Global proton pump inhibitors market is estimated to be valued at USD 4.25 Bn in 2026 and is expected to reach USD 6.31 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.8% from 2026 to 2033.
To learn more about this report, Request Free Sample
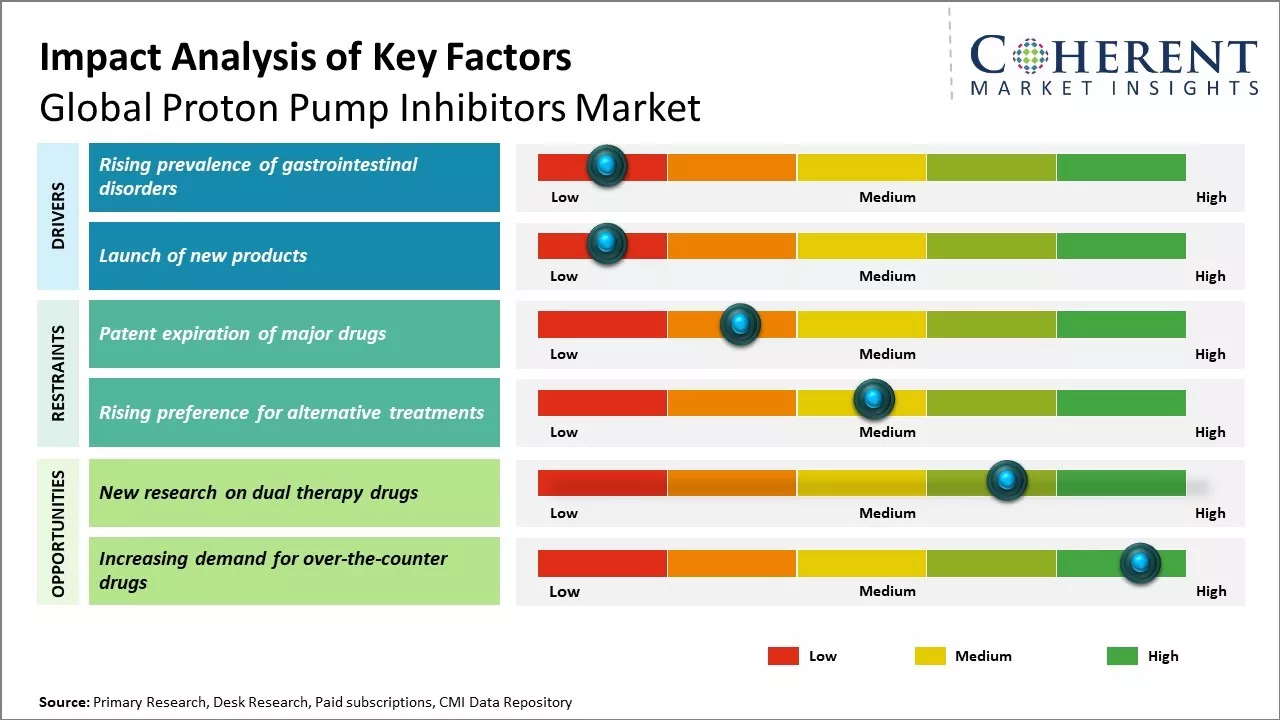
Proton pump inhibitors are being extensively used in the treatment of acid reflux diseases, peptic ulcers and other gastric diseases. Rise in geriatric population prone to such diseases, rise in awareness about treatment procedures and rise in healthcare spending in emerging economies can drive demand for proton pump inhibitors. But patent loss of leading drugs and presence of alternative therapies can restrain the market growth. Impressive pipeline of new drug candidates and growing emphasis on emerging markets by major market participants can provide fresh opportunities in the near term.
|
Current Events |
Description and its impact |
|
Increased Use of Proton Pump Inhibitors (PPIs) |
|
|
Generic PPIs Dominating the Market |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Many Medicare Part D and Part C plans with drug coverage provide reimbursement for omeprazole, a commonly prescribed proton pump inhibitor, when it is prescribed by a doctor. Some Part C plans may even allow beneficiaries to purchase omeprazole over the counter (OTC) and still receive coverage.
Coverage of omeprazole—whether the generic version or the brand-name Prilosec—depends on whether the medication is included in the specific plan’s formulary. Each Medicare Part D plan categorizes drugs into tiers within its formulary, where lower tiers generally mean lower out-of-pocket costs for the beneficiary.
Typically, more commonly prescribed or preferred medications, such as generic omeprazole, are placed in lower tiers compared to brand-name drugs like Prilosec. However, formulary placement can vary between plans.
Additionally, some Part D plans may require beneficiaries to undergo step therapy, meaning they must first try the OTC version of a medication before coverage is approved for the prescription form. This helps manage costs and ensures appropriate use of medications
Increasing incidence of gastrointestinal diseases like acid reflux, heartburn and ulcers can propel the market growth. In 2022, as per the global guidelines released by World Gastroenterology Organization, more than 25% of the world's population experience gastrointestinal problems at some point in life and this increases demand for proton pump inhibitors that provide relief from heartburn. Proton pump inhibitors act by lowering the amount of acid generated in the stomach and are used to relieve the symptoms of acid-related disorders. Many factors including changing eating habits, higher levels of stress, alcohol intake, smoking and inactivity contributes towards rising gastrointestinal disorders. As per the data released by World Health Organization, emerging economies such as India and China that are transforming towards sedentary life and urbanization experience increase in obesity, and this increases the cases of acid reflux disease.
For example, in August 2023, as per the article released by Jaypee Hospital, there has been rise in the occurrence of gastrointestinal (GI) disorders, which has led to serious health concerns. The ability of the GI system to perform digestion, absorption, and expulsion of waste is essential for the overall health. The rise in GI disorders can be attributed to several factors like dietary lifestyle, lifestyle changes, environmental conditions, and genetic inclinations.
To learn more about this report, Request Free Sample
Global proton pump inhibitors market is witnessing growth due to rising number of new product launches by major pharma companies. PPIs are commonly used to reduce excess stomach acid and provide relief from conditions like acid reflux, ulcers and gastroesophageal reflux disease (GERD). Many companies are focusing on developing advanced and more effective PPI formulations to expand their portfolios and market share. Some new products offer dual relief from acid reflux and heartburn symptoms with just one pill. These work faster and for longer duration as compared to existing drugs. Other new launches combine PPIs with other medicines to manage concurrent conditions like H. pylori infection, which causes ulcers. Some companies are also developing PPIs with improved tolerability and reduced interaction with other medications.
Expirations of patents of major drugs can hinder the growth of the global proton pump inhibitors market. When patent exclusivity expires for blockbuster drugs, generic producers can make cheap copies of the original drug. This raises competition and exerts downward pressure on prices. For instance, the patents for Nexium (esomeprazole) expired in Europe and Japan in 2014 and in the United States in 2015. Nexium was among the best-selling drugs in the world, with sales of more than US$ 6 billion annually. The introduction of generic forms of esomeprazole caused an instantaneous drop in the sales of branded Nexium drug. Some of the other blockbuster PPIs that have confronted generic competition include Prilosec (omeprazole), Prevacid (lansoprazole) and Aciphex (rabeprazole).
Novel research is investigating the possibility of dual therapy drugs, which can simultaneously target gastric acid secretion and gastric epithelial protection. This provides a significant prospect for proton pump inhibitors market across the world. Dual therapy medicines have exhibited great success in clinical trials in significantly lessening symptoms of gastroesophageal reflux disease compared to traditional PPIs. Dual therapies by inhibiting acid suppression as well as mucosal defense could be better at providing symptom relief for acid-related disorders patients. This new drug class has the potential to address some of the existing limitations of long-term PPI therapy, including decreased efficacy with time, absence of mucosal healing, and effects on antibiotic resistance with prolonged acid suppression.
Dual therapies, if approved, have the potential to gain a significant share of the market in 5-10 years. In 2021, more than 300 million individuals suffer from gastroesophageal reflux disease worldwide annually, as reported in a United Nations report. The disease significantly affects quality of life and daily functioning in many of its sufferers. Dual therapies can provide better results for this huge patient population in which standard PPIs cannot completely manage acid reflux symptoms. Their enhanced clinical effectiveness can lead to greater patient compliance with treatment regimens. This affects total disease management expense transferred by high rates of physician visits and slow symptom resolution with existing standard of care measures.
On the basis of type, omeprazole segment is likely to have the largest market share of 31.7% in 2026, thanks to its excellent brand loyalty and recognition with customers. Since the first PPI drug ever to be marketed, omeprazole set the standard as the gold standard in the treatment of ulcers and GERD. Despite the introduction of newer PPIs, omeprazole continues benefiting from the brand equity and trust it had built among consumers. Patients who were initially prescribed omeprazole have remained faithful to the brand even after their conditions were resolved, preferring its proven efficacy and tolerability over alternatives.
Besides patients, even physicians have high familiarity with omeprazole's clinical profile, and are more likely to prescribe it as the first line treatment. Omeprazole also greatly gains from the brand pull effect through word-of-mouth referrals to new patients from social and business networks. Having an older molecule, omeprazole gains relatively less in terms of generics competition compared to other PPIs.
To learn more about this report, Request Free Sample
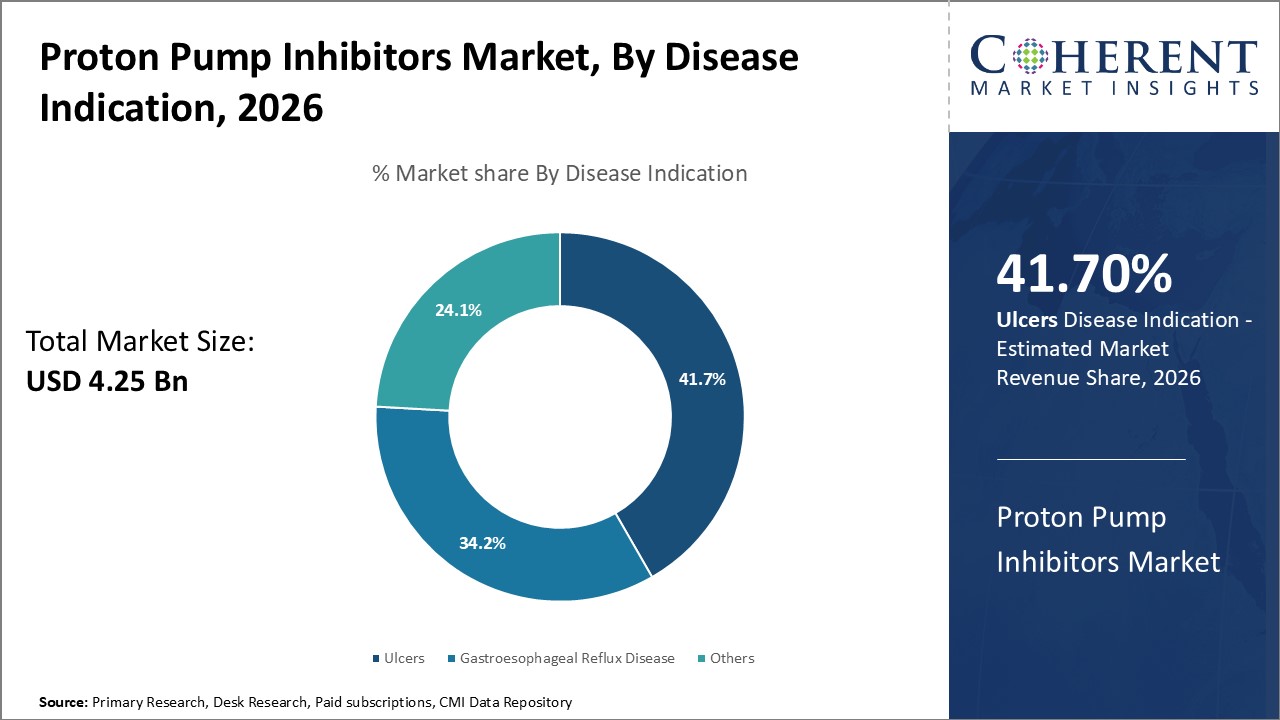
Based on the indication of the disease, the ulcers segment is projected to have the maximum market share of 41.70% during 2026, due to the increasing prevalence of gastroesophageal reflux disease (GERD). GERD is a long-term illness where stomach acid flows back up into the throat. If not treated, it may lead to esophageal damage over a period of time, which may result in ulcers. Since lifestyle modifications such as rising levels of stress, late-night meals, alcohol/tobacco overuse, and lack of exercise have become endemic across the world, GERD cases have doubled in the past two decades.
Since GERD is a leading risk factor for ulcers, its increasing prevalence has directly increased the incidence and treatments for ulcers. PPI medications such as omeprazole that deliver effective relief of gastrointestinal acidity are first-line options for the management of both GERD and consequential ulcers. Provided their capacity to cure current ulcers and likewise prevent recurrence, PPIs now account for more than 80% of overall ulcer treatment regimens.
Based on the dosage form, tablets segment is anticipated to account for the maximum market share of 61.21 % in 2026 due to ease and popularity compared to other forms. As an oral solid dose, tablets ensure ease of intake and regular dosing without complex administration processes. This ensures better treatment compliance for PPIs that require them to be administered frequently over prolonged periods.
Also, tablets possess a robust global presence within healthcare systems compared to other modes. Their global acceptability is supported by qualities of mass producibility, extended shelf life, and inexpensive dissemination even in the farthest reaches. Capsules are also very portable but are a less desirable mode because of swallowing challenges for some patients. Injections have very limited application because PPI therapy is usually of long-term outpatient nature. Since tablets address treatment requirements in the least inconvenient way, these become the leading segment.
To learn more about this report, Request Free Sample
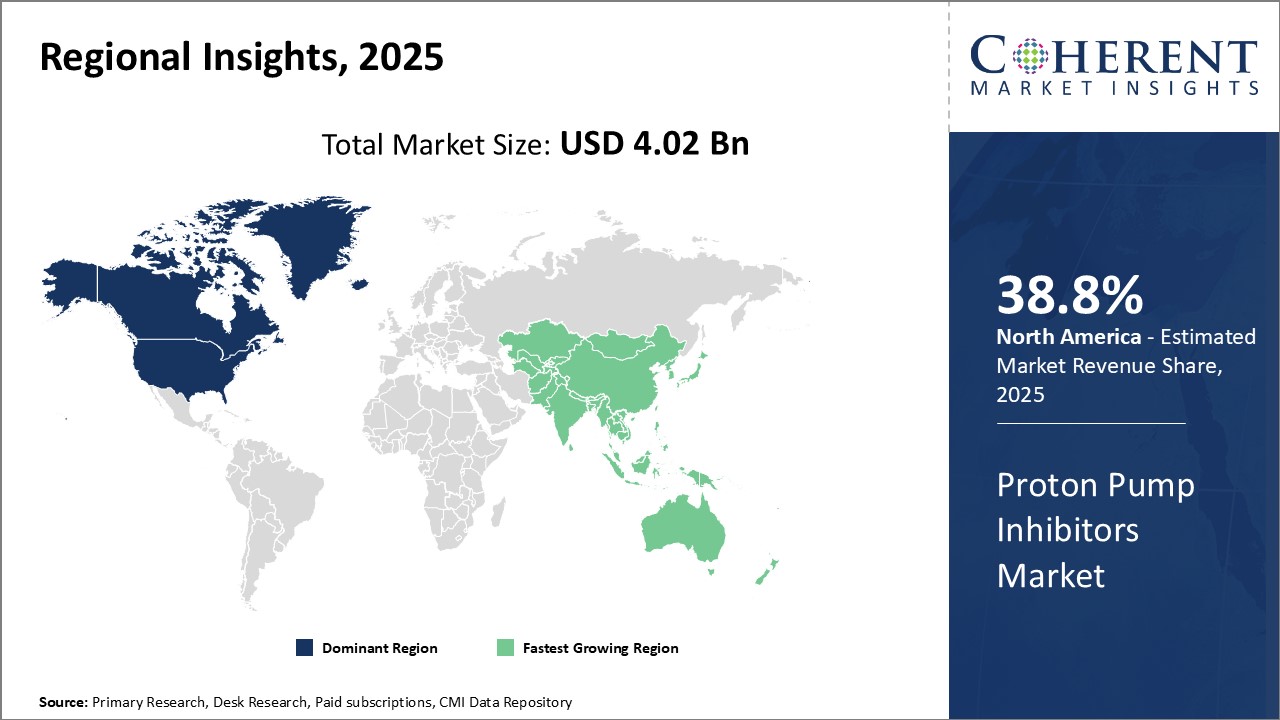
North America has proved to be the leading regional proton pump inhibitor market worldwide with an estimated market value of 38.8% in 2026. Favourable reimbursement policy and increased expenditure on healthcare by the U.S. and Canada are the growth drivers for the regional market. The region serves as the country of origin as well as home to the global headquarters of giant pharmaceutical companies, enabling high levels of R&D spending and solid product pipelines. There is a high concentration of industry players such as Pfizer and AstraZeneca that are renowned for their brand loyalty with existing products.
Additionally, the region supports medical consciousness and utilization of preventive health practices for gastric complications such as ulcers. This has made it possible for proton pump inhibitors to find mainstream therapy as an option. Nevertheless, generic and competitor pricing pressure could hinder potential future growth for innovator brands in North America.
Asia Pacific region has become the fastest growing market for proton pump inhibitors globally. Increasing healthcare spending at a rapid pace, growing obesity as a result of lifestyle and dietary changes have driven demand for acid-reducing medications. India, China and Japan have a huge patient base with gastric diseases, due to their aging population and genetic risks. Local players in these markets have consolidated their dominance with affordable generic equivalents.
Global brands are also making an entry in this market with product licensing, joint ventures and imports. But absence of strict regulation and enforcement has encouraged proliferation of sub-standard imitations. This threatens to wreak havoc on successful innovators while accruing benefits to local unorganized players.
The United States accounts for a major share of the Proton Pump Inhibitors (PPI) market due to high healthcare spending, a huge patient base of gastrointestinal disorder patients, and well-established reimbursement systems. Prescription PPIs such as omeprazole, pantoprazole, and esomeprazole are prescribed in large volumes and are reimbursed by Medicare, Medicaid, and private insurance. Demand for prescription and over-the-counter PPIs continues to increase because of the rising population, as well as improving awareness about gastrointestinal health.
Germany is among the top European markets for PPIs, boasting a strong healthcare system and strong prescription rates for PPIs. The public health insurance system in Germany, which is well-regulated, guarantees that patients suffering from gastrointestinal diseases have access to PPIs, thereby making them a first-line therapy. Moreover, the increasing number of acid reflux and GERD cases in Germany ensures constant demand for PPIs.
Japan is an important PPI market within the Asia-Pacific region. Increasing lifestyle-related gastrointestinal disorders, and the aging population in Japan, are the primary drivers of PPI treatment demand. The Japanese national health insurance system provides for wide availability of drugs, such as PPIs, enhancing their use as a treatment option for chronic illnesses like GERD and peptic ulcers.
China is experiencing fast growth in the market for PPI, primarily because of advancements in healthcare infrastructure and growing awareness of gastrointestinal disorders. With urbanization and aging population in the country, the need for PPI to treat diseases like GERD and peptic ulcers will grow. Government-sponsored health insurance programs have helped make PPIs available throughout the population.
The Proton Pump Inhibitors (PPI) Market has seen rapid growth in the last few years, fueled by the expanding disease incidence of gastrointestinal conditions like acid reflux, ulcers, and GERD (gastroesophageal reflux disease). The pipeline of PPIs indicates a number of developments to enhance efficacy, decrease side effects, and provide longer duration of treatment. Major players in the industry are interested in creating new PPI formulations with a focus on specific patient segments, including resistance to existing therapy or alternative routes of delivery (e.g., once-daily administration).
Research and development focus on new drugs that combine PPIs with other therapeutic agents to treat root causes of acid-related diseases, enhancing symptom control and disease management. Also in the pipelines are formulations with greater bioavailability and fewer adverse effects. There is also increased emphasis placed on the geriatric and paediatric segments, whose unique PPI needs must be addressed. There is competition for the market that exists between mature pharma and the biotech’s that produce new-generation medicines.
The intellectual property environment for the Proton Pump Inhibitors (PPI) market is ever-changing, with a high degree of intellectual property ownership by the large pharmaceutical corporations. Well-established PPIs like omeprazole, lansoprazole, and esomeprazole have had their initial patents lapse, opening up space for generic alternatives and more market competition. But several of these firms have taken their patent cover in other directions, including secondary patents covering novel forms, combination drugs, or novel delivery systems.
The major players in the PPI market, such as AstraZeneca, Takeda, and Pfizer, are currently engaged in patenting novel PPI compounds or formulations with enhanced pharmacokinetics, decreased side effects, or increased patient compliance. Much of the patent applications are aimed at extended-release products, fixed-dose combinations with other medications, and formulations to decrease the risk of PPI-associated side effects like osteoporosis or kidney disease. Upstart biotech firms are also making innovations in the PPI market, with targeted treatments or non-oral administration routes. The patent situation also indicates active work on increasing the application of PPIs to indications outside classic acid reflux, such as conditions such as Helicobacter pylori eradication and Zollinger-Ellison syndrome.
Proton Pump Inhibitors Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.25 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.8% | 2033 Value Projection: | USD 6.31 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
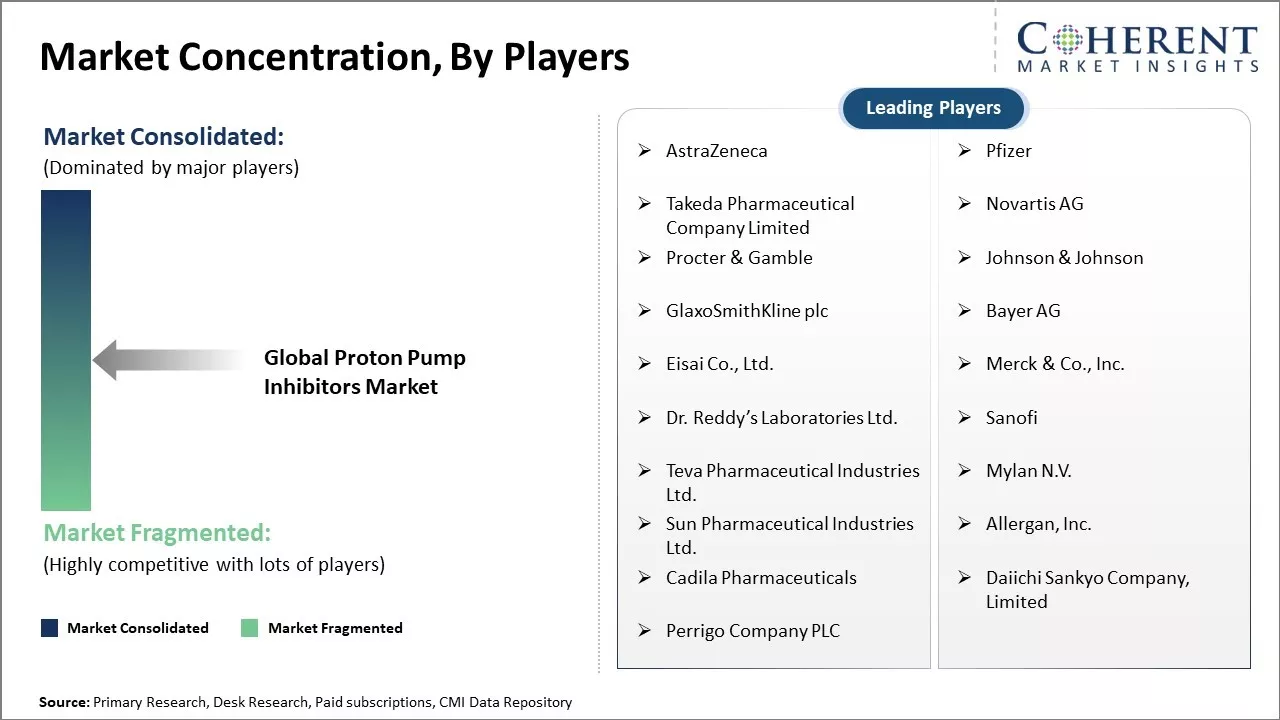
AstraZeneca, Pfizer, Takeda Pharmaceutical Company Limited, Novartis AG, Procter & Gamble, Johnson & Johnson, GlaxoSmithKline plc, Bayer AG, Eisai Co., Ltd., Merck & Co., Inc., Dr. Reddy’s Laboratories Ltd., Sanofi, Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Allergan, Inc., Cadila Pharmaceuticals, Daiichi Sankyo Company, Limited, Perrigo Company PLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Proprietary CMI Existing Repository of Information (Last 8 Years)
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients