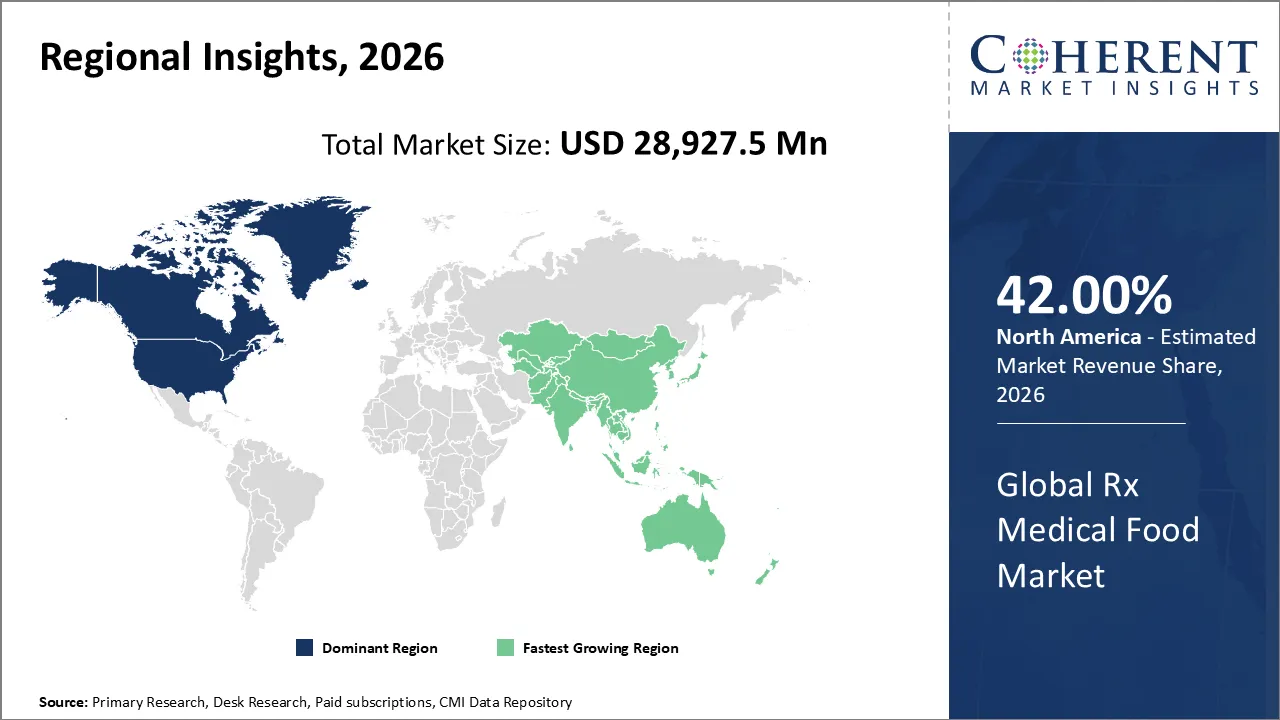
Rx medical food market is estimated to be valued at USD 28,927.5 Mn in 2026 and is expected to reach USD 46,711.8 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.1% from 2026 to 2033.
Medical food is consumed or internally administered under the supervision of a physician for the treatment of a disease or specific dietary management. Medical food is used for various purposes such as healthy aging, maintaining brain health, and for treating chronic medical conditions such as irritable bowel syndrome and metabolic disorders. Medical food imitates therapeutic agents used in the treatment of clinical conditions such as pain, sleep disorders, and cognitive disorders. It provides relief from pain or helps treat the condition without exhibiting any risk or side effects that are usually associated with pharmaceutical drugs. Medical food products claiming treatment of such medical conditions or any other diseases are required to support their claim with proper scientific and clinical study results. Medical food is a category of prescribed products which offer various advantages over prescription drugs such as lesser side effects, lower product costs, and nutritional benefits.
|
Current Event |
Description and its Impact |
|
Global Regulatory Harmonization and FDA Policy Changes |
|
|
Healthcare System Transformation and Reimbursement Evolution |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the pills segment is expected to lead the market with 42.7% share in 2026, due to their convenience, have a precise dose, and are preferred by many doctors. Patients trust pill formulations for long-term conditions, which makes sure they take them and that they work. Compared to powders, liquids, or chewable, they are the easiest to get as they are portable, stable, and well-known.
In terms of therapeutic application, the neurological disorders segment is expected to hold 33.2% share of the market in 2026, owing to the rise in cases of Alzheimer's, Parkinson's, and cognitive decline. There are increasing number of prescriptions for medical foods that are good for brain health, due to massive investments in research and development. The world's aging population is driving up demand, making neuro-nutrition the most important area of medicine, even more so than pain or metabolic disorders.
In terms of distribution Channel, the retail pharmacies segment is projected to account for 68.4% share in 2026, making the majority of the sales. They are the best way to buy as they are easy to find, build trust, and are based on prescriptions. Patients depend on physical outlets for advice and compliance, and regulatory oversight makes their role even stronger. Online pharmacies are growing steadily, but they are still not as popular as brick-and-mortar stores.
To learn more about this report, Request Free Sample
North America is expected to dominant the Rx medical food market with 42% share in 2026, due to chronic diseases are increasingly prevalent, strong physician awareness, reimbursement policies are getting stronger, and healthcare infrastructure is getting better. Aging populations, neurological and metabolic disorders, and people's preference for clinically proven nutrition are all factors that drive adoption and keep its dominant market share in the region.
For instance, in October 2025, to address malnutrition, Clemson Rural Health started the Protein Rx program in Upstate South Carolina. This is the first program of its kind to include protein supplementation as part of medical care, bringing nutrition into the delivery of healthcare. It connects directly to Rx medical food, which meets dietary needs through prescribed nutrition interventions, unlike retail pharmacy expansions.
Asia Pacific is expected to exhibit the fastest growth, due to more individuals are getting chronic diseases, the middle class is growing, and healthcare costs are rising. Growing awareness of clinical nutrition, cities are growing quickly, and the governments of China and India are helping people adopt it. The fastest growth is driven by a strong need for managing neurological and metabolic disorders.
The U.S. Rx Medical Food market is strong in 2026 as there are many cases of chronic diseases, doctors are good at prescribing them, and there are good reimbursement systems in place. The country's leading market position is strengthened by an aging population, rising neurological and metabolic disorders, advanced clinical research, and consumers' preference for evidence-based nutrition.
For instance, in December 2025, to combat food insecurity and chronic disease, VCU Health in Richmond, Virginia started a program called Filling a Prescription for Nutrition. As part of their care, patients get groceries that are good for their health and counseling. This program combines nutrition with treatment, which is in aligned with the Rx medical food concept, which claims that food can be used as medicine to improve health.
China's Rx Medical Food market continues to be competitive in 2026 due to its large population, the rising number of people with diabetes and neurological disorders, and the growing spending on healthcare by the middle class. China is the biggest and most important market in Asia-Pacific owing to government support for clinical nutrition, rapid urbanization, and doctors' growing awareness of the issue.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 28,927.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.1% | 2033 Value Projection: | USD 46,711.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Danone S.A., Mead Johnson & Company, LLC, Fresenius Kabi AG, Nestle S.A., Primus Pharmaceuticals, Inc., Targeted Medical Pharma Inc., Pivotal Therapeutics Inc., VAYA Pharma, Inc., Lanfam and AlfaSigma S.P.A. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rise in chronic diseases around the world, like diabetes, neurological disorders, gastrointestinal conditions, and metabolic syndromes, is driving the use of medical foods. Medical foods are different from regular diets or supplements because they are made to meet the nutritional needs of patients where regular nutrition fails. The Rx medical food market share is growing quickly due to a growing number of healthcare providers are prescribing these products as part of integrated treatment plans for long-term disease management.
For instance, in June 2025, to help diabetic patients, About Fresh and Neighborhood Healthcare started the Fresh Connect produce prescription pilot in Southern California. The program gives out prescriptions for fresh fruits and vegetables in order to improve diet quality, blood sugar control, and health in general. This program is a good example of the Rx medical food approach, which uses nutrition as a form of therapy.
As the number of senior citizens around the world grows, so do health problems that come with age, like malnutrition, dementia, and muscle loss. Older adults are being given medical foods to help their brain health, keep their metabolism in balance, and addressing any nutritional deficiencies. This change in demographics is making the Rx medical food market growth extremely lucrative, as older people need special nutrition to deal with complicated health problems. The rising need for physician-supervised dietary interventions positions medical foods as a critical component of geriatric healthcare.
The Rx medical food industry is putting a growing amount of effort into making disease-specific formulations to help with neurological disorders, diabetes, gastrointestinal diseases, and malnutrition related to cancer. These specialized products help with nutrition where regular diets don't work, and they can be used for therapeutic purposes under the supervision of a doctor. Companies can reach more people and build trust with healthcare providers by making solutions that are specific to different patient groups. This targeted approach is likely to have significant impacts on the Rx medical food market forecast, leading to steady growth in a wide range of therapeutic areas.
The Rx medical food market is growing due to chronic and metabolic disorders are becoming more widespread and there is more focus on nutritional therapies that are specific to managing these diseases. In 2024, worldwide sales of prescription medical food products reached over 187 million units. Pills and powders accounted for the largest parts of this market, showing that doctors prefer formats that are easy to use and give the right dose. Pills made up the majority of prescribed units, especially for neurological and metabolic conditions. Powder formulations were also commonly used for gastrointestinal and metabolic support. People who have trouble swallowing or who need certain nutrients have started to like liquids and other forms like chewable and bars.
North America is still a leading region for Rx medical food adoption as it has well-established healthcare systems and strong distribution networks for outpatient and hospital pharmacies. Medical foods are included in both short-term and long-term dietary management plans due to their can be used to treat a wide range of conditions, from pain and neurological disorders to metabolic and GI problems. In markets like Asia-Pacific, increasing numbers of individuals are using clinical nutrition as individuals are becoming more aware of it and the rules are changing.
The oral route, which includes pills and powders, is still the most common way to give medication since it is easy to use and patients are more likely to follow the instructions. However, liquid forms are becoming more popular in pediatric and geriatric care settings. Technological advances in formulation, improved nutrient stability, and tailored therapeutic outcomes are broadening the scope of Rx medical food applications, solidifying its strategic importance within comprehensive nutritional and clinical care pathways.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients