Global plasma derived medicine market is estimated to be valued at USD 21.38 Bn in 2026 and is expected to reach USD 35.70 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.6% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The market growth is driven by rising prevalence of life-threatening diseases and increasing demand for plasma derived products for treating rare diseases. Plasma derived medicines play a crucial role in the treatment of various disease indications such as primary immunodeficiency, bleeding disorders, and other conditions where replacement clotting factors are required. With no effective substitutes available for plasma derived products and development of newer therapies, there will be huge demand for plasma derived medicines over the forecast period.
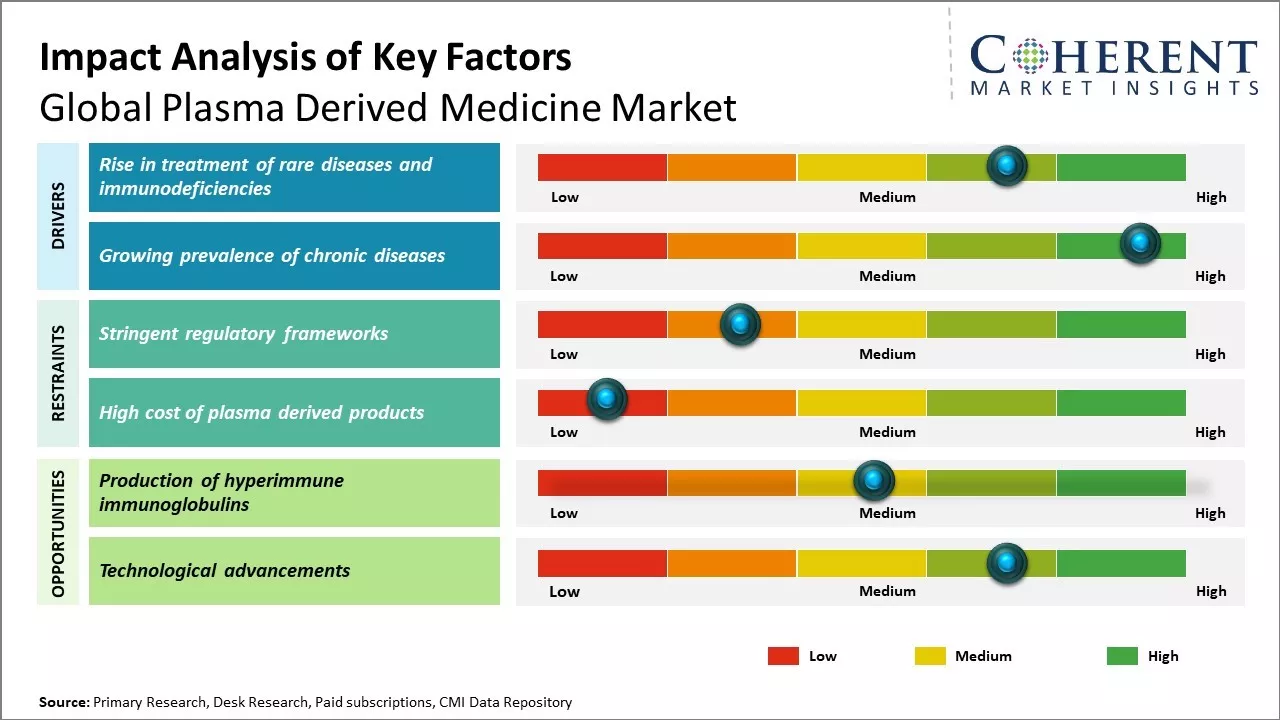
Market Driver - Rise in treatment of rare diseases and immunodeficiencies
There has been increase in usage of plasma derived medicines for the treatment of rare diseases and immunodeficiencies. Plasma derived medicines contain therapeutic plasma proteins that come from donated human blood and serve as crucial treatment options for patients who lack naturally occurring proteins in their blood. Various immunodeficient conditions such as primary immunodeficiencies that weaken patient’s immune systems rely majorly on plasma derived medicines. Growing diagnosis rates of these rare conditions coupled with expanding knowledge about their clinical manifestations and available therapeutic interventions has boosted demand for plasma derived products.
Rising survival rates of patients with rare and complex diseases due to improved management has led to increase in patients requiring long term management with plasma therapies. Multiple companies are actively involved in research and development of plasma derived therapies for rare diseases as many of these conditions lack alternative treatment options. Regulatory bodies have also simplified approval pathways and provided market exclusivity incentives for orphan drugs, encouraging pharmaceutical firms to invest in developing medicines for unmet medical needs of small patient populations. Furthermore, patient advocacy groups have raised significant awareness about rare diseases, helping to accelerate diagnosis and expand the pool of patients identified for long term plasma replacement therapies.
Get actionable strategies to beat competition: Request Free Sample
Growing prevalence of chronic diseases
Rising prevalence of chronic diseases worldwide boosts uptake of plasma derived therapies. Conditions like primary immune thrombocytopenia (ITP), hemophilia and neurological disorders demand lifelong management, often with plasma derived medicines such as immunoglobulin therapies. Growing geriatric population prone to age related chronic illnesses also boost need for plasma products as replacement therapies. It is estimated that over 50% of plasma collected globally is utilized in manufacturing medicines for chronic diseases.
Lifestyle changes, expanding urbanization and increased life expectancy have led to rise in non-communicable chronic ailments. Developed nations have witnessed substantial disease burdens from chronic conditions which now account for the majority of national healthcare budgets. Emerging countries are also experiencing an epidemiological shift towards chronic illnesses due to rapid economic growth and changing dietary patterns. All of these factors have elevated the pool of patients requiring long term or repeated use of plasma therapies. Chronic diseases represent one of the biggest therapeutic areas for plasma derived medications.
Key Takeaways from Analyst:
Global plasma derived medicine market growth is driven by rising prevalence of life-threatening diseases like hemophilia and increasing plasma collection worldwide. Plasma derived medicines play a crucial role in the treatment of immunodeficiency disorders.
North America dominates the market currently due to high healthcare expenditures and rising healthcare awareness. However, Asia Pacific is expected to witness the fastest growth due to growing healthcare facilities and disposable income in emerging economies like China and India.
Stringent regulations surrounding plasma collection and production processes can pose challenges for market players. Unfavorable reimbursement policies especially in developing nations can hamper the market growth.
Alternatives like gene therapy and recombinant proteins can hamper the market share of plasma derived medicines. However, ongoing R&D in novel plasma products and growing acceptance of intravenous immunoglobulins (IVIG) for various neurological and inflammatory diseases can provide growth opportunities.
Overall, with rising healthcare infrastructure, the global plasma derived medicine market is well-positioned to grow steadily driven by increase in plasma volume collection and production of new products.
Market Challenge - Stringent regulatory frameworks
Global plasma derived medicine market growth can be hampered due to presence of stringent regulatory frameworks governing the manufacturing and approval of plasma derived medicines. The production of plasma derived medicines involves collection of human plasma, which raises several safety and ethical concerns. To ensure the safety of plasma collection process and final medicines, regulations bodies across countries have implemented strict rules for plasma collection centers and manufacturers. Complying with various international standards such as cGMP further increases the production and operating costs for market players. Obtaining regulatory approvals for new plasma derived drugs or significant changes to the existing manufacturing processes has also become a time-consuming process due to complex standards. The cumbersome regulatory conditions can delay the market entry of new products and significantly affect the revenue generation capabilities of companies.
Market Opportunity: Production of hyperimmune immunoglobulins
The production of hyperimmune immunoglobulins can offer growth opportunity for players in the global plasma derived medicine market. Immunoglobulins are antibodies that are derived from blood plasma of donors with high antibody count against specific diseases. Hyperimmune immunoglobulins are immunoglobulin preparations produced from plasma specifically collected from donors with high levels of antibodies against targeted diseases. Increasing production of hyperimmune immunoglobulin products for different indications can offer growth opportunities. For example, manufacturers can focus on developing and commercializing hyperimmune products for diseases like rabies, hepatitis B, varicella zoster and tetanus, where immunoglobulin therapy delivers high clinical effectiveness. With growing spending on healthcare and emphasis on developing targeted therapies, there will be huge demand for hyperimmune immunoglobulins in the near future.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
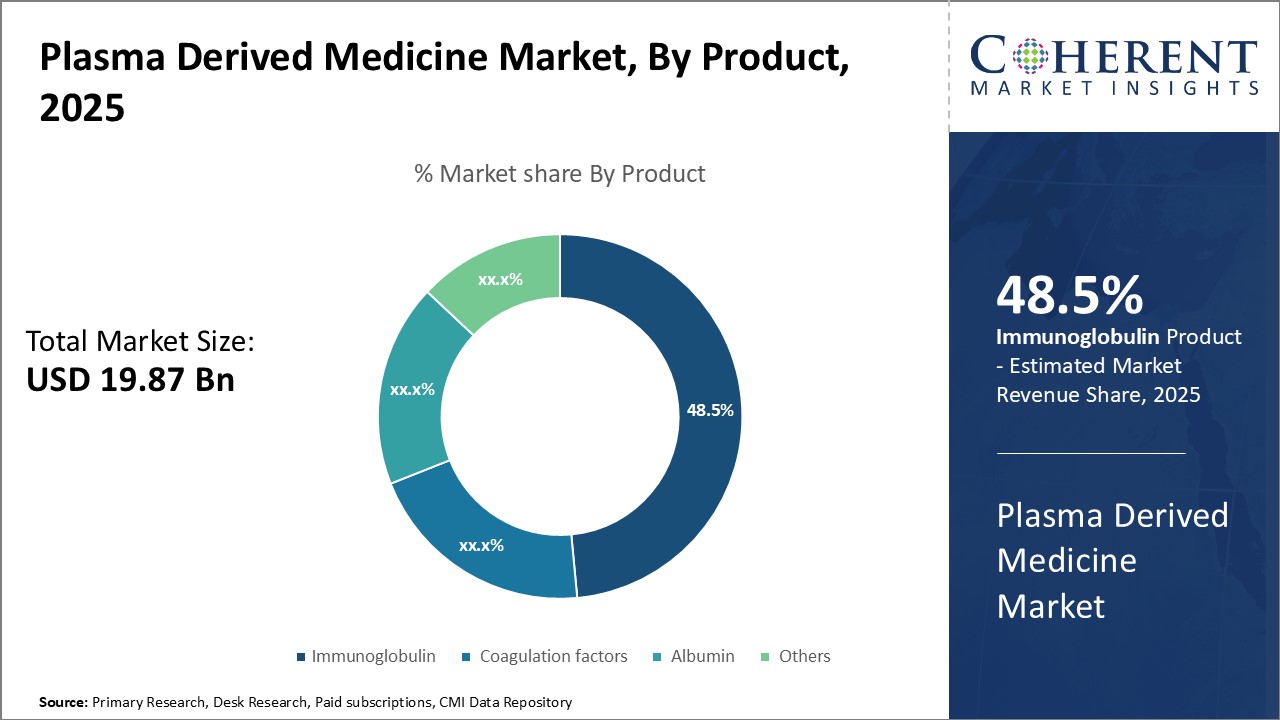
By Product - Driving immunity through immunoglobulin
In terms of product, immunoglobulin segment is expected to contribute the highest market share of 49.5% in 2026, owing to its ability to treat a wide range of immune disorders and diseases. Immunoglobulin is used for treating numerous conditions that are caused due to immunodeficiency or failure of the immune system to defend against foreign bodies. It helps to replace missing or non-functional antibodies in the body, which are essential for fighting infections and pathogens. As a result, immunoglobulin plays a vital role in improving immunity and reducing severity and frequency of infections. Its applications range from treating primary immunodeficiencies and neurological disorders to providing passive immunity. Moreover, continual innovations allowing subcutaneous administration of immunoglobulin have boosted patient compliance and demand. Steady approval of its new indications can drive the segment growth. Rising prevalence of immunological disorder can boost demand for immunoglobulin derived plasma medicines globally.
To learn more about this report, Request Free Sample
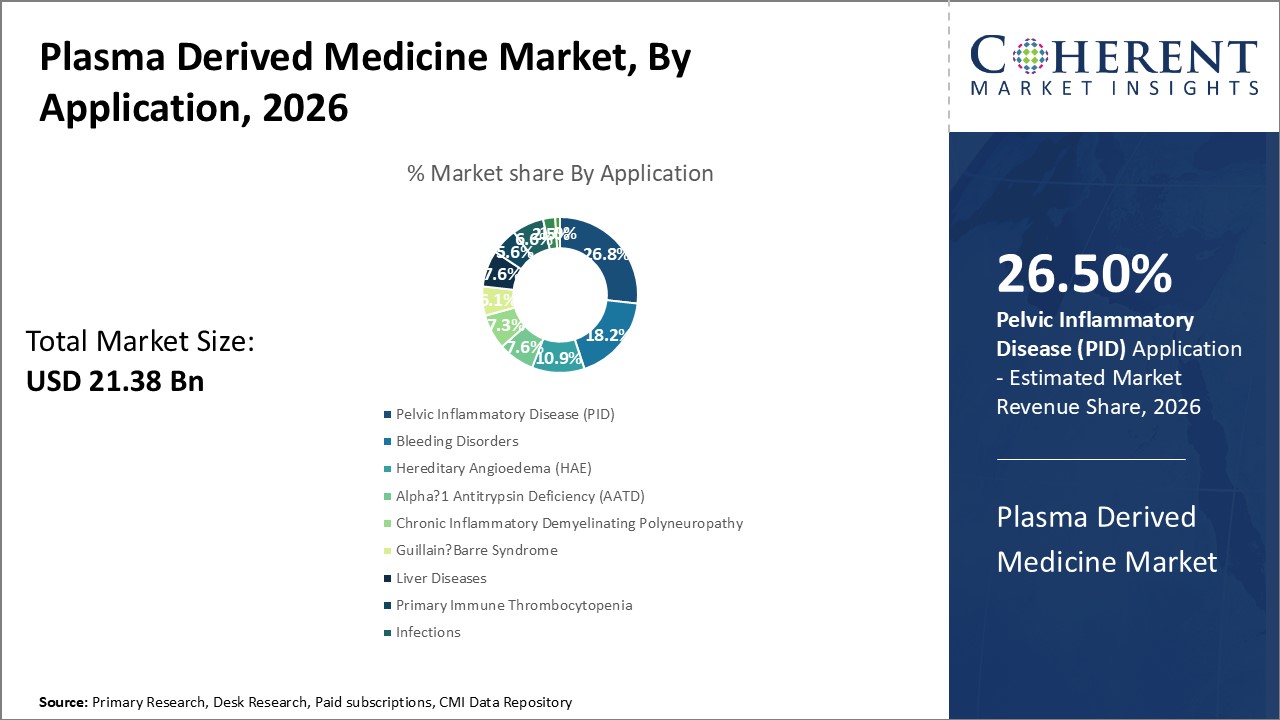
By Application- Treating bleeding disorders through coagulation factors
In terms of application, pelvic inflammatory disease (PID) segment is expected to contribute the highest market share of 26.5% in 2026, owing to the wide-ranging treatments offered by plasma derived coagulation factors. Conditions like hemophilia where coagulation factors are deficient or dysfunctional are effectively managed using plasma derived replacement therapy. Regular prophylactic treatment helps prevent painful spontaneous internal bleeding episodes in patients. It substantially improves their quality of life by enabling an active lifestyle with reduced joint damage risks. Growing hemophilia patient base accompanied by enhanced diagnosis has majorly propelled this segment growth. Rising life expectancies have boosted demand for lifesaving replacement therapy worldwide.
By Distribution Channel- Convenient hospital administration through pharmacies
In terms of distribution channel, hospital pharmacies segment is expected to contribute the highest market share of 49.5% in 2026, due to the convenience offered to patients, physicians as well as the healthcare system. Most plasma derived medicines demand refrigeration and have short shelf lives requiring careful handling. Their administration also needs monitoring under medical supervision. Hospital settings offer the most structured environment for distributing and administering such therapeutics. It ensures proper storage, timely availability as well as supervised usage. The distribution chain is also streamlined to hospitals' inventory management and billing systems. This eases logistics and improves treatment adherence for conditions requiring lifelong therapy like primary immunodeficiencies. It maximizes access and uptake of life-saving plasma medicines relevant to different disease areas.
Need a Different Region or Segment? Request Free Sample
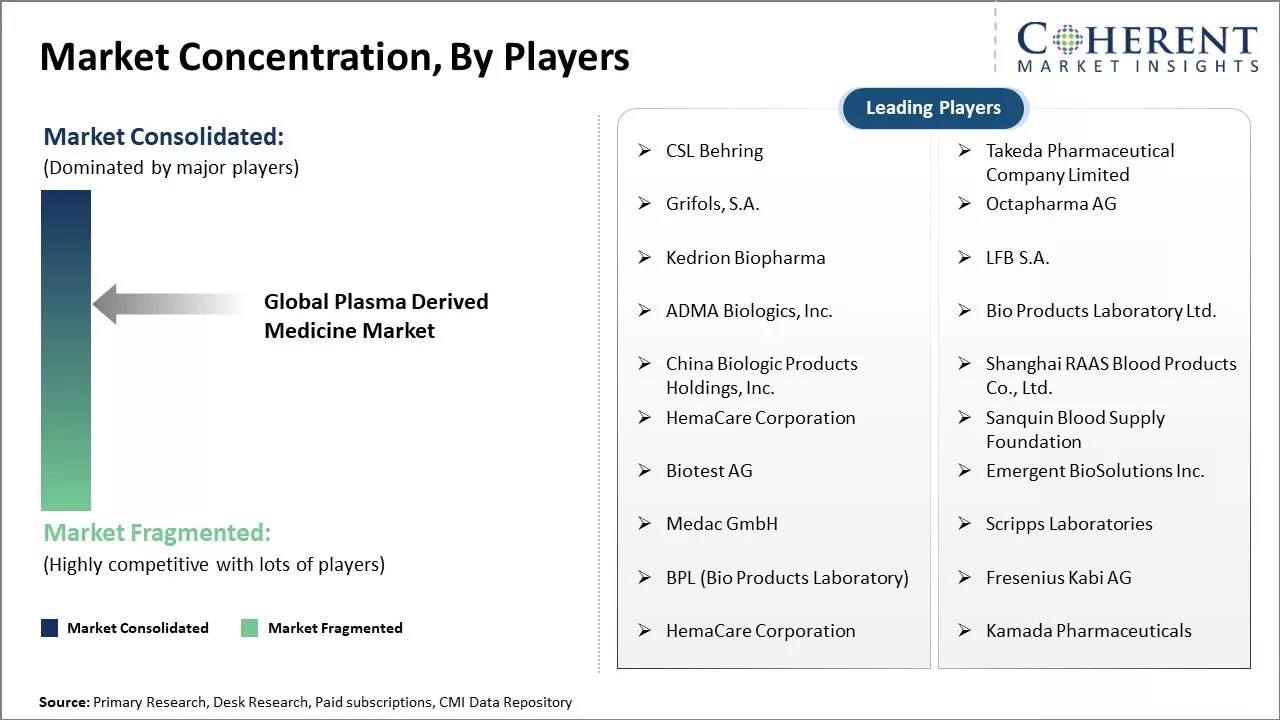
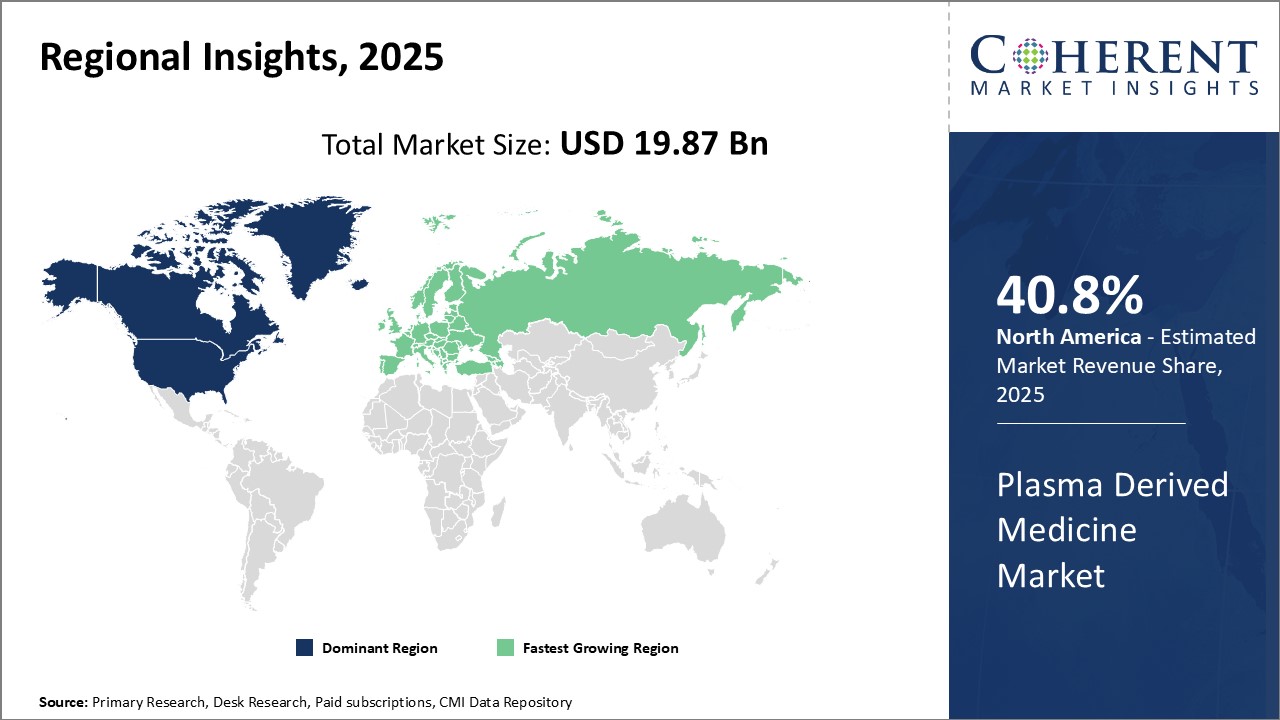
North America has established itself as the dominant region in the global plasma derived medicine market with an estimated market share of 40.8% in 2026. The presence of major industry players such as Shire, CSL Behring and Grifols has boosted the market growth in the region. These leading companies have continued to invest heavily in R&D as well as in expanding their product portfolios. Rising healthcare expenditure and presence of advanced medical facilities have boosted demand for plasma derived drugs. The favorable regulatory environment under FDA also attract new players to set up their manufacturing facilities in the region.
Europe has the most developed plasma collection infrastructure with countries like Germany and Austria at the forefront. The region witnesses millions of plasma collection on a regular basis. This abundant plasma resource has enabled European companies to establish themselves as leading exporters of plasma derived medicines. Key markets like Germany and U.K. dominate the imports for rest of the world. However, increasing presence of U.S players has intensified the competition for European companies in their home markets.
China has emerged as the fastest growing regional market for plasma derived medicine. Rapid economic growth, growing medical needs and rising healthcare expenditures drive the market growth. The government's push for developing domestic biologics industry and reducing imports dependence has attracted significant investments from multinational companies for setting up local plasma fractionation facilities. Regional self-sufficiency holds the potential to make China a global production powerhouse.
Plasma Derived Medicine Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.38 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.6% | 2033 Value Projection: | USD 35.70 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
CSL Behring, Takeda Pharmaceutical Company Limited, Grifols, S.A., Octapharma AG, KedrionBiopharma, LFB S.A., ADMA Biologics, Inc., Bio Products Laboratory Ltd., China Biologic Products Holdings, Inc., Shanghai RAAS Blood Products Co., Ltd., HemaCare Corporation, Sanquin Blood Supply Foundation, Biotest AG, Emergent BioSolutions Inc., Medac GmbH, Scripps Laboratories, BPL (Bio Products Laboratory), Fresenius Kabi AG, and Kamada Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global plasma derived medicine market includes pharmaceutical therapies extracted and purified from human blood plasma. These medications are essential for treating various rare diseases and immune disorders that result from deficiencies in specific proteins found in blood plasma. Key products in this market include immunoglobulins, coagulation factors, albumin, and other specialized plasma-derived treatments. These vital medicines are crucial for enhancing patients' quality of life by aiding in the management of chronic conditions.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients