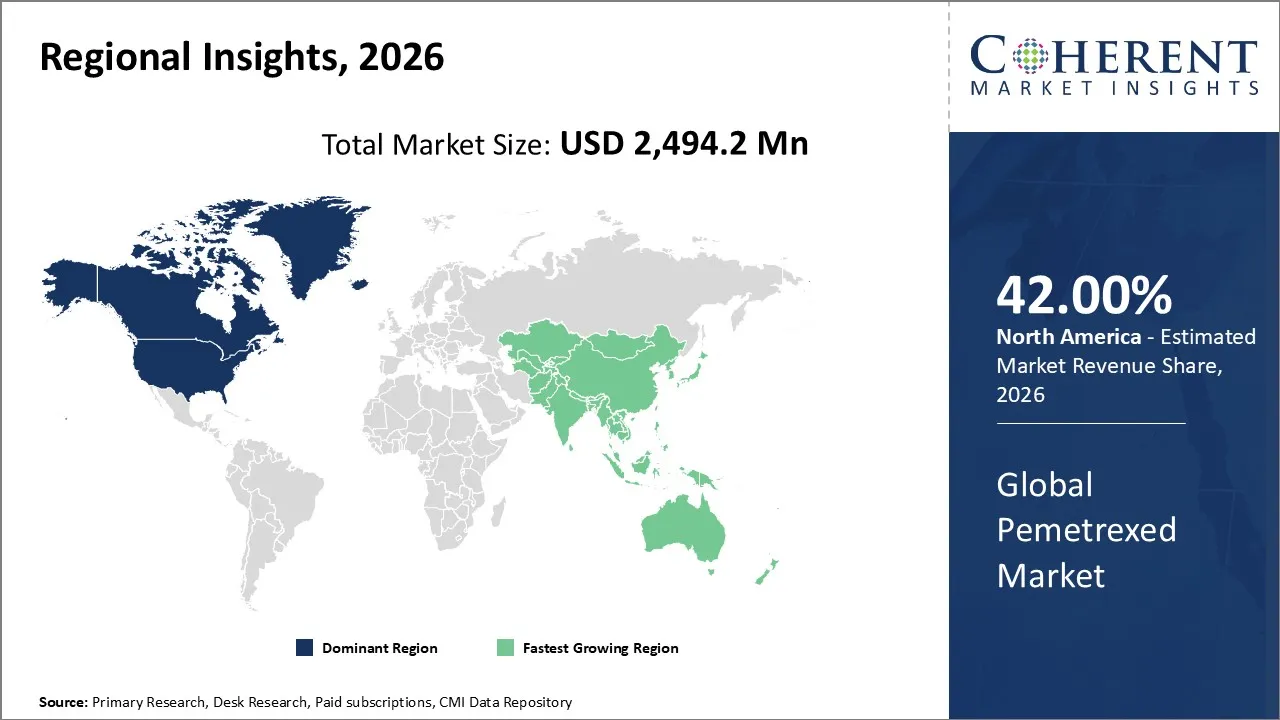
Pemetrexed market is estimated to be valued at USD 2,494.2 Mn in 2026 and is expected to reach USD 2,907.6 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 2.2% from 2026 to 2033.
Increase in incidence of lung cancer and malignant pleural mesothelioma worldwide is one of the major factor fueling demand for chemotherapy drugs involving pemetrexed. Pemetrexed, used as part of combination chemotherapy, is commonly applied in chemotherapy treatment of non-small cell lung cancer, which accounts for a large share of worldwide cancer incidence rates. Increasing general public awareness of the importance of early diagnosis of cancers, along with improved access to oncology treatment services, also contributes to steady growth in chemotherapy treatment of cancer.
|
Current Events |
and its impact |
|
Regulatory and Patent Landscape Changes |
|
|
Geopolitical Trade Tensions and Supply Chain Disruptions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on strength, the 500 mg segment is likely to generate the highest level of market share, considered to be 62% of the total market share in 2026. Pemetrexed 500 mg vials are generally employed in the treatment of non-small cell lung cancer and malignant pleural mesothelioma with platinum-based regimens in combination with immunotherapy.
For instance, in April 2024, Amneal Pharmaceuticals launched PEMRYDI RTU, a pemetrexed formulation in ready-to-use injectables, including the 500 mg/50 mL vial preparation, which does not require reconstitution, dilution, or refrigeration.
Based on distribution channel, the hospital pharmacies segment is expected to contribute the highest market share of around 60% in 2026. As pemetrexed is administered intravenously and under strict medical supervision, a significant number of treatments take place in the oncology departments of hospitals and specialty cancer centers. Hospital pharmacies manage the procurement, storage, preparation, and dispensing of drugs according to regulatory standards for cytotoxic medications.
For instance, Amneal Pharmaceuticals and Shilpa Medicare jointly announced the commercial launch of "PEMRI DI RTU," the first "Ready to Use" Pemetrexed Injection that does not require reconstitution, dilution, and refrigeration.
To learn more about this report, Request Free Sample
North America is poised to account for the largest share of 42% in the pemetrexed market in 2026. Advanced healthcare infrastructure, a high prevalence of lung cancer and malignant pleural mesothelioma, strong reimbursement policies, and early adoption of chemotherapy treatments are some of the factors contributing to market growth in the region. Demand from hospital pharmacies and oncology centers remains high as pemetrexed continues to be a standard chemotherapy drug used in the treatment regimens.
For instance, in January 2025, Johnson & Johnson announced that Health Canada had given its Notice of Compliance (NOC) to RYBREVANT (amivantamab) in combination with carboplatin and pemetrexed to treat patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) expressing certain EGFR mutations and who had previously received therapy.
The Asia Pacific market is expected to be the fastest-growing market for pemetrexed because of the increasing rate of cancer incidence, investment in health infrastructure, and increasing access to chemotherapy and cancer awareness regarding early detection of cancer. The availability of generics of pemetrexed at a lower cost is driving the market in emerging countries such as China, India, and Japan.
For instance, in January 2026, Shilpa Medicare highlighted the growing use of PEMRYDI RTU® in hospital pharmacies across Asia Pacific, emphasizing its ready-to-use formulation that reduces preparation time and supports safe, efficient administration of chemotherapy in institutional settings.
The United States pemetrexed market is still rising due to an increase in cancer prevalence, well-developed healthcare infrastructure, and rising institutional use of chemotherapy. It has remained a consistent drug in chemotherapy regimens used to treat NSCLC and malignant pleural mesothelioma, with an established support base generated from hospital pharmacy networks.
For instance, in April 2024, Amneal Pharmaceuticals launched PEMRYDI RTU® in the U.S. — a ready‑to‑use pemetrexed injection available in 500 mg and 100 mg strengths that eliminates reconstitution and refrigeration requirements.
China’s rapid expansion of healthcare infrastructure and growing access to oncology treatments are driving increased adoption of pemetrexed therapies. The government’s focus on improving cancer diagnostics and treatment availability, along with expanded reimbursement coverage, supports institutional procurement of chemotherapy drugs and broadens the patient base that can access pemetrexed.
For instance, in June 2024, China’s National Medical Products Administration (NMPA) approved AstraZeneca’s Tagrisso (osimertinib) in combination with pemetrexed and platinum‑based chemotherapy.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,494.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.2% | 2033 Value Projection: | USD 2,907.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eli Lilly and Company, Stada Arzneimittel AG, Eagle Pharmaceuticals, Inc, Fresenius Kabi USA, Teva Pharmaceuticals USA, Inc, Biocon, Pfizer Inc., Apotex Inc, Dr. Reddy’s Laboratories Ltd, Cadila Pharmaceuticals., Accord Healthcare, Accure Labs Pvt. Ltd, Shanghai Henlius Biotech, Inc., Janssen Global Services, LLC, and Checkpoint Therapeutics, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of NSCLC and malignant pleural mesothelioma, especially in North America and the Asia Pacific, propels the demand for pemetrexed. Since the medication represents a first-line chemotherapeutic agent in standard treatment regimen, hospital pharmacies and oncology centers are also posting regular growth in consumption. The growing awareness about early detection of cancer and better diagnostic skills further enhances the adoption of pemetrexed in clinical practice.
The ready-to-use pemetrexed injections offer growth potential, simplifying the hospital pharmacy process by eliminating major steps of reconstitution and refrigeration. Besides this, the increasing availability of generic pemetrexed formulations in major markets such as the U.S., China, and Europe ensures cost-effective options for healthcare providers, thus improving access to treatment and driving market growth. There is also an opportunity to integrate pemetrexed into combination therapies with targeted agents, increasing its usage across diverse patient populations.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients