The progressive supranuclear palsy treatment market is anticipated to grow at a CAGR of 12.2% with USD 313 Mn in 2026 and is expected to reach USD 689 Mn in 2033. Growth in this market is due to better research and more awareness of this rare brain disorder. Increasing incidence of progressive supranuclear palsy (PSP) (Globally, 1 per 100,000, with a prevalence rate of five per 100,000 individuals) is expected to boost the market. Doctors and scientists are learning more about Progressive Supranuclear Palsy and focusing on diagnosing it early and giving treatments that fit each patient. This approach can help patients feel better and live healthier lives.
Dopamine is projected to account for the largest share of drug type in 2026, representing approximately 60% of the total volume. Owing to its established therapeutic efficacy in managing the motor symptoms associated with PSP as well as its widespread acceptance among neurologists globally. Rates of HIGH/VERY HIGH DA dosing increased with patient age. Twice as many neurologists (31.1%) prescribed HIGH/VERY HIGH doses vs other specialties.
Dopamine drugs are most used because they treat the main problem in PSP, which is a big loss of dopamine in certain parts of the brain, especially the basal ganglia and brainstem.
The National Institute of Neurological Disorders and Stroke (NINDS) recognizes dopaminergic therapy as a cornerstone treatment approach for PSP patients, despite the generally limited response compared to Parkinson's disease.
Levodopa, the most commonly prescribed dopaminergic medication, remains the first-line treatment recommendation from major neurological associations globally, including the American Academy of Neurology as well as the European Federation of Neurological Societies, primarily because it provides the most predictable symptomatic relief for bradykinesia as well as rigidity symptoms in PSP patients.
To learn more about this report, Request Free Sample
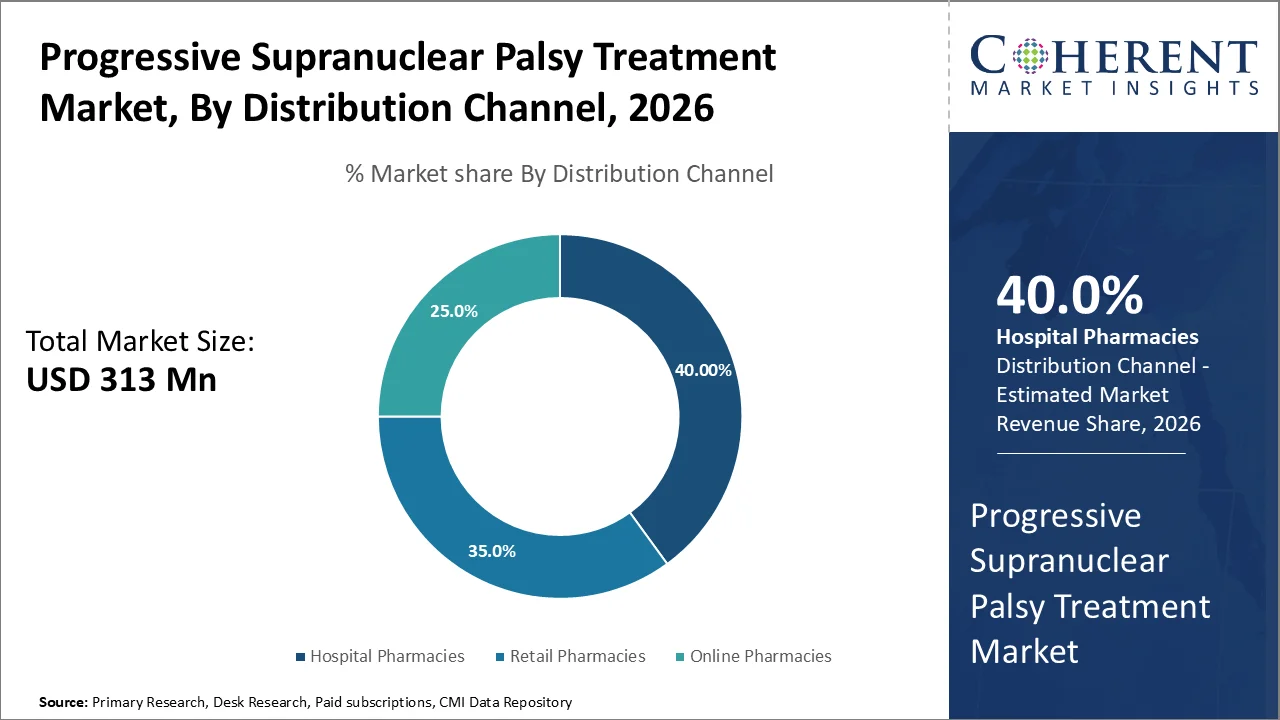
Based on distribution channel, hospital pharmacies dominates the market, accounting for a significant 40% share in 2026, owing to the specialized nature of Progressive Supranuclear Palsy (PSP) as a rare neurodegenerative disorder that needs comprehensive medical supervision as well as expert neurological care.
The dominance of hospital pharmacies in this therapeutic area stems from the complex diagnostic process along with the need for multidisciplinary treatment approaches that are typically available within hospital environment.
According to the Journal of Research in Medical and Dental Science, PSP affects about 6 to 7 people in every 100,000. Treating it needs special expertise, usually found in big hospitals and research centers. These hospitals have brain specialists who treat rare disorders, so patients usually visit hospital pharmacies first for PSP treatment.
Tau aggregation inhibitors are small molecules and antibodies designed to prevent tau protein clumping, a key factor in neurodegeneration. Leading companies include Alector Inc. with AL001, AbbVie with ABBV-8E12, as well as TauRx with methylene blue derivatives (LMTM).
Early trial results are promising: TauRx’s Phase 3 tests with LMTM showed small improvements in thinking for PSP patients, leading to wider access in many countries. Alector’s AL001 cut a marker of nerve damage by 50% in Phase 1b trials, showing it might slow the disease.
|
Current Event |
Description and its Impact |
|
FDA Rare Disease Guidance Update (2025) |
|
|
EU Orphan Drug Regulation Revision (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
North America account 50% market share in 2026, owing to its robust healthcare infrastructure, advanced medical research facilities, and comprehensive patient care networks that have established the region as the global epicenter for Progressive Supranuclear Palsy (PSP) treatment innovation. According to the data published in UCSF Health, about 20,000 Americans, or one in every 100,000 people over age 60 — have PSP.
The presence of world-renowned neurological research institutions such as the Mayo Clinic, Johns Hopkins Hospital, the National Institute of Neurological Disorders and Stroke (NINDS), etc., which have been at the forefront of PSP research and clinical trials.
The region benefits from substantial federal funding through the National Institutes of Health (NIH), which allocated major resources to rare neurological disorder research in recent years. The National Institute of Neurological Disorders and Stroke (NINDS) is a part of the U.S. National Institutes of Health (NIH). It conducts as well as funds research on brain and nervous system disorders and has a budget of just over USD2.03 billion.
The CurePSP foundation, headquartered in New York, has been instrumental in driving awareness and research funding, organizing annual conferences that bring together leading neurologists from across North America.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. Driven by rapidly aging populations as well as increasing diagnostic capabilities across key markets including Japan, South Korea, Australia, etc. The region's growth is particularly evident in Japan, where the Ministry of Health, Labour and Welfare designated PSP as a specified intractable disease in 2022, effectively improving patient access to specialized treatments which is responsible for the growth of the market.
The total population of Japan is 124.35 million people as of October 1, 2023. The number of the population aged 65 and over is 36.23 million. The percentage of the population aged 65 and over (percentage of the elderly) is 29.1%., creates an expanding patient base requiring PSP interventions. Furthermore, the opening of special PSP treatment centers, like the new movement disorder clinic at the National Center of Neurology and Psychiatry in Tokyo, shows Japan’s commitment to treating this rare brain disease using both standard therapies and new protective drugs.
The U.S. contributes the highest share in the Progressive Supranuclear Palsy (PSP) treatment market in North America owing to its advanced healthcare infrastructure, substantial research and development investments, and higher prevalence of diagnosed PSP cases. The United States benefits from having world-renowned neurological research centers such as the Mayo Clinic and Johns Hopkins, which have established dedicated PSP research programs and treatment protocols.
The National Institute of Neurological Disorders and Stroke (NINDS) has funded many PSP treatment studies, with over 15 active trials listed on ClinicalTrials.gov. The U.S. also has big drug companies, like AbbVie, which recently moved their PSP treatments through Phase II trials, showing strong efforts to develop new drugs.
The CurePSP Foundation in New York has the largest PSP patient registry in North America, with over 3,000 patients mostly in the U.S., giving important data for developing treatments. U.S. hospitals have special care programs for PSP, with movement disorder clinics in more than 200 medical centers.
Japan contributes the highest share in the Progressive Supranuclear Palsy Treatment Market in Asia Pacific owing to its advanced healthcare infrastructure, sophisticated neurological research capabilities, along with established pharmaceutical regulatory framework. The nation's demographic profile, characterized by one of the world's most rapidly aging populations, makes a patient pool requiring specialized neurological care. The World Economic Forum, in 2024, the country had a record 36.25 million people aged 65 and over, accounting for 29.3% of the total population. This share is projected to rise to 34.8% in 2040 and 36.3% in 2045.
Japan’s health ministry has made strong healthcare policies for brain diseases. They set up special centers for rare neurological disorders in big cities like Tokyo, Osaka, and Kyoto.
The Japanese Society of Neurology has helped make standard rules for diagnosing and treating PSP, which helps find the disease earlier and manage it better.
Some of the major key players in Progressive Supranuclear Palsy Treatment Market include, AbbVie Inc., Acorda Therapeutics Inc., Biogen Inc., Bristol-Myers Squibb Co., Merck & Co. Inc., UCB Biopharma, AlzProtect, Asceneuron Therapeutics, TauRx Pharmaceuticals, GlaxoSmithKline PLC, Novartis AG, and Teva Pharmaceutical Industries Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 313 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.2% | 2033 Value Projection: | USD 689 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AbbVie Inc., Acorda Therapeutics Inc., Biogen Inc., Bristol-Myers Squibb Co., Merck & Co. Inc., UCB Biopharma, AlzProtect, Asceneuron Therapeutics, TauRx Pharmaceuticals, GlaxoSmithKline PLC, Novartis AG, and Teva Pharmaceutical Industries Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients