Tissue Glue And Bio Adhesive Sealants Market is estimated to be valued at USD 2,541.2 Mn in 2026 and is expected to reach USD 4,440.5 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033.
Analysts’ Views on Global Tissue Glue And Bio Adhesive Sealants Market:
The global tissue glue and bio adhesive market's growth can be hindered by the presence of a variety of traditional sealants, and sutures. The increasing prevalence of bone-related disorders is expected to drive the global tissue glue and bio-adhesive sealants market over the forecast period. For instance, according to an article published by the National Library Of Medicine, an estimated 1.5 million individuals suffer a fracture caused by bone disease annually. In fact, fractures are the most common musculoskeletal condition requiring hospitalization among Medicare enrollees (who are age 65 and older). Approximately one-third (500,000) of fracture patients are hospitalized), while many more suffer fractures that do not require or result in hospitalization.
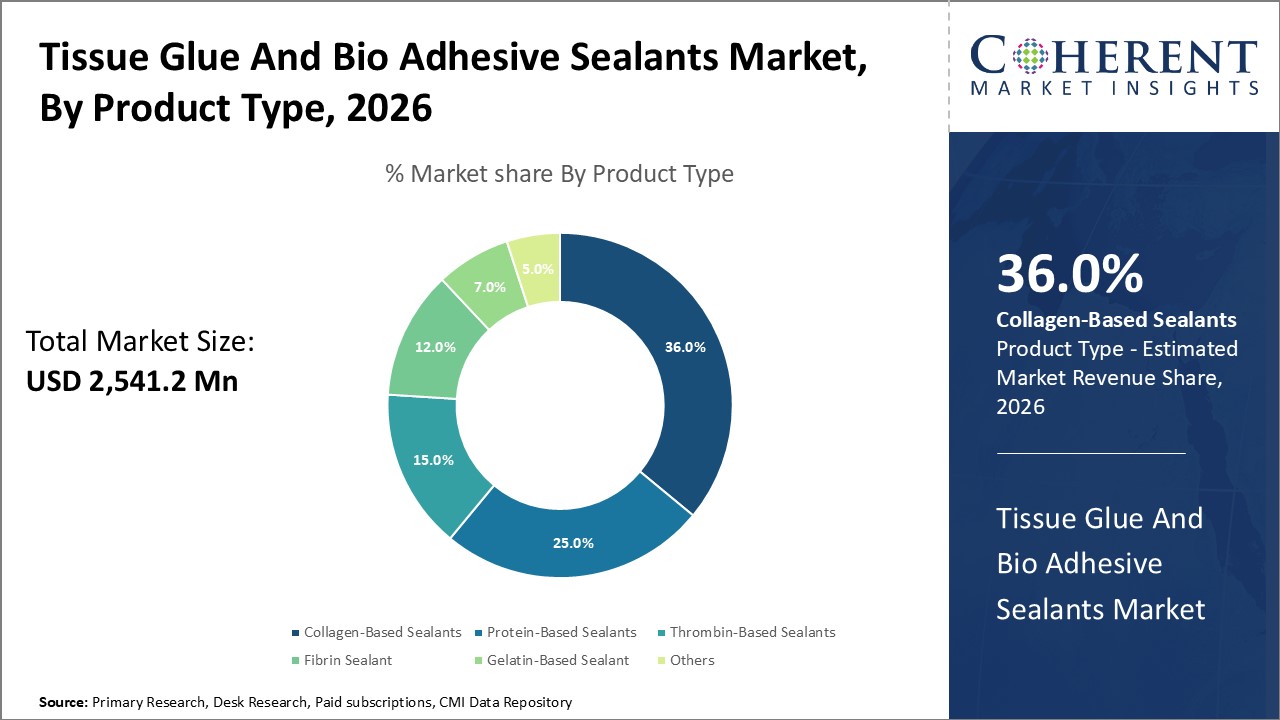
Figure 1. Global Tissue Glue And Bio Adhesive Sealants Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Tissue Glue And Bio Adhesive Sealants Market – Driver
Increasing prevalence of bone-related disorders
Increasing prevalence of bone-related disorders is expected to propel the growth of the global tissue glue and bio-adhesive sealants market over the forecast period. The prevalence of bone diseases is going to increase significantly as the population ages. In the U.S., the number of people aged 65 and older is expected to rise from 35 to 86 million between 2000 and 2050, while the number of people aged 85 and older will increase from 4 to 20 million.
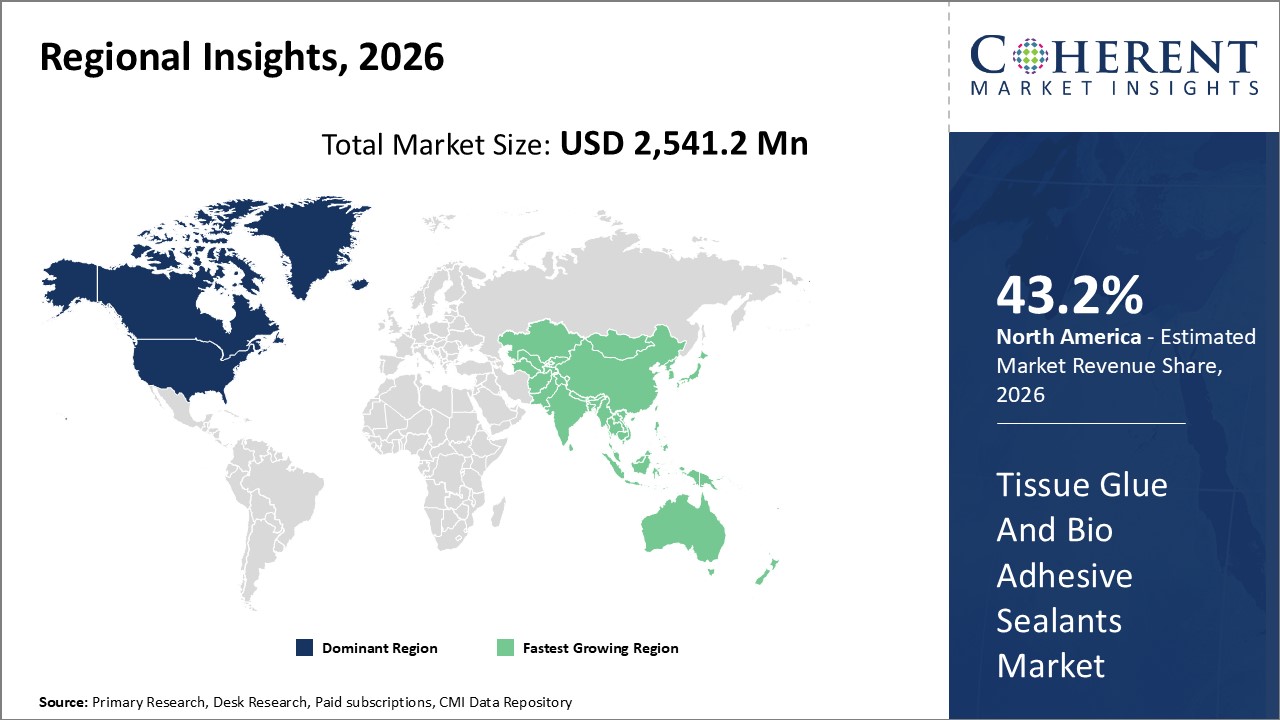
Figure 2. Global Tissue Glue and Bio-adhesive Sealants Market Value (US$ 2,007.6 Million), by Region, 2026
To learn more about this report, Request Free Sample
Global Tissue Glue and Bio-adhesive Sealants Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global tissue glue and bio-adhesive sealants market over the forecast period. North America holds 43.2% % of the market share due to the increasing prevalence of hospitals, surgical centers, and medical associations in the region. For instance, in 2020, according to the Centre for Medicare & Medicaid Services (CMS), the U.S. government made a national expenditure of around US$ 4.1 trillion in 2020.
Tissue glue and bio-adhesive sealants have major applications in hospitals, surgeries centers , and clinics. Moreover, various surgery centers, hospital units, and wound care and treatment services were delayed. The hospitals saw a shortage of beds, care professionals, medical equipment, and others. For instance, in 2020, according to the United States Census Bureau, the revenues of medical and diagnostic laboratories saw a fall of 10.9% in the 2nd quarter of 2020.
Tissue Glue And Bio Adhesive Sealants Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,541.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 4,440.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cryolife, Baxter International Inc., Luna Innovations Incorporated, B. Braun Melsungen AG, Johnson and Johnson, Dentsply Sirona Inc., Cohera Medical, Inc., Chemence Medical, Inc., Tissuemed Ltd., C.R. Bard Inc., and Integra LifeSciences Corporation. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Tissue Glue and Bio-adhesive Sealants Market Segmentation:
The global tissue glue and bio-adhesive sealants market report is segmented into product type, application, end user, and region
Based on Product Type, the market is segmented into protein based sealants, collagen based sealants, thrombin-based sealants, fibrin sealant, gelatin-based sealant, and others. Out of which, the collagen based sealants segment is expected to hold a dominant position in the global tissue glue and bio-adhesive sealants market during the forecast period and this is attributed because they are widely accepted and help to reduce the scars and it is also used for the post-operative infections.
Based on Application, the market is segmented into cardiovascular surgery, laparoscopic surgeries, burns and skin grafting, digestive, cosmetic, reconstructive and hand surgery, general surgeries, and others. Out of which, the cosmetic, reconstructive and hand surgery segment is expected to hold a dominant position in the tissue glue and bio-adhesive sealants market during the forecast period and this is attributed to the increasing usage of tissue adhesives in cosmetic surgeries
Based on End User, the market is segmented into hospitals, specialty clinics, and ambulatory surgical centers (ASCs). Out of which, the hospital segment is expected to dominate the market over the forecast period and this is attributed to the increasing number of surgeries and treatment procedures.
Among all segmentation, the product type segment has the highest potential due to the increasing prevalence of collagen based sealants over the forecast period. For instance, according to the data published by the MDPI, FloSeal and Proceed are two collagen-based adhesives approved for use in the U.S., and they are made from a combination of bovine collagen and bovine thrombin and are also relatively inexpensive
Global Tissue Glue and Bio-adhesive Sealants Market Cross Sectional Analysis:
In product type segment, collagen based sealants hold a dominant segment in North America region due to the risk of a fracture increasing with age. For instance, in 2020, according to an article published in the National Library of Medicine, the risk of a fracture increases with age and is greatest in women. Roughly 4 in 10 white women age 50 or older in the U.S. will experience a hip, spine, or wrist fracture sometime during the remainder of their lives. Looking ahead, the lifetime risk of fractures will increase for all ethnic groups as people live longer. Osteoporosis is the most common cause of fractures. Roughly 10 million individuals over age 50 in the U.S. have osteoporosis of the hip. An additional 33.6 million individuals over age 50 have low bone mass or “osteopenia” of the hip and thus are at risk of osteoporosis and its potential complications later in life.
Global Tissue Glue and Bio-adhesive Sealants Market: Key Developments
Global Tissue Glue and Bio-adhesive Sealants Market: Key Trends
Increasing adoption of inorganic activities by market players
Increasing adoption of inorganic activities by market players such as acquisition to expand the product portfolio of their manufacturer is expected to drive the global tissue glue and bio-adhesive sealants market growth over the forecast period. For instance, on October 5, 2022, Collagen Matrix, Inc. a regenerative medicine and a Linden Capital Partners portfolio company, announced the acquisition of Polyganics, B.V., a medical technology company that develops and manufactures bioresorbable medical devices. The acquisition expands Collagen Matrix’s portfolio of bioresorbable solutions and technology platforms for bone and tissue repair.
Global Tissue Glue and Bio-adhesive Sealants Market: Restraint
Lack of adoption of tissue glue and bio-adhesive sealants
The global tissue glue and bio-adhesive sealants market can be hindered by the lack of adoption of tissue glue and bio-adhesive sealants. This can be attributed to a lack of awareness regarding the benefits, clinical efficacy, safety, and cost-effectiveness of tissue glue and bio-adhesive sealants compared to substitute products or procedures and tissue repair therapies.
Global Tissue Glue and Bio-adhesive Sealants Market - Key Players
Major players operating in the global tissue glue and bio-adhesive sealants market include Cryolife, Baxter International Inc., Luna Innovations Incorporated, B. Braun Melsungen AG, Johnson and Johnson, Dentsply Sirona Inc., Cohera Medical, Inc., Chemence Medical, Inc., Tissuemed Ltd., C.R. Bard Inc., and Integra LifeSciences Corporation.
Global Tissue Glue and Bio-adhesive Sealants Market– Insights
Tissue glue is used as an alternative method for wound closure that is painless, fast, and does not require a follow-up visit for removal. Tissue glue is made from cyanoacrylate compounds. Tissue glue finds application in superficial lacerations, low wound tension, and after placement of deep sutures to close the skin.
Bio-adhesive sealants are developed from proteins, collagen, thrombin, fibrin, gelatin, and polysaccharides such as chitosan and are used for acute thoracic aortic dissection repair, controlling intraoperative bleeding in vascular procedures, and for soft tissue sealing.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients