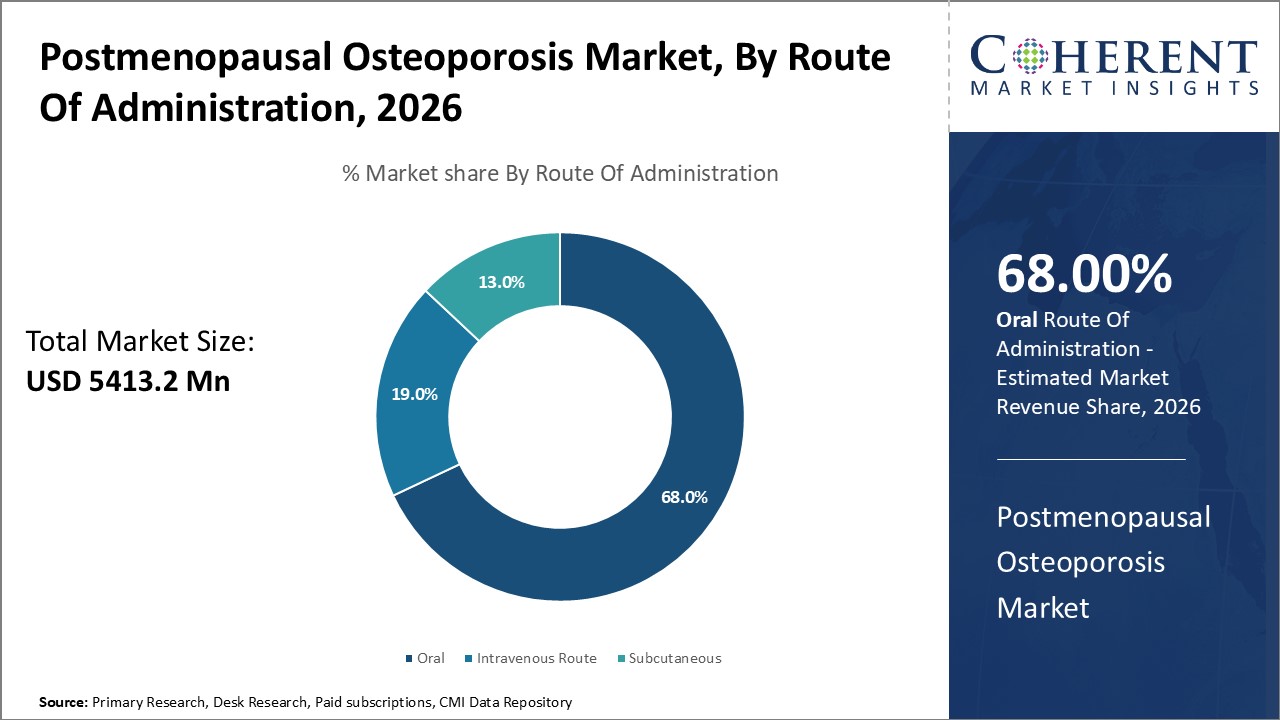
Postmenopausal Osteoporosis Market is estimated to be valued at USD 5,413.2 Mn in 2026 and is expected to reach USD 7,416.0 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of4.6% from 2026 to 2033.
The postmenopausal osteoporosis market is experiencing significant growth, driven by several key factors. The aging global population, particularly the increasing number of postmenopausal women, has led to a higher prevalence of osteoporosis, thereby boosting demand for effective treatments. Advancements in drug development, including the introduction of biologics and combination therapies, have improved treatment efficacy and patient outcomes. Additionally, increased awareness and education about osteoporosis have led to earlier diagnosis and proactive management, further propelling market growth. The expansion of healthcare infrastructure and reimbursement policies in emerging economies has also facilitated greater access to osteoporosis treatments. These combined factors are expected to continue driving the market's expansion in the coming years.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Changes |
|
|
Drug Development & Market Dynamics |
|
|
Global Health Initiatives |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial Intelligence (AI) is improving drug research, diagnosis, and treatment customisation, which is revolutionizing the postmenopausal osteoporosis market. By more precisely evaluating bone mineral density scans (like DEXA), AI-powered diagnostic tools are enhancing early osteoporosis identification by allowing physicians to spot minute changes that the human eye might overlook. Additionally, machine learning algorithms are being used to predict fracture risk using patient-specific information such as age, medical history, and genetic markers. This allows for more individualized treatment plans. AI in drug discovery expedites the process of finding new therapeutic targets and, by forecasting patient reactions, improves the design of clinical trials.
For example, UCB plans to license its AI technology for enhanced osteoporosis detection to ImageBiopsy Lab.
Medicare covers a number of osteoporosis-related services in the United States. Outpatient screenings including bone density tests and some injectable therapies like denosumab (Prolia) are covered by Medicare Part B if certain requirements are met. For example, if a woman has osteoporosis, is eligible for Medicare home health services, and suffers a related fracture, Part B may pay for Prolia injections. Different reimbursement schemes for osteoporosis treatments are found in European nations. After receiving bisphosphonates, denosumab is covered in France as a second-line treatment for postmenopausal women who are at higher risk of fracture. For some patients, particularly those who are intolerant to bisphosphonates, selective estrogen receptor modulators (SERMs), such as raloxifene, are advised and compensated.
The proven effectiveness of bisphosphonates in halting bone loss and lowering the risk of fractures makes them a mainstay in the treatment of postmenopausal osteoporosis. By preventing osteoclast-mediated bone resorption, these drugs help to preserve or improve bone mineral density. Alendronate, risedronate, and ibandronate are often prescribed drugs that are widely accessible in oral and injectable formulations, providing therapeutic flexibility according to patient requirements and preferences. Generic bisphosphonates' low cost contributes to their broad use, especially in healthcare systems with tight budgets. According to an article published by Versus Arthritis, bisphosphonates are used to lower the risk of fractures in the hip and spine caused by osteoporosis, which is further contributing to the postmenopausal osteoporosis market growth.
To learn more about this report, Request Free Sample
In the treatment of postmenopausal osteoporosis, oral treatments are essential because they provide many patients with a practical and efficient option. These drugs are frequently prescribed because they have been shown to be effective in lowering the risk of fractures and because they are simple to use, especially oral bisphosphonates like alendronate and risedronate. Healthcare professionals can customize treatment strategies to meet the needs of each patient by using flexible dose schedules, such as daily, weekly, or monthly, which improves adherence and results. Furthermore, accessibility and affordability have increased due to the availability of generic formulations, particularly in healthcare settings where costs are a concern. Apart from this, people prefer oral therapy which is further accelerating the postmenopausal osteoporosis market demand. For example, 77% of patients preferred oral therapy, with 70.4% feeling it disrupted their daily life less, 86.9% having no swallowing difficulties, and 56.8% not worried about missing doses.
Hospital pharmacies are crucial distribution channels for pharmaceuticals, especially for patients in need of specialist care, and are therefore important players in the market for postmenopausal osteoporosis treatments. The administration of cutting-edge osteoporosis therapies, including injectable biologics like denosumab and romosozumab, depends heavily on these pharmacies. Hospital pharmacies allow for easy collaboration between pharmacists, doctors, and other healthcare professionals because they are located within medical facilities. By ensuring prompt drug delivery, tracking patient reactions, and modifying treatment regimens as needed, this integration improves patient outcomes overall. Hospital pharmacies serve both inpatient and outpatient patients by giving them prescription drugs both while they are in the hospital and after they are discharged.
To learn more about this report, Request Free Sample
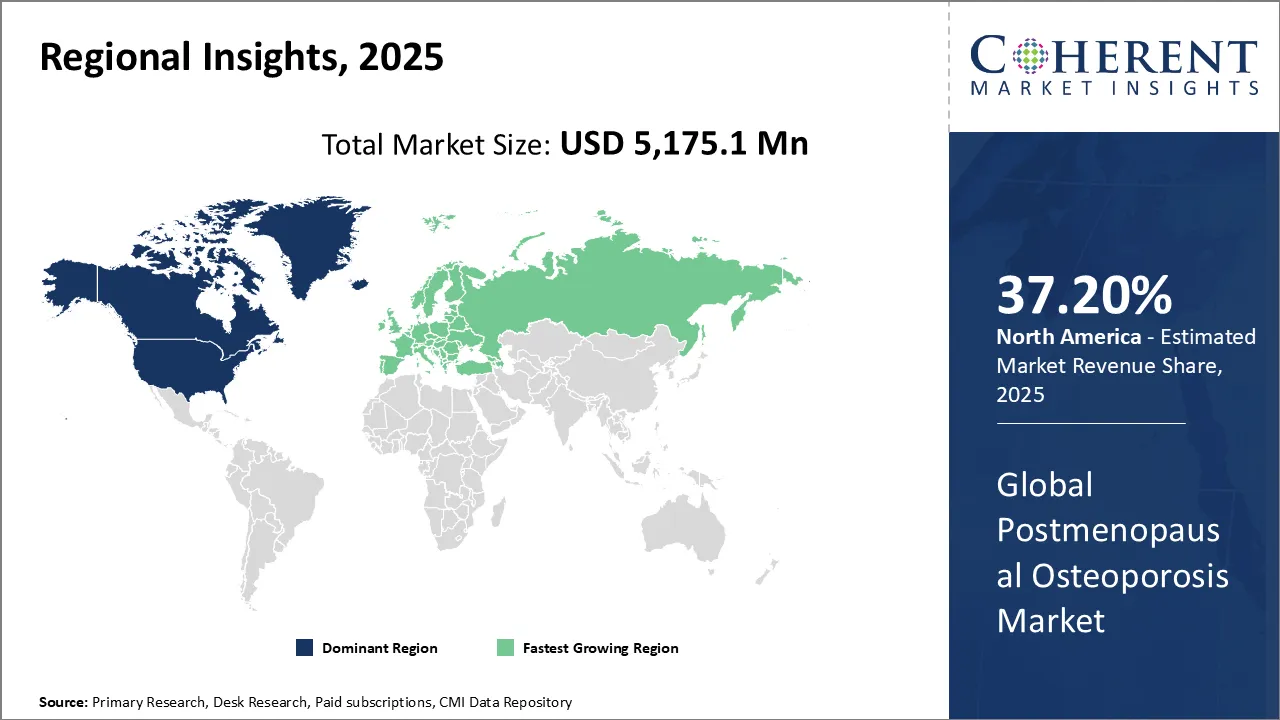
An aging population, greater awareness, and improved treatment choices are all contributing to the postmenopausal osteoporosis market growth in North America. North America acquires 37.2% market share in 2026, with the United States dominating because of its highly developed healthcare system and high diagnostic rates. About 71 million Americans are expected to have osteoporosis or reduced bone mass by 2030, demonstrating the high incidence of osteoporosis among women 50 and older. Furthermore, RANKL inhibitors like denosumab are becoming more popular; in 2024, the FDA approved biosimilars of Jubbonti and Wyost, improving accessibility and affordability. For Instances, The US Food and Drug Administration (FDA) approved Wyost (denosumab-bbdz) and Jubbonti (denosumab-bbdz), the first and only FDA-approved denosumab biosimilars, to treat postmenopausal women with osteoporosis, Sandoz, a major player in generic and biosimilar medications.
The postmenopausal osteoporosis market in Asia Pacific is expanding significantly due to changes in the population, heightened awareness of healthcare issues, and improvements in available treatments. At the front of this expansion are nations like China, Japan, India, and South Korea, each of which makes a distinct contribution to the marketplace dynamics. Reports indicate that approximately 60 million people in India suffer from osteoporosis, with women accounting for 80% of cases. The peak onset of osteoporosis in India happens 10 to 20 years earlier compared to Western countries. Edirol (eldecalcitol), a medication licensed for the treatment of postmenopausal osteoporosis, government assistance, and a rapidly aging population are the main drivers of this rise. The introduction of innovations like romosozumab, a monoclonal antibody that promotes bone growth and reduces bone resorption, in nations like Japan has improved postmenopausal women's treatment options. This is further contributing to the postmenopausal osteoporosis market share.
Growing awareness of bone health, improved treatment choices, and an aging population are all contributing to the postmenopausal osteoporosis industry in the United States. Because of their ability to lower fracture risk and maintain long-term tolerability, bisphosphonates continue to be the most prescribed medication and enjoy a substantial market share. The U.S. Food and Drug Administration (FDA) has approved Prolia™ (denosumab) for treating postmenopausal women with osteoporosis who are at high risk of fractures, further contributing to the postmenopausal market share.
With life expectancy increasing, a significant portion of India's population is aging. For example, an article by the UNFPA states that India’s elderly population, currently at 153 million people aged 60 and above, is projected to rise dramatically to 347 million by 2050. This demographic shift has led to a higher prevalence of osteoporosis, particularly among women, as bone density naturally decreases with age.
The growing use of digital health tools, including wearable technology, AI-powered risk assessment tools, and smartphone applications, is revolutionizing the treatment of osteoporosis by encouraging adherence and facilitating real-time monitoring. Government initiatives and nonprofit campaigns that inform women on lifestyle modifications, calcium and vitamin D consumption, and early screening are fueling the growing movement in favor of preventive treatment. Consultations and follow-ups for osteoporosis are becoming easier with the use of telemedicine, particularly for older women living in remote or disadvantaged locations.
The global increase in the population of women aged 50 and above is a major driver of the PMO market. Postmenopausal women are at high risk of developing osteoporosis due to declining estrogen levels, making them prime targets for treatment. This demographic trend is particularly pronounced in developed nations and rapidly aging regions such as East Asia and Eastern Europe, where healthcare systems are investing more in elderly care.
With the expiration of patents for some leading PMO drugs, there is a growing opportunity for biosimilars to enter the market. These cost-effective alternatives are making treatment more accessible in low- and middle-income countries. Pharmaceutical companies that develop high-quality biosimilars can tap into a wider patient base, particularly where healthcare affordability is a concern.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,413.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.6% | 2033 Value Projection: | USD 7,416.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., F. Hoffmann-La Roche Ltd, Mylan N.V. (Viatris Inc.), Fresenius Kabi AG, Hikma Pharmaceuticals PLC, Novartis AG, Teva Pharmaceutical Industries Ltd., GlaxoSmithKline plc, Sun Pharmaceutical Industries Ltd, Ligand Pharmaceuticals Incorporated, Paras Biopharmaceuticals Finland Oy, Eli Lilly and Company, TRANSCENTA HOLDING, Amgen, Gedeon Richter Plc. Enzene Biosciences, Samsung BioepisL Radius Health, Inc., Alvotech, and AryoGen Pharmed |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients