Global Alzheimer’s drugs market is estimated to be valued at USD 6.24 Bn in 2026 and is expected to reach USD 12.72 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.7% from 2026 to 2033.
To learn more about this report, Request Free Sample
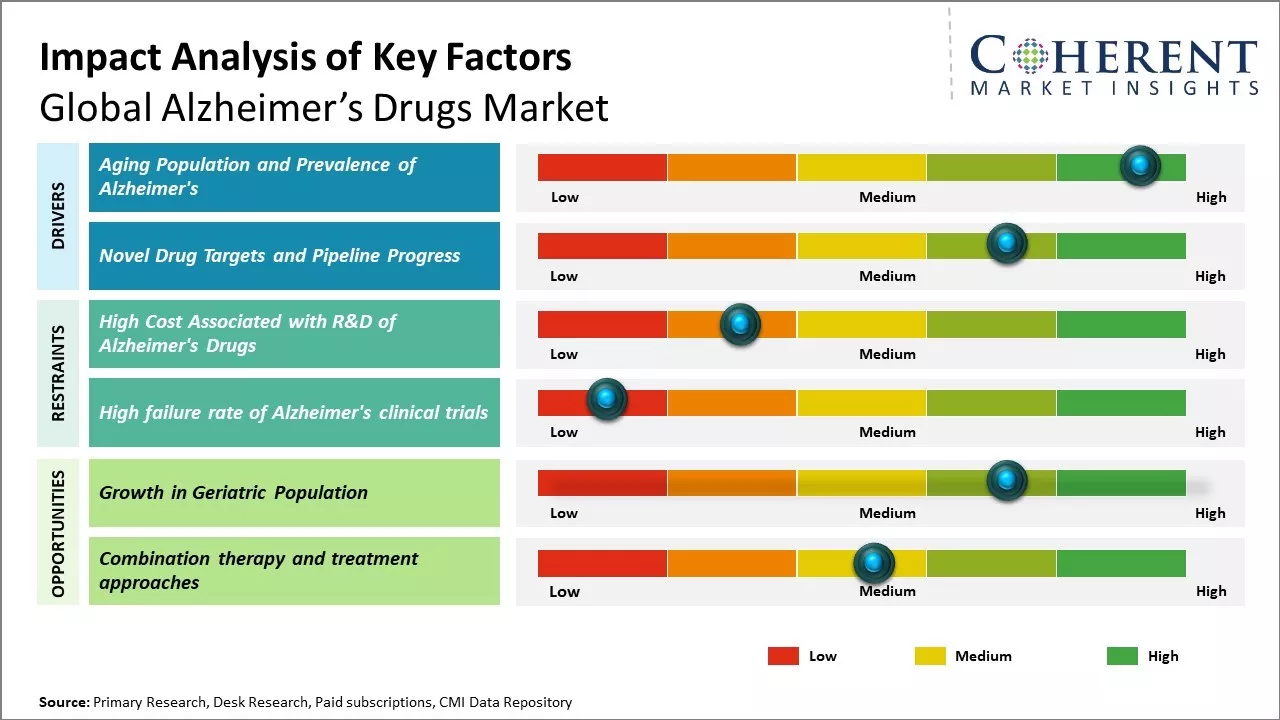
Global Alzheimer’s drugs market growth is driven by the increasing incidence and prevalence of Alzheimer’s disease worldwide due to the rising geriatric population. As age remains the most significant risk factor, countries with aging demographics are witnessing heightened healthcare needs, fueling the Alzheimer’s drugs market demand. New product launches by key players can also drive the market growth. The launch of new drugs that target the underlying causes and slow disease progression rather than just control symptoms can drive the market growth.
|
Current Event |
Description and its Impact |
|
FDA Approval of Donanemab in the U.S. (July 2024) |
|
|
DIAN-TU Gantenerumab Extension Study Results (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Reimbursement policies for Alzheimer’s drugs remain highly restrictive and vary across regions, reflecting the high costs and limited efficacy of available treatments. In the United States, Medicare—the primary payer for elderly patients has taken a cautious approach. For example, it initially limited coverage for Aduhelm to clinical trial participants due to uncertainties about clinical benefit versus cost. Similarly, coverage for Leqembi was expanded in 2023 but only under a “coverage with evidence development” model, requiring patients to be part of a registry to monitor real-world effectiveness. Japan has approved Leqembi for national health insurance reimbursement, but with strict monitoring and budget controls. A growing trend across countries is the adoption of conditional reimbursement frameworks, such as risk-sharing agreements or registry-linked coverage, where funding is tied to actual treatment outcomes. These measures aim to contain public spending while ensuring that only clinically effective drugs receive broad reimbursement.
In terms of drug class, cholinesterase inhibitors segment is estimated to contribute the highest Alzheimer’s drugs market share of 41.5% in 2026, owing to its ability to meet significant unmet needs. Cholinesterase inhibitors work by increasing levels of a chemical messenger in the brain called acetylcholine, which can help slow decline in thinking skills, memory and other functions. These are some of the first treatments approved specifically for Alzheimer's disease and remain a core therapeutic option for many patients. The mechanism of action targeting acetylcholine neurotransmission addresses a key pathological process in Alzheimer's and provides symptomatic benefit. Cholinesterase inhibitors offer oral administration convenience as compared to other treatment approaches in development. The efficacy demonstrated on cognitive and functional outcomes has led to their widespread adoption as the frontline pharmacotherapy for mild-to-moderate Alzheimer’s disease.
In August 2024, A Swedish registry study led by Karolinska Institute, published in Alzheimer’s & Dementia, evaluated 1,095 dementia with Lewy bodies (DLB) patients over a decade. Findings show that cholinesterase inhibitors (ChEIs), such as rivastigmine, donepezil and galantamine, significantly slowed cognitive decline, dropping MMSE (Mini-Mental State Examination) scores by just ‑0.39 points yearly versus ‑2.49 (memantine) and ‑2.50 (no treatment). Additionally, ChEI users faced a 34% lower risk of death in the first-year post-diagnosis (adjusted HR 0.66).
To learn more about this report, Request Free Sample
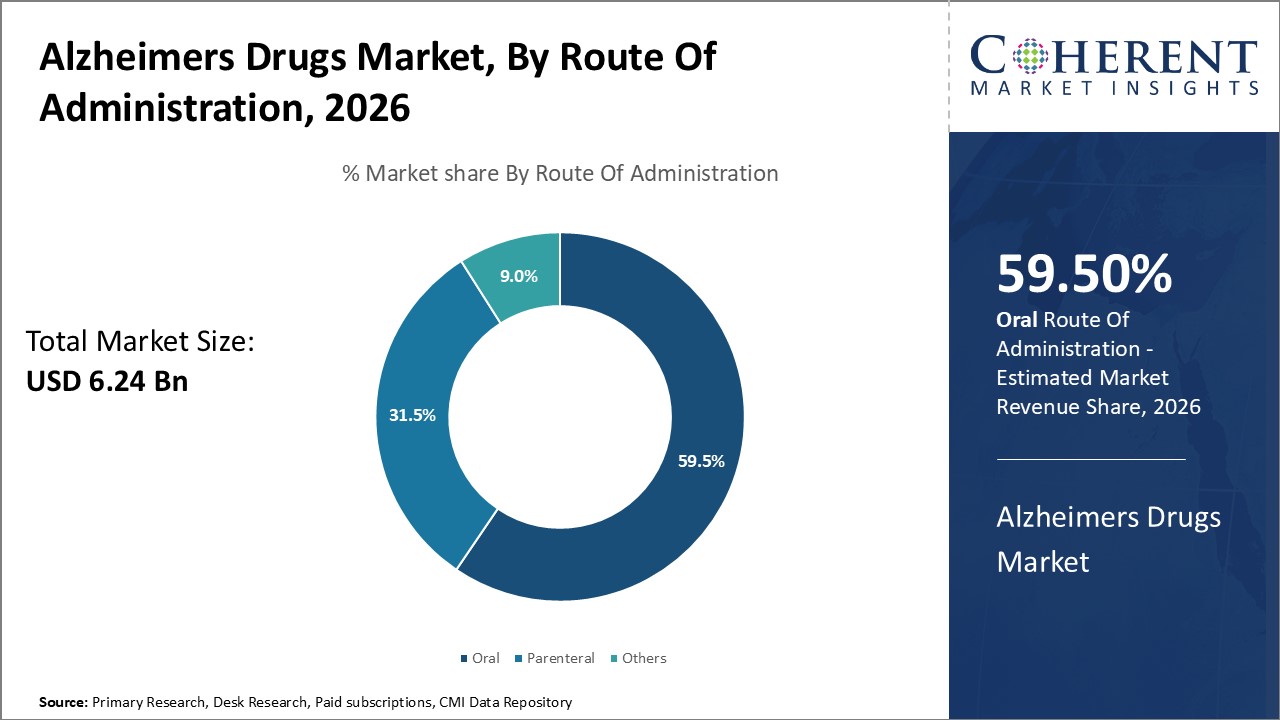
In terms of route of administration, oral segment is estimated to contribute the highest market share of 59.5% in 2026, owing to its advantage of maximizing patient adherence to treatments. For most Alzheimer's patients in the mild-to-moderate stages of the disease, oral administration of medicines is preferred as it is non-invasive and can be self-administered at home without need for clinic visits. The convenience of oral dosing encourages compliance with the treatment regimen over an extended period of time that may span years. This is critically important for Alzheimer's where continuous management is required as symptoms progress. Administering medicines orally also avoids introduction of potential infection risks as compared to parenteral routes and additional cost burdens to healthcare systems as compared to injection administration. Positive effects on cognition and function rely upon consistent intake of therapies as prescribed over the long term.
In May 2023, the FDA granted supplemental approval for Rexulti (brexpiprazole) oral tablets, the first drug certified to treat agitation in dementia due to Alzheimer’s disease. Two 12‑week Phase 3 trials demonstrated significant reductions in caregiver‑reported agitation at 2–3 mg doses. Starting at 0.5 mg, doses can escalate to 3 mg based on patient response.
In terms of distribution channel, hospital pharmacies segment is estimated to contribute the highest market share of 46.5% in 2026, owing to its role in reinforcing integrated care models for Alzheimer’s patients. Alzheimer's treatment necessitates a multidisciplinary approach beyond just pharmacotherapy alone. Hospital pharmacies play a central part in coordinating the delivery of comprehensive Alzheimer's care. These work closely with memory clinics, geriatric specialists, nursing staff and social workers to ensure medications are prescribed appropriately and patients transition smoothly between inpatient and outpatient settings as their needs change. Many hospitals also run specialized dementia day programs, providing therapy and respite to delay nursing home placements. The hospital pharmacies act as an anchor, supporting a holistic care model where medication optimization occurs in the context of multifactorial assessment and non-drug interventions.
In December 2024, IDIBAPS–Hospital Clínic Barcelona took part in the HUB Alzheimer Barcelona initiative.
To learn more about this report, Request Free Sample
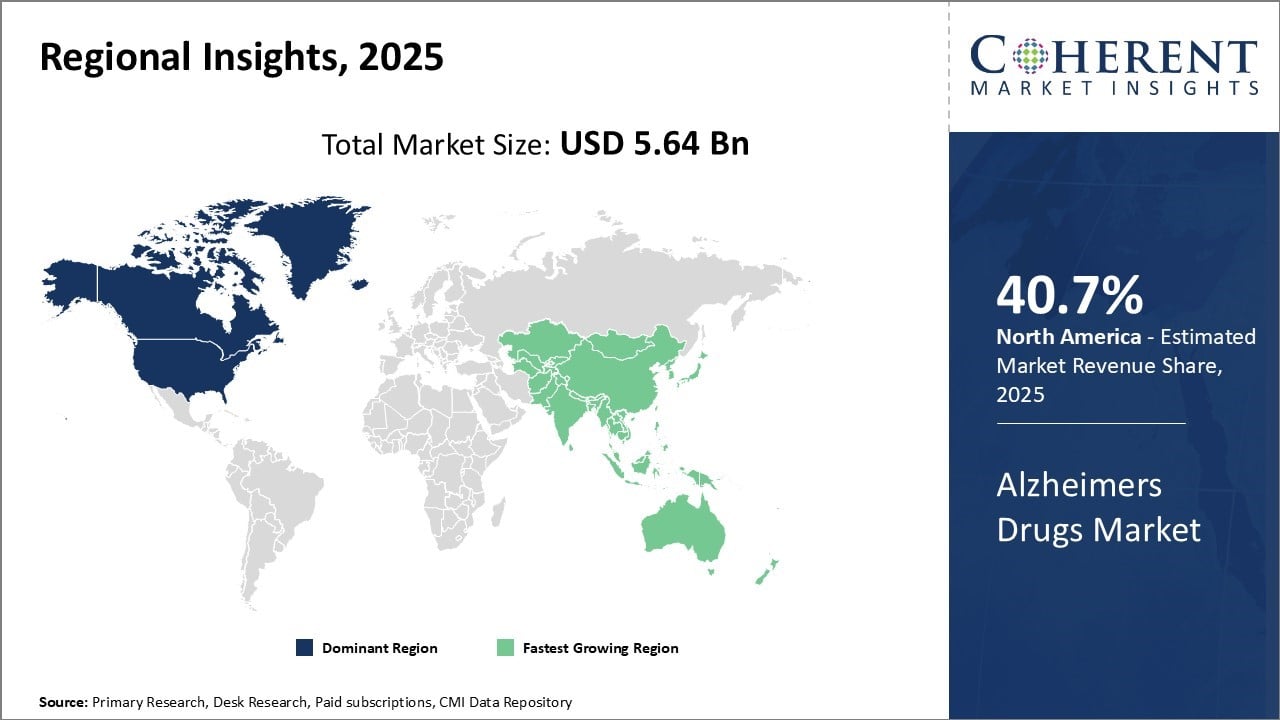
North America dominates global Alzheimer's drugs market with an estimated market share of 40.7% in 2026. The region accounts for the largest market share due to presence of major pharmaceutical companies undertaking research and clinical trials for new drugs. The National Institute on Aging (NIA) in Bethesda, Maryland is leading NIH's efforts in clinical, behavioral, and social research in Alzheimer's and related dementias through efforts aimed at finding ways to treat and ultimately prevent the disorder. Furthermore, aging population and rising prevalence of Alzheimer's disease in the region also drives the market growth. Countries like U.S. have also approved use of several disease-modifying drugs under their regulatory pathways, thus, boosting the Alzheimer drugs market revenue.
Asia Pacific region has emerged as the fastest growing market for Alzheimer's drugs. Asia Pacific market is expected to witness highest CAGR, owing to the increasing focus of international drug makers to tap unmet needs. Countries like China and India are proving lucrative with their huge patient pool and growing healthcare expenditure. For instance, Eli Lilly's Kisunla (donanemab) received regulatory approval in China (Dec 2024) and Japan (Sept 2024), offering once‑monthly infusion and flexible dosing a market first. Local companies are also ramping up their R&D activities in collaboration with global innovators. Regional players like TauRx (Singapore) and Agharkar Institute (India) are advancing tau‑targeted molecules and cholinesterase inhibitors through early‑stage trials. This has led to higher availability of both generic and patented drugs across major Asian countries. Lack of stringent regulations compared to Western nations makes Asia Pacific region an attractive destination for clinical trials of new molecules.
The United States stands as the world’s largest Alzheimer’s drugs market and diagnostics, driven by a rapidly aging population, strong public health infrastructure, and a progressive regulatory landscape. As of 2026, more than 6.7 million Americans aged 65 and older are living with Alzheimer’s disease, a number projected to more than double. This growing burden has spurred significant demand for early diagnosis, effective therapeutics, and long-term care solutions.
Key government initiatives such as Medicare coverage expansion and FDA fast-track pathways have been instrumental in accelerating access to novel therapies. The approval of disease-modifying drugs like Leqembi (lecanemab) and Kisunla (donanemab) under the FDA's Accelerated Approval Program represents a turning point in treatment, shifting focus from symptom management to slowing disease progression. These developments not only enhance patient outcomes but also attract sustained investment from both domestic and global pharmaceutical companies.
With a mature healthcare system, widespread clinical trial networks, and strong advocacy from organizations such as the Alzheimer’s Association, the U.S. continues to lead the charge in Alzheimer’s innovation, access, and care delivery.
China’s Alzheimer’s drugs market is driven by the world’s largest Alzheimer’s patient, with over 10 million diagnosed cases, a number projected to more than double by 2050 due to the country’s rapidly aging demographic.
The country’s rising middle class and increased public awareness are driving strong demand for both early diagnosis and advanced treatment options. Government initiatives such as the "Healthy China 2030" plan aim to expand access to senior healthcare, including cognitive screening and dementia drugs.
International pharmaceutical companies are increasingly focusing on China due to its growing healthcare expenditure, which exceeded USD 1.3 trillion in 2023. China’s regulatory reforms, such as the inclusion of foreign drugs in the National Reimbursement Drug List (NRDL) and the fast-track approval of innovative therapies, have made it an attractive launchpad for new Alzheimer’s treatments. For instance, Eli Lilly’s Kisunla (donanemab) was approved in China in late 2024, marking a significant shift in the country's acceptance of disease-modifying therapies.
Additionally, China plays a pivotal role in global clinical research. The country hosts numerous clinical trial sites for Alzheimer’s therapies, through its vast patient pool, modern hospital infrastructure, and streamlined trial approval process. Leading hospitals and research institutes in Beijing, Shanghai, and Guangzhou are frequently involved in late-stage studies of both local and foreign drug candidates.
As a result, China is emerging not only as a high-demand commercial market but also as a global R&D hub for Alzheimer's drug development, accelerating innovation and access across the Asia-Pacific region.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.24 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.7% | 2033 Value Projection: | USD 12.72 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie, Inc., Teva Pharmaceuticals Ltd., Eisai Co., Ltd., Janssen Pharmaceuticals, Inc. (Johnson & Johnson), Novartis AG, Zydus Lifesciences Ltd (CADILA), Sun Pharmaceutical Industries Ltd., Dr.Reddy's Laboratories Ltd, Aurobindo Pharma Ltd, Amneal Pharmaceuticals Inc., Macleods Pharmaceuticals Ltd., Viatris Inc., Lupin Ltd, Cipla Ltd., Torrent Pharmaceuticals Ltd., Unichem laboratories ltd, Lannett Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Due to rising life expectancy, there has been increase in number of elderly people aged 65 years and over. According to WHO, the proportion of the world's population over 60 years will nearly double from 12% to 22% between 2015 and 2050. Elderly population is particularly susceptible to neurodegenerative conditions such as Alzheimer's and dementia due to natural age-related cognitive decline. Alzheimer's disease is the most common cause of dementia among older adults. As more people survive into their 80s and beyond, even a small increase in diagnosis rates in the very old can cause a dramatic rise in prevalence numbers. In 2023, According to Alzheimer’s Disease International report, around 55 million people have dementia worldwide, the number of people affected is set to rise to 139 million by 2050, with the greatest increases in low- and middle-income countries. Growing elderly populations can increase Alzheimer's cases without effective prevention or cure strategies.
After multiple past failures in treatment development for Alzheimer's, the currently approved medicines can only manage symptoms temporarily without altering disease progression. There has been promising pipeline activity that explores novel mechanisms and molecular targets beyond the amyloid hypothesis. Several companies are developing disease-modifying drugs targeting tau tangles, neuroinflammation, synaptic dysfunction, and other pathways increasingly implicated in Alzheimer's pathology. Large late-stage trials are ongoing to evaluate anti-amyloid antibodies as well as anti-tau antibodies and small molecules for preventing or slowing cognitive decline. Some potential first-in-class therapies for early Alzheimer's include anti-Tau antibody crenezumab from Genentech and anti-amyloid gamma secretase modulator verubecestat from Merck.
The global rise in the elderly population presents a significant opportunity for Alzheimer’s drugs market forecast, driven by increased demand for effective treatments among aging demographics. Alzheimer’s typically affects individuals 65 years and older, and as global life expectancy climbs, so does the number of potential patients. For instance, a recent tool developed by Duke, Harvard, and the University of Otago called DunedinPACNI uses MRI data to predict dementia risk, highlighting the urgency for early intervention before irreversible damage occurs. Moreover, new therapies are emerging: in late 2023, the FDA granted accelerated approval to donanemab (Kisunla) and approved Zunveyl, a prodrug version of galantamine with fewer gastrointestinal side effects, offering patients more tolerable options. These advances underscore a shifting focus toward interventions that delay the onset or progression of Alzheimer’s. As a result, pharmaceutical companies and healthcare providers are well-positioned to serve an expanding cohort of older adults seeking cognitive-preserving treatments, positioning the Alzheimer's drugs market for sustained growth.
*Definition: Global Alzheimer's drugs market consists of pharmaceutical drug therapies that are used for treating patients suffering from Alzheimer's disease. These drugs aim to temporarily slow down the progression of dementia symptoms by targeting neurotransmitters in the brain like acetylcholine and glutamate. Some of the drugs include cholinesterase inhibitors, NMDA receptor antagonists, and others. The market has seen significant growth in recent years due to the increasing prevalence of Alzheimer's disease worldwide.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients