North America Toxicology Laboratories Market is estimated to be valued at USD 306.3 Mn in 2026 and is expected to reach USD 411.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.3% from 2026 to 2033.
Analysts’ Views on North America Toxicology Laboratories Market:
Factors affecting market growth includes presence of a well-established pharmaceutical industry in the area, high R&D spending, the dominance of major service providers, and growing trend of pharmaceutical and biopharmaceutical companies outsourcing analytical testing. For instance, according to the Organization for Economic Co-operation and Development (OECD's) in October 2022, gross domestic product (GDP) for pharmaceutical spending in the U.S., Canada, and Mexico, in 2020 were 2.08%, 1.72%, and 1.34%, respectively. This demonstrates the substantial participation of big businesses, manufacturers, and governmental bodies in production of product . The major causes of this increase in expenditure are the emphasis on obtaining an edge over competitors and the substantial returns achieved on newly manufactured goods.
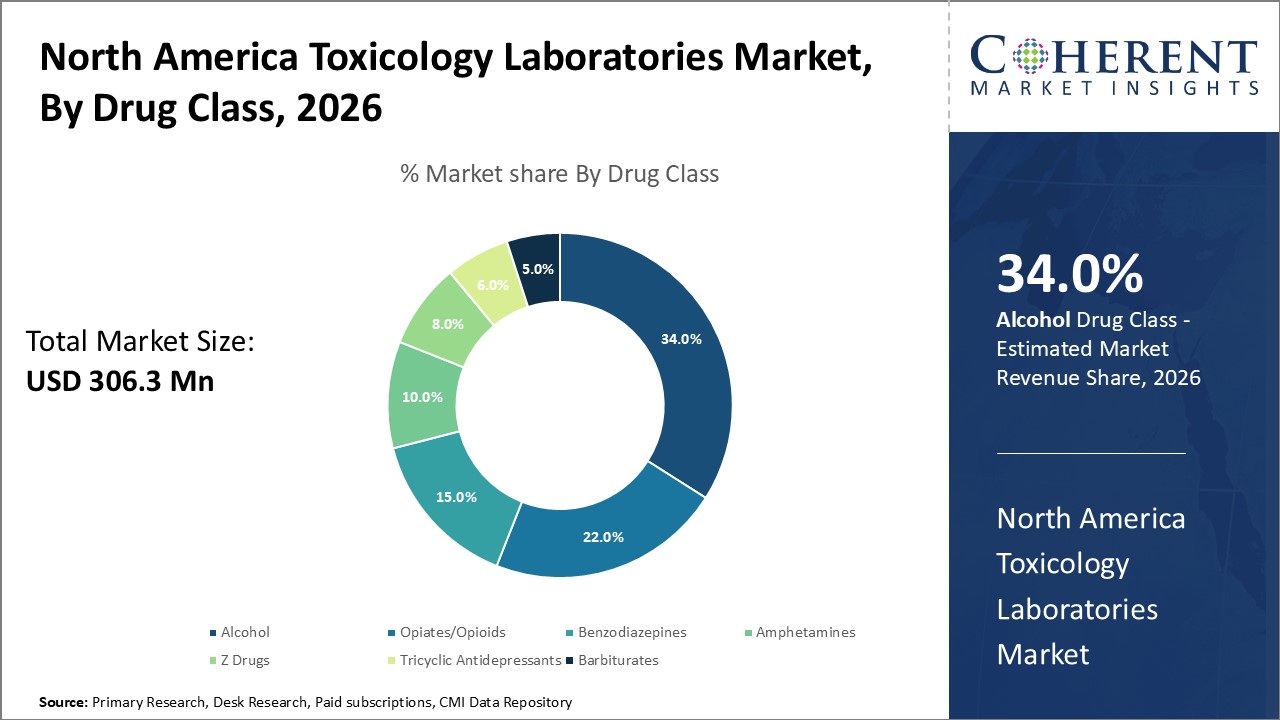
Figure 1. North America Toxicology Laboratories Market Share (%), By Drug Class, 2026
To learn more about this report, Request Free Sample
North America Toxicology Laboratories Market – Driver
Increasing Awareness about Conventional Toxicology Testing Devices
Increasing awareness about conventional toxicology testing devices is expected to drive the North America toxicology laboratories market growth over the forecast period. There is increase in demand for conventional toxicology testing devices in the U.S. and Canada, owing to large consumption of controlled substances such as opioids, depressants, etc. in these countries. Most of the publically funded toxicology laboratories (forensic laboratories) are dependent on conventional testing equipment, due to the high performance offered by these devices at a minimal cost. Toxicology laboratories in hospitals and diagnostic testing centres rely on conventional technologies, due to the cost benefits.
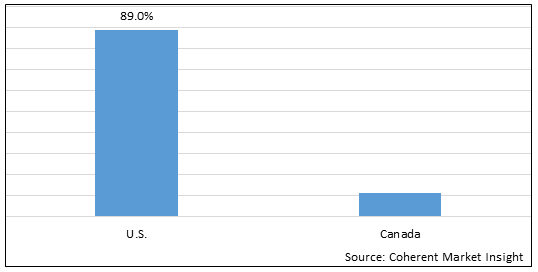
Figure 2. North America Toxicology Laboratories Market Value (US$ Million), By Country, 2026
To learn more about this report, Request Free Sample
North America Toxicology Laboratories Market- Country Analysis
Among country, the U.S. is estimated to hold a dominant position in the North America toxicology laboratories market over the forecast period due to the presence of established market players and the market players are focusing on research and development of new drugs, and increasing clinical trials in the U.S In January 2022, according to Regulatory Affairs Professionals Society (RAPS), 50 novel drugs were approved by Center for Drug Evaluation and Research (CDER) in 2021, among which 38 drugs were approved in the U.S.
Since the virus outbreak in December 2019, the disease has spread to over 100 countries across the globe, and the World Health Organization declared it a public health emergency on January 30, 2020.
North America Toxicology Laboratories Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 306.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.3% | 2033 Value Projection: | USD 411.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
InSphero AG, The Charles Stark Draper Laboratory, Inc., Bristol-Myers Squibb Company, Thermo Fisher Scientific Inc., Agilent Technologies, Inc., Bio Rad Laboratories Inc., Eurofins Scientific, Neogen Corporation, Enzo Life Sciences, Inc., and Vala Sciences Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
North America Toxicology Laboratories- Market Segmentation
North America toxicology laboratories market is segmented into drug class, product type, sample type, end user, and country.
Based on Drug Class, the market is segmented into Alcohol, Amphetamines, Barbiturates, Benzodiazepines, Opiates/Opioids, Tricyclic Antidepressants, and Z Drugs. Out of which, Alcohol segment is expected to hold a dominant position in the North America toxicology laboratories market during the forecast period, owing to research and development (R&D) activities.
Based on Product Type, the market is segmented into Instruments (Immunochemistry Analyzers, Polymerase Chain Reaction (PCR) Machine, Gas Chromatography (GC)-Mass Spectrometry (MS), and High Performance Liquid Chromatography(HPLC) and Reagents & Kits (Enzyme Linked Immunosorbent Assay (ELISA), PCR, Enzyme Multiplied Immunoassay Technique (EMIT), and Radio Immuno-sorbent Assay (RIA). Out of which, the Gas Chromatography (GC)-Mass Spectrometry (MS) segment is expected to hold a dominant position in the North America toxicology laboratories market during the forecast period, owing to increasing technological advancements.
Based on Sample Type, the market is segmented into Urine, Blood, Hair, and Oral Fluids. Out of which, the urine segment is expected to hold a dominant position in the North America toxicology laboratories market during the forecast period, owing to rising demand for toxicological drug screening.
Based on End User, the market is segmented into Hospitals and Trauma Centers, Forensic Laboratories, Diagnostic Laboratories, and Rehabilitation Centers. Out of which, the hospitals and trauma centres segment is expected to dominate the market over the forecast period, owing to e increase in the number of patient admissions in the hospitals and trauma centers.
Among all segmentation, the product type segment has the highest potential due to increasing product launches over the forecast period. For instance, in June 2022, PerkinElmer, Inc., a global leader focused on innovation for a healthier world, launched GC 2400 Platform, an advanced, automated gas chromatography (GC), headspace sampler and GC/mass spectrometry (GC/MS) solution designed to help laboratory teams in simplifying lab operations, driving exact outcomes, and performing more flexible monitoring.
North America Toxicology Laboratories Market- Key Developments
In January 2020, Agilent Technologies Inc., announced, Thermo Fisher Chromeleon or Agilent OpenLab CDS are the most recent applications of its mutual compatibility philosophy, allowing joint consumers of Agilent Technologies and Thermo Fisher Scientific to control analytical instruments from both companies.
In June 2022, Thermo Fisher Scientific Inc., launched a new liquid chromatography-mass spectrometry (LC-MS) system for forensic toxicology to help forensic toxicologists, clinical research toxicologists, employee drug testing facilities, and wellness Organizations stay on top of new and illegal drugs.
North America Toxicology Laboratories Market- Key Trends
Increasing Expansion Of Toxicology Serives
Key market players are focusing on expanding their toxicology services by adopting growth strategies such as acquisition, in order to support drug-development programs beyond Investigational New Drug (IND) and into Developmental and Reproductive Toxicology (DART) and carcinogenicity studies. For instance, in January 2022, Frontage Laboratories, Inc., a pharmaceutical laboratory, announced the acquisition of Experimur LLC, a laboratory and its affiliate Experimur Properties LLC. This acquisition will provide customers highest levels of quality, technology, and the ability to support customers' drug-development programs beyond IND and into Developmental and Reproductive Toxicology (DART) and carcinogenicity studies.
North America Toxicology Laboratories Market- Restraint
Introduction of Point-Of-Care Toxicology Testing Devices
Introduction of point-of-care toxicology testing devices is expected to hamper the market growth. Increasing abusive or hazardous consumption of controlled substances leads to high demand for handheld toxicology testing devices. Thus, companies are focusing on developing point-of-care toxicology testing devices. Commercialization of point-of-care toxicology The market share of traditional toxicology laboratories would eventually be taken over by testing devices, which could have a negative effect on the market's expansion. For instance, in April 2019, Neogen Corporation, launched a portable version of its Raptor Integrated Analysis Platform that offers the option of performing mycotoxin testing in the laboratory or wherever a tester may choose.
North America Toxicology Laboratories Market - Key Players
Major players operating in the North America toxicology laboratories market include InSphero AG, The Charles Stark Draper Laboratory, Inc., Bristol-Myers Squibb Company, Thermo Fisher Scientific Inc., Agilent Technologies, Inc., Bio Rad Laboratories Inc.
Definition: Toxicology is the scientific study of adverse effects that occur in living organisms due to chemicals. Toxicology laboratories are specialized labs that test for multiple forms of toxicity. It involves observing and reporting symptoms that arise after exposure to toxic substances. The substances that are assessed by toxicologists includes environmental agents and chemical compounds found in nature, as well as pharmaceutical compounds that are synthesized for medical use by humans.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients