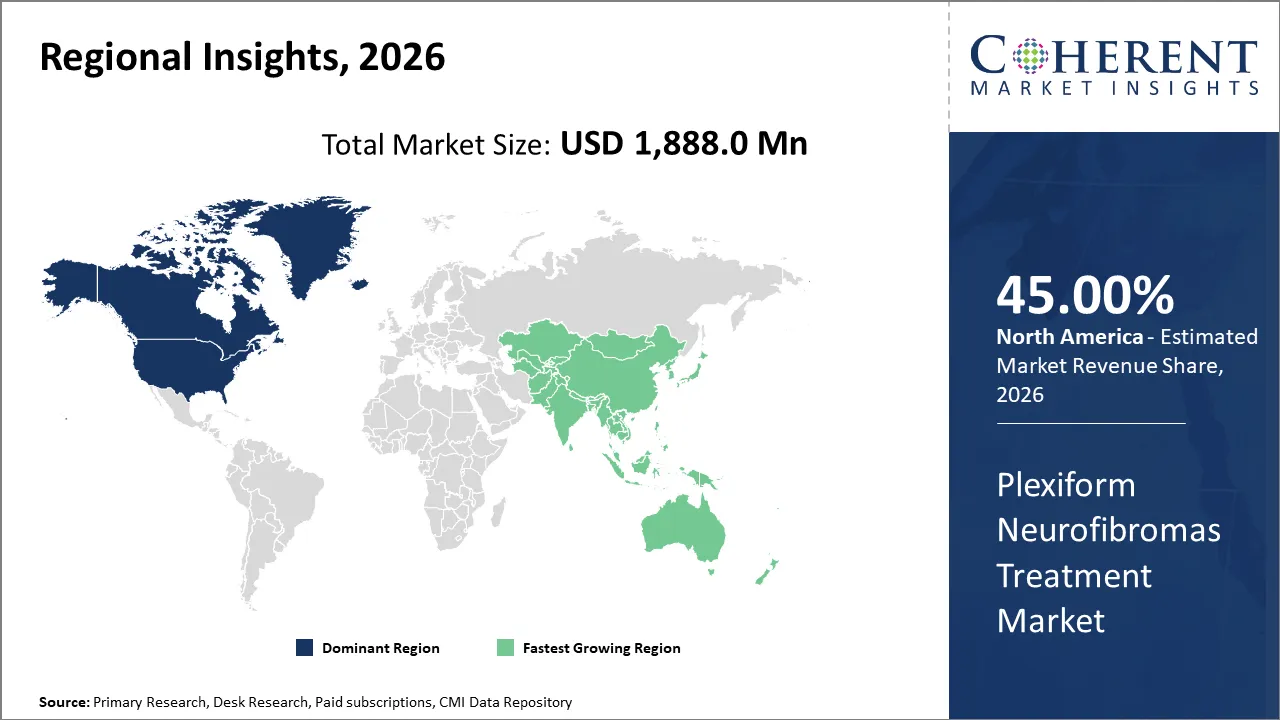
The global plexiform neurofibromas treatment market size is expected to reach approximately USD 1,888.0 Mn in 2026 and USD 3,265.0 Mn by 2033, growing at a CAGR of 8.4% throughout the forecast period (2026-2033).
Plexiform neurofibromas develop from nerve sheath cells and often present at birth or early childhood. It is complex, benign tumors that grow along nerves and their branches. They are most commonly associated with neurofibromatosis type 1 (NF1), a hereditary condition. Rising prevalence of NF1 increasing number of patients with neurofibromatosis type 1 expands the target patient population. The plexiform neurofibromas market is gaining strategic importance due to advancement in targeted therapies such as selumetinib, strong unmet medical need, supportive regulatory policies and improving healthcare access.
|
Current Event |
Description and its Impact |
|
Increasing regulatory approval of new drug treatment in the emerging markets |
|
|
Increasing Clinical Trial Studies for the Plexiform Neurofibromas Treatment |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Selumetinib, categorized by its drug class, is projected to command a significant market share, roughly 58%, by the year 2026. Selumetinib is the first drug approved by the U.S. Food and Drug Administration specifically for plexiform neurofibromas and gives a first-mover advantage and strong market penetration. It is easy to administer an oral drug and used for long-term disease management, which improves patient compliance and continuous revenue generation. The selumetinib drug is widely used in children with inoperable tumors and henceforth has high treatment demand in the pediatric segment, which boosts overall market share.
By patient population, the hospital pharmacies segment is estimated to account for a prominent market share of 61% in 2026. As the plexiform neurofibromas is early onset disease it leads to a larger pediatric patient pool. Most cases are identified at a young age due to visible symptoms and genetic screening which results in early entry into treatment pathway. Key drugs like Selumetinib were initially approved for pediatric use and drives high adoption in children compared to adults. Many clinical studies and trial focus on children leads to faster innovation and better treatment availability in paediatrics. Tumors often grow during childhood and adolescene which requires early and continuous intervention.
Based on distribution channel, hospital pharmacies segment led the global plexiform neurofibromas treatment market with share of 46% in 2026. Plexiform neurofibromas require multidisciplinary care and specialist supervision and treatments are primarily initiated in hospital environments. Innovative drugs like selumetinib are typically prescribed and dispensed through hospital systems which ensures controlled and safe drug administration. Patients require regular monitoring, side-effect management and dose adjustments which is supported by hospital pharmacies integrated clinical care. Many treatments are covered under hospital-based insurance systems which improves patient access to costly therapies. Hospitals provide oncologists, neurologists and genetic specialists which enhances treatment accuracy and drug utilization.
To learn more about this report, Request Free Sample
North America is expected to dominate the plexiform neurofibromas treatment market, accounting for a share of 45% in 2026. North America leads the market owing to early innovation adoption, advanced healthcare systems, and high treatment accessibility. Rapid regulatory approvals by the U.S. Food and Drug Administration (FDA) and early availability of advanced drugs like selumetinib ensures first-mover advantage and higher treatment adoption. North America’s patient pool has easy access to accurate diagnosis and advanced treatments due to the presence of specialized hospitals and neuro-oncology and rare disease center. Moreover, the presence of leading pharma and biotech firms are headquartered or highly active in North America which drives commercialization and innovation of new therapies. North America especially the U.S. has one of the highest healthcare spending levels globally supports adoption of premium and innovative therapies. These factors collectively enable greater access to innovative treatments in North America region and support the dominance in the market.
Asia Pacific region is estimated to grow at highest CAGR in the market owing to large patient population, rapid improvement in healthcare infrastructure, increasing awareness and diagnosis rates and supportive government initiatives. Increasing number of clinical trials and pharma activities in Asia Pacific region boosts local availability of new treatments. For instance, in December 2025, Shanghai Kechow Pharma, Inc., pharmaceutical company based in China, revealed the primary completion of the Single-arm Phase II Study to evaluate the efficacy and safety of HL-085 (tunlametinib) in the treatment of adult participants with Neurofibromatosis Type 1 (NF1). Oral administration of HL-085 is intended to show its potential for tumor reduction and better patient outcomes. Shanghai Kechow Pharma, Inc. is the sponsor of the trial. Furthermore, governments are increasing healthcare budgets and focus on innovating treatment for rare disease which supports adoption of high-cost innovative therapies.
The U.S. country held the dominant revenue share in the North America plexiform neurofibromas treatment market owing to rising prevalence, increasing approval of new drug therapy by the U.S. FDA, high adoption of genetic testing & screening and strong presence of specialized treatment centers. Patient in the U.S. are more likely to receive high-cost innovative therapies and strong willingness-to-pay supported by insurance, which drives higher revenue generation per patient. Rapid new drug approval by the U.S. Food and Drug Administration supports quick adoption of targeted drugs like Selumetinib and emerging therapies such as Mirdametinib. Early adoption of innovative therapies ensures faster availability and widespread clinical use of advanced treatments, driving market growth.
China is the dominant country in the Asia Pacific market, driven by large patient base, rapid healthcare expansion, and increasing access to advanced therapies. Expansion of local biotech and pharma companies has boosted the availability and affordability of treatments within China country. Moreover, improving health insurance coverage and rising healthcare expenditure in China supports adoption of high-cost innovative therapies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,888.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.4% | 2033 Value Projection: | USD 3,265.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca, Pfizer Inc., Sun Pharmaceuticals Industries Limited, Mallinckrodt Pharmaceuticals, SpringWorks Therapeutics, Alcaliber S.A, Teva Pharmaceutical Industries Ltd., Glenmark, Amneal Pharmaceuticals LLC, Aurobindo Pharma, Apotex Inc., Mylan N.V., GSK plc., Solara Active Pharma Sciences Ltd., Abbott, Shanghai Kechow Pharma, Inc., Endo Pharmaceuticals Inc., Purdue Pharma L.P, Merck & Co., Inc., NFlection Therapeutics, Healx, and Array Biopharma |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence of neurofibromatosis type 1 (NF1) has significantly increased the demand for plexiform neurofibromas treatment and driven the growth of the market. For instance, according to the data published by the National Center for Biotechnology Information, approximately 1 in 3,000 to 1 in 4,000 people globally have NF1, one of the most prevalent tumor risk disorders. About 1 in 2,500 to 1 in 3,000 live newborns experience it. By maturity, NF1 is fully penetrant, but as people age, many of its symptoms worsen or become more common. A hereditary condition called neurofibromatosis type 1 (NF1) increases the likelihood of developing tumors. 50% of NF1 patients have plexiform neurofibromas (PNs), which significantly increase morbidity when surgery is not an option. Hence, increasing patient pool directly increases demand for treatment.
Emergence of next-generation drugs has significantly driven the growth of the market. New treatments, like Mirdametinib, which gained approval in 2025, are broadening the possibilities for addressing plexiform neurofibromas. The major pharmaceutical companies are concentrating their efforts on creating new drugs that feature enhanced MEK inhibitors, along with refined dosing regimens and improved tolerability profiles. The emergence of gene therapies, targeted therapies, immunotherapies, and combination approaches suggests that next-generation drug could provide patients with plexiform neurofibromas more effective and tailored treatment options, potentially leading to significantly better outcomes. This is creating a dynamic drug landscape and proves to be significant trend in driving the growth of the market.
Increasing launch of advanced diagnostic technologies and integration of AI significantly drive the growth of the market. Use of digital tumor monitoring and AI-based imaging helps in early diagnosis and better treatment tracking. For efficient treatment, early detection of these tumors is important, and AI integration helps make it easy. Hence, advancements in diagnostics and integration of AI improve clinical outcomes and treatment efficiency.
Rise of personalized medicine and growing adoption of it for treatment of plexiform neurofibromas has estimated to create significant opportunity for the growth of the plexiform neurofibromas treatment market. Use of genetic profiling and molecular diagnostics in manufacturing drug treatment for plexiform neurofibromas enables patient specific treatment and better clinical outcomes. Personalized therapies are expected to be a major future growth pillar.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients