The BRAF Kinase Inhibitors market is anticipated to grow at a CAGR of 8.3% with USD 2.12 Bn in 2026 and is expected to reach USD 3.52 Bn in 2033. The market growth is driven by increasing prevalence of BRAF-mutant cancers (approximately 500,000 new cases globally), advancements in precision medicine, and rising adoption of combination therapies. Pharmaceutical companies are actively investing in R&D to expand therapeutic applications and develop next-generation inhibitors with improved safety profiles.
Vemurafenib is projected to account for the largest share of drug in 2026, representing approximately 45% of the total volume. Owing to its position as the first-in-class BRAF inhibitor that received regulatory approval and established the foundation for targeted therapy in BRAF-mutated cancers. The extensive clinical validation through landmark trials and its early market entry advantage, which allowed it to capture significant market share before competitors emerged.
About 40% of cutaneous melanomas carry BRAF mutations-more common in younger patients-and these are linked to more aggressive behavior and a higher risk of brain metastasis.
Vemurafenib’s pioneering status as one of the first BRAF inhibitors to receive approval by major regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) has established its dominant presence.
Its approval was supported by pivotal clinical trials like the BRIM-3 study, which showed its superiority over conventional chemotherapy, setting a precedent for targeted therapy in melanoma and thereby holding its position as the go-to treatment for patients harboring the relevant mutation.
To learn more about this report, Request Free Sample
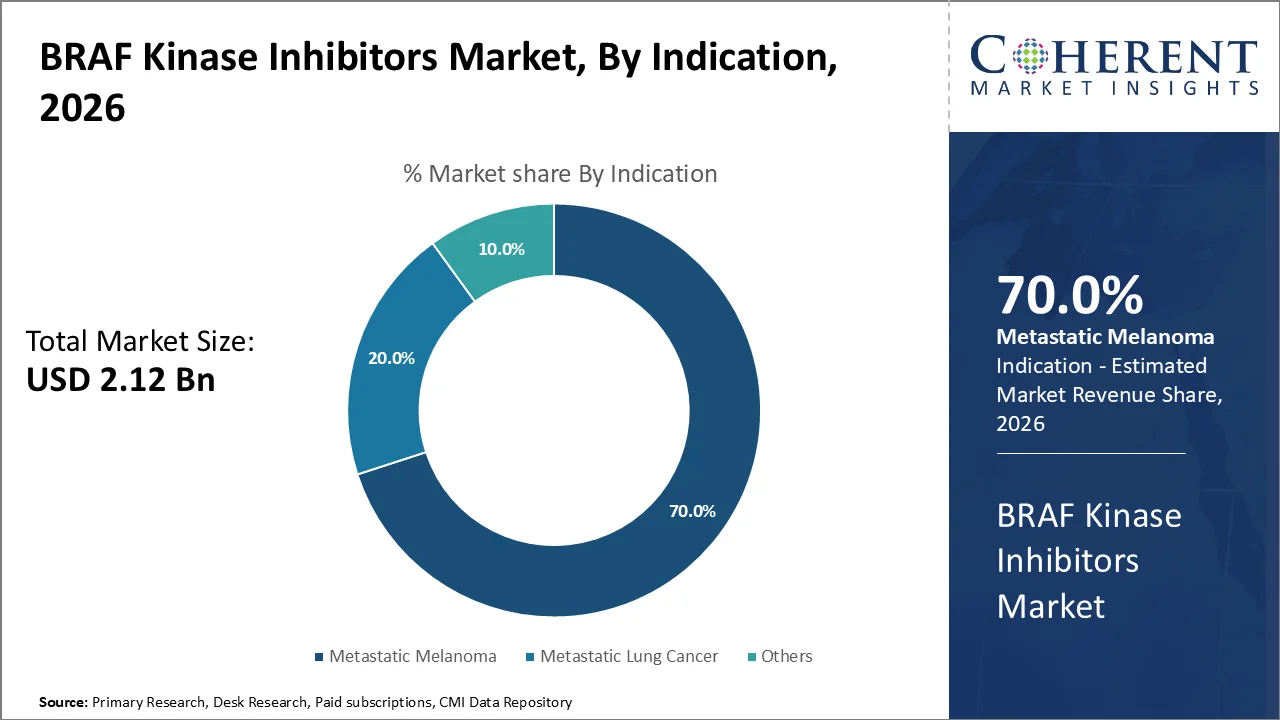
Based on indication, metastatic melanoma dominates the market, accounting for a significant 70% share in 2026, the most frequent oncogenic BRAF mutations, particularly V600E, constitutively activate the kinase, creating a highly actionable target. BRAF mutations, particularly the V600E mutation, are identified in approximately 50% of cutaneous melanomas, making targeted inhibition a highly effective therapeutic strategy.
In 2025, approximately 140,000–175,000 new BRAF-mutant melanoma cases globally are expected, largely driven by V600E. Constitutive kinase activation by V600E leads to aggressive tumor growth, making selective BRAF inhibitors highly effective in advancing response rates as well as survival.
The dominance of the Metastatic Melanoma segment is further underpinned by clinical practice guidelines established by authoritative bodies like the National Comprehensive Cancer Network (NCCN) and the American Society of Clinical Oncology (ASCO), which highly recommend BRAF inhibitors either as monotherapy or in combination with MEK inhibitors for patients harboring BRAF mutations.
Based on distribution channel, hospital pharmacies dominate the market, accounting for a significant 80% share in 2026, as they work closely with doctors, have early access to new treatments, and BRAF kinase inhibitors are very important in cancer care.
For 2025, DCI has identified more than 1,900 dispensing locations with specialty pharmacy accreditation from one or both of the two major independent accreditation organizations. The overall number of accredited locations grew by only 3% in 2025, but is more than five times larger than the 2015 figure.
Hospital pharmacies serve as the primary dispensing points for patients undergoing cancer treatment, particularly for those with malignancies such as melanoma, lung cancer, and colorectal cancer, where BRAF mutations are clinically significant.
These institutions are usually the initial point of drug administration following oncologist prescription, ensuring careful adherence to therapy protocols as well as close monitoring of treatment efficacy and side effects.
Selective BRAF inhibitors are made using advanced computational modeling and X-ray crystallography to map the three-dimensional structure of the kinase domain. These structure-based approaches allow researchers to precisely target the active site and optimize drug interactions for higher efficacy and safety.
Companies such as Plexxikon/Roche (vemurafenib), GlaxoSmithKline (dabrafenib), and Array BioPharma/Pfizer (encorafenib) have applied these techniques to fuel drug development. Adoption of structure-based design has shortened development timelines from 15+ years to 8–10 years, improved selectivity by 100–1000-fold over first-generation inhibitors, as well as reduced late-stage failures by around 40%.
Fragment-Based Drug Discovery (FBDD) involves screening small molecular fragments (150–250 Da) that bind weakly to target proteins and then optimizing them into potent inhibitors. This way allows precise identification of binding sites, including novel and allosteric sites, improving drug design efficiency.
Companies like Plexxikon (vemurafenib), Astex Pharmaceuticals (AT9283), SGX Pharmaceuticals, etc., have leveraged FBDD for kinase inhibitor development. The method has stimulated hit-to-lead success rates from 1–2% to 15–20%, lower the size of compound libraries by 90%, and facilitated the discovery of novel targets and allosteric inhibitors.
|
Current Event |
Description and its Impact |
|
FDA Approval of Lifirafenib (2025) |
|
|
EMA Regulatory Guidance on Combination Therapies (2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
North America account 50% market share in 2026, North America, owing to a well-established healthcare infrastructure, advanced research facilities, and early adoption of innovative oncology treatments. As of January 1, 2025, about 18.6 million people were living in the United States with a history of cancer, and this number is projected to exceed 22 million by 2035.
The three most prevalent cancers are prostate (3,552,460), melanoma of the skin (816,580), and colorectum (729,550) among males and breast (4,305,570), uterine corpus (945,540), and thyroid (859,890) among females.
The United States, which is a major part of this segment, has been at the forefront of developing and approving targeted therapies for cancers involving BRAF mutations, such as melanoma and non-small cell lung cancer (NSCLC).
For example, the U.S. Food and Drug Administration (FDA) has granted multiple approvals to BRAF kinase inhibitors like vemurafenib as well as dabrafenib, which highlights a strong regulatory environment that supports rapid introduction and utilization of these therapies.
Moreover, the U.S. National Cancer Institute (NCI) data shows a high incidence rate of melanoma as well as other BRAF mutation-associated cancers, fueling demand for more effective treatments. The American Cancer Society’s estimates for melanoma in the United States for 2026 are: About 112,000 new melanomas will be diagnosed (about 65,400 in men and 46,600 in women). About 8,510 people are expected to die of melanoma (about 5,500 men and 3,010 women).
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.2%. Growth is being driven by better cancer diagnosis in emerging countries. Programs like China’s Healthy China 2030 aid in investing in cancer care and making new treatments, including BRAF inhibitors, more available.
China has added about 270 cancer drugs to its national reimbursement list. This shows the government wants to make new cancer treatments easier to get. As part of the Healthy China 2030 program, this helps more patients use precision treatments like BRAF inhibitors and improves cancer diagnosis and treatment across the country.
Japan’s PMDA has made it faster to approve new cancer treatments. This lets new BRAF inhibitors, designed for Asian patients with specific genetic traits, reach the market more quickly.
The U.S. contributes the highest share in the BRAF Kinase Inhibitors market in North America owing to its sophisticated healthcare infrastructure, extensive pharmaceutical research ecosystem, as well as established regulatory framework that facilitates drug development along with commercialization.
The Food and Drug Administration's accelerated approval pathways have been instrumental in bringing BRAF inhibitors like vemurafenib, dabrafenib, and encorafenib to market rapidly, with the FDA being among the first regulatory bodies globally to approve these targeted therapies for melanoma as well as other BRAF-mutated cancers.
The National Cancer Institute's SEER database indicates that melanoma incidence rates in the United States have been consistently high, creating a substantial patient population having targeted BRAF inhibitor treatments.
Prominent drug companies like Roche/Genentech, Novartis, and Pfizer have set up strong manufacturing and distribution networks in the U.S. This ensures BRAF inhibitors are available at cancer treatment centers as well as major hospitals.
For instance, in February 2026, Pfizer Inc. announced that the U.S. Food and Drug Administration (FDA) has granted full approval to BRAFTOVI (encorafenib) in combination with cetuximab (marketed as ERBITUX®) and fluorouracil-based chemotherapy for the treatment of adult patients with metastatic colorectal cancer (mCRC) with a BRAF V600E mutation based on results from the global Phase 3 BREAKWATER trial (NCT04607421).
Japan contributes the highest share in the BRAF kinase inhibitors market in the region owing to a combination of advanced healthcare infrastructure, strong government support for oncology research, and early adoption of precision medicine technologies. Japan has one of the most developed healthcare systems in Asia, with significant investments made into cancer treatment as well as genomics-based therapies.
The National Cancer Center Japan says that melanoma and other cancers with BRAF mutations are being treated more with advanced therapies. This is supported by strong clinical trials and early approval of BRAF inhibitors.
In November 2025, Ono Pharmaceutical Co., Ltd. announced that Ono received a supplemental approval for BRAFTOVI (generic name: encorafenib) Capsule (“Braftovi”), a BRAF inhibitor, in combination with cetuximab, an anti-human EGFR monoclonal antibody, and chemotherapy for the indication of unresectable, advanced or recurrent colorectal cancer (CRC) with BRAF mutation.
For example, the approval of BRAF kinase inhibitors like dabrafenib and vemurafenib under the “Sakigake” fast-track designation by the Ministry of Health, Labour and Welfare showcases Japan’s proactive stance in facilitating early patient access to innovative oncology drugs.
Some of the major key players in BRAF kinase inhibitors market are, F. Hoffmann-La Roche Ltd., Novartis AG, Array BioPharma Inc. (Pfizer), Bayer AG, Plexxikon Inc. (Daiichi Sankyo), Blueprint Medicines, Kinnate Biopharma Inc., IDEAYA Biosciences, Mirati Therapeutics, Loxo Oncology (Eli Lilly), BeiGene Ltd., Spectrum Pharmaceuticals, Ono Pharmaceutical, Chugai Pharmaceutical, BridgeBio Pharma
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.12 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 3.52 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche Ltd., Novartis AG, Array BioPharma Inc. (Pfizer), Bayer AG, Plexxikon Inc. (Daiichi Sankyo), Blueprint Medicines, Kinnate Biopharma Inc., IDEAYA Biosciences, Mirati Therapeutics, Loxo Oncology (Eli Lilly), BeiGene Ltd., Spectrum Pharmaceuticals, Ono Pharmaceutical, Chugai Pharmaceutical, BridgeBio Pharma |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients