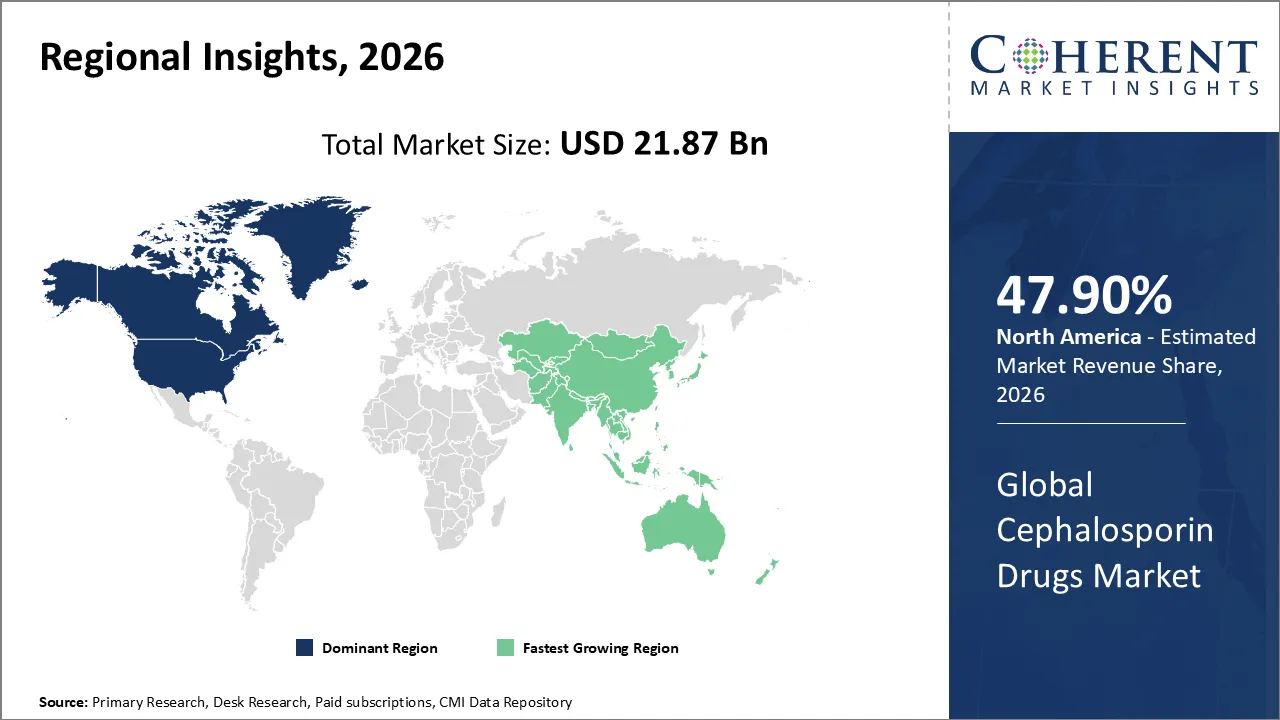
The Cephalosporin Drugs Market is estimated to be valued at USD 21.87 Bn in 2026 and is expected to reach USD 23.65 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 3.76% from 2026 to 2033.
The market for cephalosporin medications is a well-established sector of the worldwide antibiotics industry. These drugs are widely used to treat bacterial infections in both hospitals and outpatient settings. Healthcare practitioners use cephalosporins because they work against a wide range of bacteria, are safe, and are available in many generations, formulations, and ways of giving them. The market's global presence is still strong because of rising hospital demand, rising infection rates, the extensive availability of generics, and the fact that they are always included in treatment guidelines.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological and Scientific Advances |
|
|
Public Health and Epidemiological Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
First-generation Cephalosporin hold the largest market share of 34.3% in 2026. First-generation cephalosporins are still quite popular in the cephalosporin medications market because doctors use them a lot since they work well against Gram-positive bacteria and have been accepted in clinical settings for a long time. Hospitals and outpatient clinics often prescribe these medications to prevent surgery and treat simple skin and soft tissue infections, which keeps demand steady. Broad generic availability, low treatment costs, and continuing inclusion in standard treatment guidelines all help to increase uptake, especially in healthcare systems that are sensitive to costs and in emerging countries. For instance, in December 2024, Baxter International launched five injectable pharmaceuticals in the U.S., including Micafungin, Cyclophosphamide, Pantoprazole Sodium, Cefazolin, and Levetiracetam, with Cefazolin serving as a single-dose, first-generation cephalosporin for treating and preventing infections.
Generic Cephalosporin Drugs expected to hold largest market share of 53.3% in 2026. Generic cephalosporins drive the market by offering affordable, effective alternatives to branded antibiotics. Widely available in hospitals, clinics, and pharmacies—especially in emerging markets—they are frequently prescribed for bacterial infections due to their safety and cost-effectiveness. Large-scale production from India and China ensures steady supply, supporting both institutional and outpatient care.
Oral acquired the prominent market share of 52.7% in 2026. Oral cephalosporins help the cephalosporin medications industry grow by making it easier to take them outside of hospitals. Doctors provide them to people who have infections in the community so that they can finish their treatment at home and stick to it better. The fact that they work on a wide range of problems and come in different forms, such tablets and suspensions, makes people want to use them even more. Healthcare professionals and pharmacies in both established and new markets are turning to oral choices because they are cost-effective, outpatient care is growing, and people are becoming more aware of how important it is to treat infections quickly. For instance, in August 2025, Basilea Pharmaceutica has entered an exclusive licensing deal with Venatorx Pharmaceuticals to obtain global rights to ceftibuten-ledaborbactam etzadroxil, a phase 3–ready oral therapy combining a cephalosporin antibiotic for treating complicated urinary tract infections.
Skin Infection captures the largest market share of 33.9% in 2026. Healthcare providers are using cephalosporins more and more to treat bacterial skin infections such cellulitis, impetigo, and wound infections. This is driving up demand. Because they work well against common Gram-positive bacteria and are safe, they are a popular choice in hospitals and for outpatient care. More people are getting skin infections because of long-term health problems, and more people are aware of the need for early treatment, which leads to more prescriptions. The fact that oral and injectable forms are readily available makes them even more likely to be used in a wide range of healthcare settings. For instance, in November 2025, Dechra has launched Solovecin™ (cefovecin sodium) in the U.S., a long-acting injectable cephalosporin antibiotic for treating skin infections in dogs and cats.
Hospital Pharmacies hold the largest market share of 41.7% in 2026. Hospital pharmacies keep the cephalosporin pharmaceuticals industry going by always having enough antibiotics on hand for operations, emergency care, and inpatient care. Hospitals are where doctors get injectable and oral cephalosporins to treat serious infections that people get in the hospital. Hospital pharmacies become important distribution channels by managing bulk purchases, centralizing stocks, and following therapeutic recommendations. Their ability to quickly provide important medicines increases demand and makes healthcare institutions even more reliant on cephalosporins.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 47.90% in 2026. The North American cephalosporin market is growing because hospitals and outpatient clinics need more antibiotics to treat infections that don't respond to other treatments and to care for patients after surgery. Strong healthcare infrastructure, insurance coverage, and access to both brand-name and generic pharmaceuticals make it easier to use. Ongoing research, new approvals, stewardship initiatives, and more people becoming aware of bacterial infections are all helping the business grow in the region. For instance, Actavis has made AVYCAZ™ (ceftazidime-avibactam), a cephalosporin-based antibiotic, available for hospital ordering across the United States.
The market for cephalosporin medications in the Asia Pacific region is growing because more people are getting sick, healthcare is becoming more accessible, and more people are getting bacterial infections. Cephalosporins are often prescribed by hospitals and clinics to treat infections of the skin, respiratory system, and urinary tract. Affordable generics come from India and China, and government programs make healthcare easier to get, which helps the market grow even more. Patients and healthcare providers are putting more and more importance on rapid infection management and outpatient treatment. This is helping to spread the use of cephalosporin treatments across the region. For instance, in June 2024, India-developed enmetazobactam is set for local launch as part of an approved antibiotic combination with cefipime, a fourth-generation cephalosporin, for treating moderate to severe bacterial infections.
The market for cephalosporin medications in the United States is still growing because doctors are using these antibiotics more and more to treat infections that people get in hospitals and in the community. Doctors and nurses aggressively give cephalosporins to people with skin, urinary, and respiratory infections, as well as to people who are going to have surgery. Strong healthcare infrastructure, wide insurance coverage, and simple access to both brand-name and generic pharmaceuticals all make sure that people utilize them often. New drugs, ongoing research, and antibiotic stewardship programs all help doctors prescribe the right drugs. Hospitals and outpatient settings are also more likely to use these programs when people know more about how to prevent bacterial infections. For instance, in May 2025, Innoviva Specialty Therapeutics has made its novel cephalosporin antibiotic Zevtera (ceftobiprole medocaril sodium for injection) commercially available in the United States.
The market for cephalosporin medications in China keeps growing because more and more doctors are prescribing them to treat skin, urinary, and respiratory infections. Hospitals and clinics utilize both oral and injectable cephalosporins to fulfill the needs of more and more patients. Pharmaceutical companies are making more drugs, and the fact that cheap generics are widely available and the government is working to make healthcare more accessible are also helping the market develop. More and more, healthcare providers and patients are putting infection control, outpatient care, and standardized treatment protocols at the top of their lists. This is leading to more widespread use of cephalosporin medicines across the country.
The cephalosporin market is witnessing a notable shift toward generic drugs due to their affordability and comparable effectiveness to branded options. Hospitals, clinics, and retail pharmacies increasingly stock generics to reduce treatment costs and improve accessibility, especially in emerging markets. Manufacturers in India, China, and other regions are scaling production to meet demand, supporting widespread adoption. This trend enables broader patient reach, encourages competition, and drives growth while maintaining clinical efficacy in both inpatient and outpatient settings.
Injectable cephalosporins remain a key focus as hospitals rely on them for severe infections, surgical prophylaxis, and critical care. The need for rapid, reliable treatment in inpatient settings drives higher adoption of intravenous and intramuscular forms. End-users prefer injectables for complicated infections and hospital-acquired cases, while pharmaceutical companies focus on improving stability, solubility, and ease of preparation. This trend highlights the ongoing reliance on hospital infrastructure to deliver advanced cephalosporin therapies efficiently.
The rising prevalence of resistant bacterial strains presents a strong opportunity for developing next-generation cephalosporins with enhanced efficacy. Pharmaceutical companies can focus on drugs targeting multi-drug resistant Gram-negative and Gram-positive bacteria. Innovations in combination therapies, improved pharmacokinetics, and extended-spectrum formulations allow healthcare providers to manage complicated infections effectively. Introducing novel cephalosporins can strengthen clinical adoption, expand hospital and outpatient use, and differentiate products in a highly competitive market, offering significant growth potential globally.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.87 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.76% | 2033 Value Projection: | USD 23.65 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co. Inc., GlaxoSmithKline plc., Pfizer Inc., Allergan plc., Abbott Laboratories, Basilea Pharmaceutica Ltd., Sanofi S.A., F. Hoffmann-La Roche AG, Lupin Limited, Teva Pharmaceutical Industries Ltd., and B. Braun Melsungen AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients