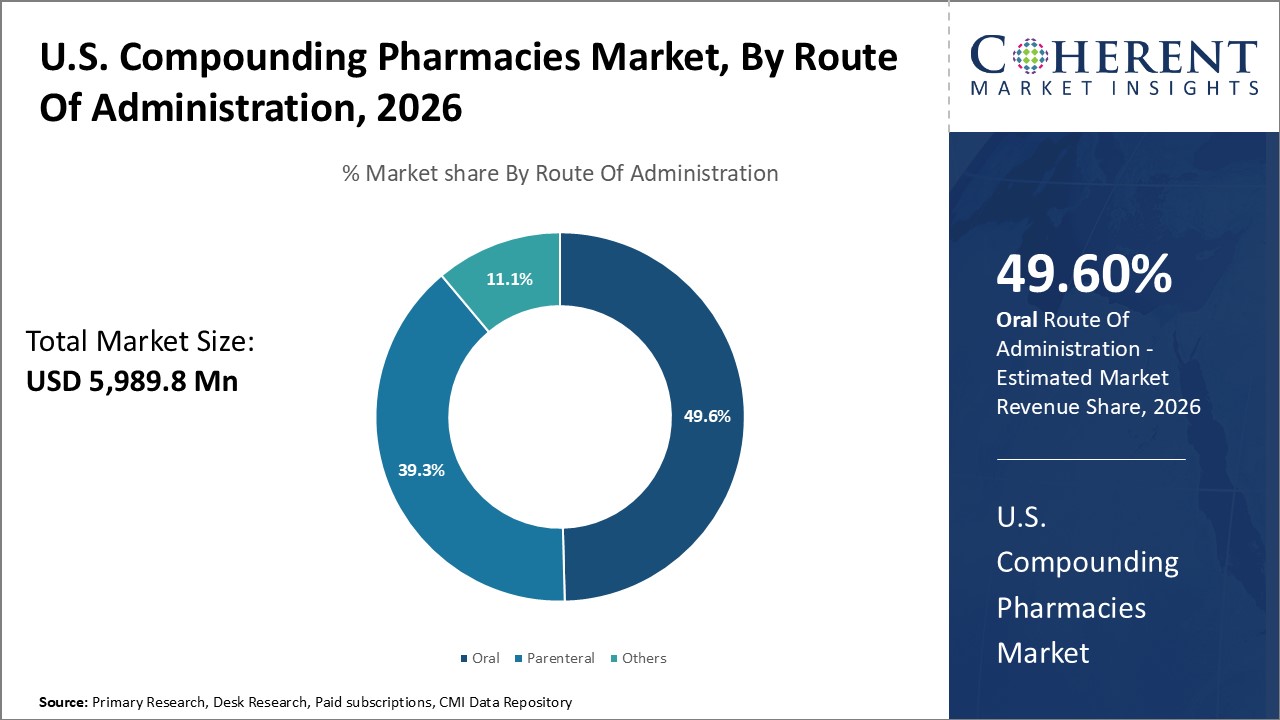
The U.S. compounding pharmacies market is estimated to be valued at USD 5,989.8 Mn in 2026 and is expected to reach USD 9,308.0 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.5% from 2026 to 2033.
To learn more about this report, Request Free Sample
The compounding pharmacies market demand is poised for positive growth over the forecast period, driven by increasing demand for personalized medicines, a rise in chronic diseases, and a growing geriatric population. The need for compounded drugs tailored to specific doses, delivery methods, or ingredient combinations presents significant opportunities for market players.
|
Current Event |
Description and Its Impact |
|
Technological Advancements and Digital Integration |
|
|
Public Health and Epidemiological Factors |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Automation in compounding pharmacies is transforming the way personalized medications are prepared, improving safety, efficiency, and accuracy. Traditionally manual and time-consuming, compounding is now enhanced by robotic systems, software, and precision tools that standardize processes and reduce human error.
In May 2025, Finnish healthtech company CurifyLabs raised €6.7 million, bringing its total funding to €17.6 million, to advance the automation of compounded medication production. Compounded drugs are essential when standard medications aren't suitable due to factors like dosage needs, allergies, or shortages. CurifyLabs’ Compounding System Solution integrates excipient bases, active ingredients, hardware, software, and quality control to automate personalized drug manufacturing. Its desktop-sized Pharma Printer mixes high-quality excipient bases with active ingredients—from raw materials or crushed tablets—and dispenses them in tailored forms such as tablets or liquids.
In terms of the therapeutics area, the hormone replacement therapy segment is expected to hold 33.9% of the market share in 2026, owing to the high incidence of hormonal imbalances among the U.S. population. As people age, it is common for hormone levels in the body to fluctuate or decline. This can cause a wide variety of uncomfortable and unpleasant symptoms. Issues like hot flashes, night sweats, mood swings, decreased libido, vaginal dryness in women, and other signs of hormonal decline are quite common in middle-aged and older individuals. The stress of modern lifestyles also takes a toll on hormone balance in many individuals. This growing need to mitigate symptoms of hormonal imbalance and maintain quality of life has driven the demand for compounded hormone replacement therapy medications.
For instance, recent advancements in hormone replacement therapy (HRT) are poised to revolutionize care for individuals with hormone deficiencies. Innovative approaches, including the use of hormone-filled microbeads in hydrogels, offer steady, extended release after monthly self-injections. These developments aim to provide patients with more convenient and effective treatment options, potentially improving outcomes and quality of life.
To learn more about this report, Request Free Sample
In terms of route of administration, the oral segment is expected to hold 49.6% of the market share in 2026 due to the popularity and convenience of oral medications for pain management. Pain affects a significant proportion of the U.S. population at some point. From minor headaches to severe chronic conditions like arthritis or back pain, effective pain relief is highly sought after. While conventional oral painkillers are sufficient for some, many individuals require customized medications to truly get their pain under control. Compounded drugs allow for tailoring of drug types, strengths, and formulations according to an individual's unique needs, biochemistry, and tolerance levels. This has significantly boosted the demand for compounded oral medications for effective pain management.
For instance, in March 2025, the U.S. Food and Drug Administration (FDA) approved Arbli™ (losartan potassium) Oral Suspension, a 10 mg/mL formulation developed by Scienture Holdings, Inc. This marks the first ready-to-use liquid version of losartan in the U.S., providing a standardized alternative to compounded preparations. Arbli is indicated for treating hypertension in patients over six years old, reducing stroke risk in those with hypertension and left ventricular hypertrophy, and managing diabetic nephropathy in certain type 2 diabetes patients.
In terms of application, the human segment is expected to hold 57.7% of the market share in 2026 due to the growing demand for individualized healthcare. Modern medical practice places great emphasis on personalized care and treatment according to an individual's specific conditions, genetics, and other factors. However, not all conventional medications are suitable or optimally effective for all patients. Compounded medications offer a solution, allowing the formulation of customized drugs and site-specific therapies based on human physiology. They also help address issues like allergies to inactive ingredients or the need for specific dosage forms not commercially available. This expanding need for individualized clinical care has fueled growth in the demand for human-oriented compounded medications and has driven major market share for the human segment.
For instance, in July 2025, Texas Star Pharmacy, under the leadership of Raman Bhaumik, launched a new provider portal aimed at simplifying the ordering and compliance processes for compounded prescriptions. The platform offers enhanced features for secure ordering, real-time tracking, and streamlined documentation, ensuring adherence to regulatory standards. Designed with input from healthcare providers, the portal aims to reduce administrative burdens and improve patient care by providing a more efficient and transparent system for managing compounded medications.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,989.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.5% | 2033 Value Projection: | USD 9,308.0 Mn |
| Segments covered: |
|
||
| Companies covered: |
Avella Specialty Pharmacy,Ltd, Rx3 Compounding Pharmacy, B Braun SE, QuVa Pharma, ITC Compounding Pharmacy, PharMEDium Services, Cantrell Drug Company, Triangle Compounding Pharmacies, Hoye’s Pharmacy, Vertisis Custom Pharmacy, Smith Caldwell Drug Store, Nephron Pharmaceuticals Corporation, Clinigen Group PLC, Lorraine’s Pharmacy, and Precision Compounding Pharmacy |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
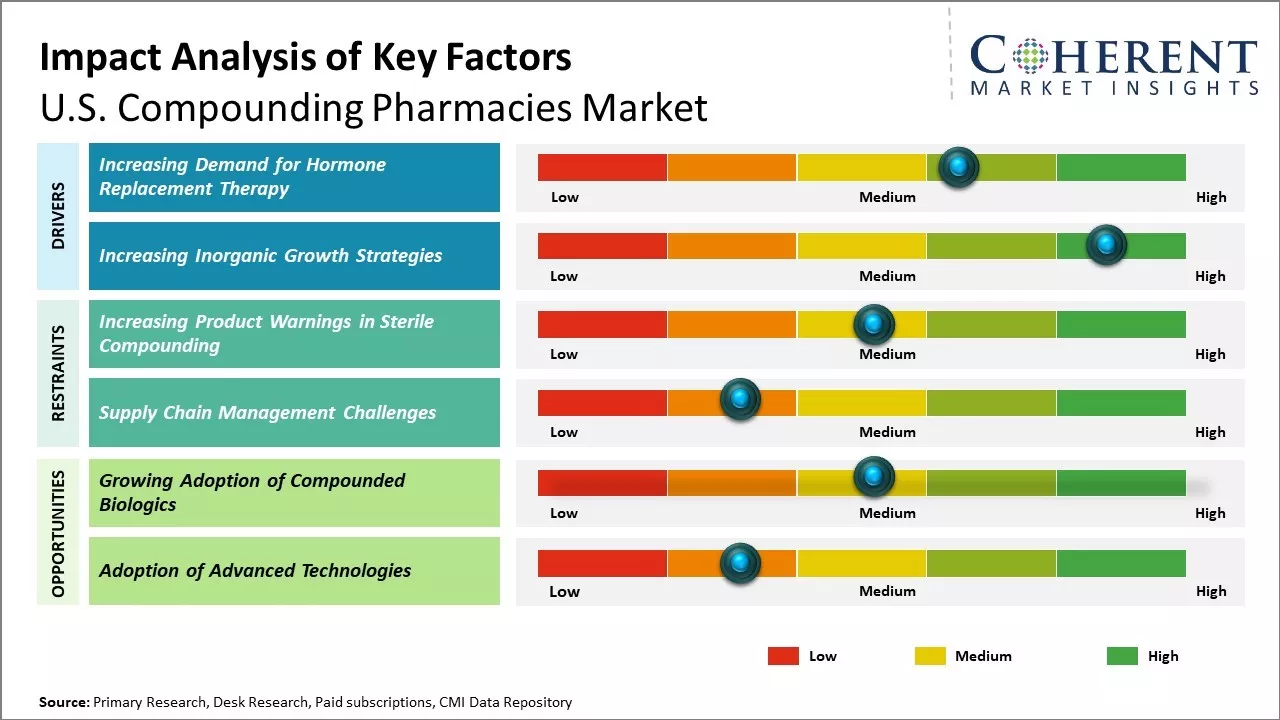
The U.S. compounding pharmacies market is growing due to rising demand for hormone replacement therapy (HRT). Compounding pharmacies provide personalized HRT formulations tailored to individual needs, including various hormone types and delivery methods. Increased awareness of HRT benefits among the aging population and hormone-related health issues is driving more patients toward customized treatments, boosting the market demand for compounded hormone therapies. For instance, in July 2025, Dr. Jay Bhaumik spearheaded the launch of bio-identical hormone pellets at Texas Star Pharmacy, aiming to enhance hormone replacement therapy (HRT) with improved convenience and consistency. These pellets, designed to deliver a steady dose of hormones, offer patients a more reliable and user-friendly alternative to traditional HRT methods.
In the U.S. compounding pharmacies market, inorganic growth strategies such as mergers, acquisitions, and partnerships are crucial for rapid expansion and competitive advantage. These moves help companies broaden product offerings, enter new regions, and access advanced technologies. Acquiring smaller pharmacies and collaborating with healthcare providers improve market share, regulatory compliance, and customized formulation development. Overall, inorganic growth supports innovation and strengthens companies’ positions amid rising demand for personalized medicines.
For instance, in April 2024, Myonex, a global clinical trial supply company, announced that it had acquired Saveway Compounding Pharmacy, a compounding pharmacy in Delaware, U.S. The acquisition helped Myonex to better supply patients in clinical trials with medication and equipment as needed. It will also help the company expand its clinical trial designs and include direct-to-patient and home healthcare provider capabilities.
The growing adoption of compounded biologics presents a significant opportunity for the U.S. compounding pharmacies market forecast. As biologic drugs become more prevalent in treating complex medical conditions like cancer and chronic diseases, the demand for customized medication alternatives is growing. Compounding pharmacies allow for specialized biologics to be created to better suit individual patient needs. This is important as not all U.S. Food and Drug Administration-approved drugs work uniformly across different genetic profiles and clinical factors in each person. As biologics continue to revolutionize the treatment of serious diseases, compounded versions provide an important avenue for optimizing patient care through personalization. This emerging opportunity is well-positioned to support significant expansion of the U.S. compounding pharmacies market in the coming years as the chronic illness burden escalates across communities. Catering to this specialized niche with alternatives for hard-to-treat populations can prove integral to the long-term growth trajectory of the compounding pharmacies market.
The U.S. Compounding Pharmacies Market value is positioned at a critical juncture characterized by evolving regulatory scrutiny and increasing demand for personalized therapeutics. Notably, the market’s trajectory is being shaped less by broad pharmaceutical trends and more by nuanced shifts in patient-specific care paradigms and regulatory enforcement dynamics. The heightened focus on individualized medicine, evident through the rising prevalence of chronic diseases and complex comorbidities—underscores the indispensable role of compounding pharmacies in delivering tailored formulations that standard drugs cannot adequately address.
From a regulatory perspective, the recent intensification of FDA oversight has prompted compounding pharmacies to enhance their quality assurance and compliance infrastructures. This regulatory tightening, while posing operational challenges, serves as a catalyst for elevating industry standards and fostering greater trust among healthcare providers and patients. For instance, the implementation of stricter USP <797> and USP <800> guidelines have compelled pharmacies to invest substantially in sterile compounding facilities and personnel training, a necessary evolution that aligns with patient safety imperatives.
Moreover, compounding pharmacies that have successfully integrated advanced automation and digital tracking systems report measurable improvements in production accuracy and traceability. These technological adoptions not only mitigate risks associated with manual compounding errors but also streamline workflows, resulting in enhanced capacity to meet the growing demand for customized medications, particularly in oncology, dermatology, and pediatrics.
The market’s resilience is further evidenced by the expanding geriatric demographic, which increasingly requires medications adjusted for polypharmacy and age-specific pharmacokinetics. Leading compounding providers catering to this segment have reported up to a 25% increase in demand for hormone replacement therapies and pain management formulations tailored to elderly patients.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients