Real World Data (RWD) Market size is estimated to be valued at USD 2.73 Bn in 2026 and is expected to reach USD 7.08 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 14.6% from 2026 to 2033.
The increasing adoption of RWD in drug development and approvals, market access, and post-market surveillance is driving the growth of the market. RWD is data collected from real-world settings, such as electronic health records (EHRs), claims data, and patient registries. It can be used to generate evidence on the safety and efficacy of drugs and medical devices, as well as to assess the impact of treatments on patient outcomes.
|
Event |
Description and Impact |
|
Regulatory Developments Impacting RWD Integration |
|
|
Technological Advancements in Data Infrastructure |
|
|
Geopolitical Data Governance Challenges |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The Real-world Data (RWD) market growth is supported by a robust pipeline of pharmaceutical candidates across multiple phases of clinical development. These candidates are expected to generate substantial real-world evidence upon approval and commercialization, enhancing insights into safety, efficacy, and treatment outcomes in diverse patient populations.
These pipeline molecules span various therapeutic areas, reflecting the growing interest of sponsors and innovators in leveraging RWD to support regulatory approvals, post-marketing surveillance, and personalized medicine strategies.
The RWD market forecast is experiencing a rapidly evolving and expanding ecosystem of patents which is fuelled by investments from industry leaders and new market entrants seeking to gain competitive edges in analytic, integration, and application technologies. Patents concerning RWD are mainly concerned with advancements in infrastructure of data collection systems, data anonymization, AI-based analytics, and interoperability issues.
Furthermore, some of the world’s largest pharmaceutical companies, IT business firms, and healthcare analytics companies are actively patenting new algorithms and platforms to automate real-world evidence generation, enrich the data, and ensure compliance with regulatory requirements. Recent patents also focus on the use of blockchain and other safe methods of data sharing to remove privacy and security risks associated with sensitive health information, enhancing the overall privacy and security of health data.
The geographic concentration of this patenting activity is in North Amercia and Europe, where there are advance and well-defined regulatory frameworks for patents and high-quality intellectual property systems. However, the new patent applications from Asia Pacific jurisdictions suggest some increased interest and innovative activity in the RWD technologies in this region.
In general, the evolving patents’ geography and focus of emerging patents indicate an increased competition in the RWD markets and frontiers highlights the growing importance of intellectual property related to technological development and market differentiation.
The reimbursement dynamics of Real-world Data (RWD) is influenced by a myriad of coding schemes, guidelines, and insurance models across the globe. Critical coding frameworks like RWD's ICD-10, CPT, and HCPCS provide appropriate classification of services tied to RWD’s ancillary services and data privacy legislations like GDPR in Europe as well as HIPAA in the US safeguards the data.
FDA (U.S.), EMA (Europe), and PMDA (Japan), as regulatory authorities, are key players utilizing RWD for decisions related to drug and device approval and reimbursement for both purposes. HTA bodies evaluate the clinical and economic value of interventions that have RWD applied to them for informing coverage policies.
In regards to spending, RWD associated expenditures only account for a minute portion of global healthcare spending: 1-2% of Medicare’s $799.4 billion in the US, approximately 0.5-1% of NHS £129.7 billion budget from the UK, along with similar portions from Japan and Australia. It’s estimated that China’s Basic Medical Insurance system spends around 0.3-0.5% of their total budget on RWD-related services.
While the direct costs of RWD are low, their importance is growing due to increased use of RWD in regulatory approvals and value-based care. This emerging context of reimbursement policy signals a paradigm shift in the value of RWD in the health care system.
The Real-World Data (RWD) supplement is pivotal for healthcare providers and practitioners as a patient’s condition changes. A clinician's dependability on RWD shifts depending on the particular line of treatment as well as the disease stage. While first-line treatments adhere strongly to clinical trials and set guidelines, there is more RWD reliance when it comes to second line and later treatments due to necessity and importance of insight into how medications work and their safety in “real-life” situations as opposed to trial settings.
The disease progressing also changes the RWD spectrum that is used; early-stage disease management tends to follow evidence that comes from clinical trials, while more advanced or complicated stages will rely heavily on RWD to fine-tune the required therapies. For instance, prescribers tend to use established medications where dominant trials support their efficacy, but further down the timeline may change their stance and look at other options which RWD showcases performing better via alternative or combination therapies.
Other reasons changing preferring RWD includes patient characteristics (age group, existing illness, ethnicity), the type of practice (academic or community practice), provided incentives by the healthcare system (value based or fee-for-service models), and the use of RWD in the form of electronic health records and other decision-support tools.
Overall in these case scenarios, where there are boundaries to evidence, prescribers have shown to depend more on RWD incorporating more of it when it comes to creating more personalized approaches to treatment plans.
In terms of Application, Post Market Surveillance segment is projected to hold the largest Real World Data (RWD) market share of approximately 31% by 2026, driven primarily by the increasing regulatory emphasis on ongoing safety and performance monitoring of products after their release. As regulatory authorities intensify post-market requirements to ensure continuous compliance and mitigate risks, organizations are prioritizing comprehensive surveillance strategies.
Post market surveillance systems enable manufacturers and regulatory bodies to systematically collect, analyze, and act on real-world data related to product performance and adverse events. These systems enhance transparency by facilitating timely identification of safety concerns, thereby protecting end-users and supporting proactive risk management.
Furthermore, the integration of advanced data analytics and real-time reporting tools strengthens decision-making processes, ensuring swift corrective actions and regulatory compliance. Consequently, the growing focus on patient safety and regulatory oversight continues to drive the adoption of post market surveillance solutions across industries.
To learn more about this report, Request Free Sample
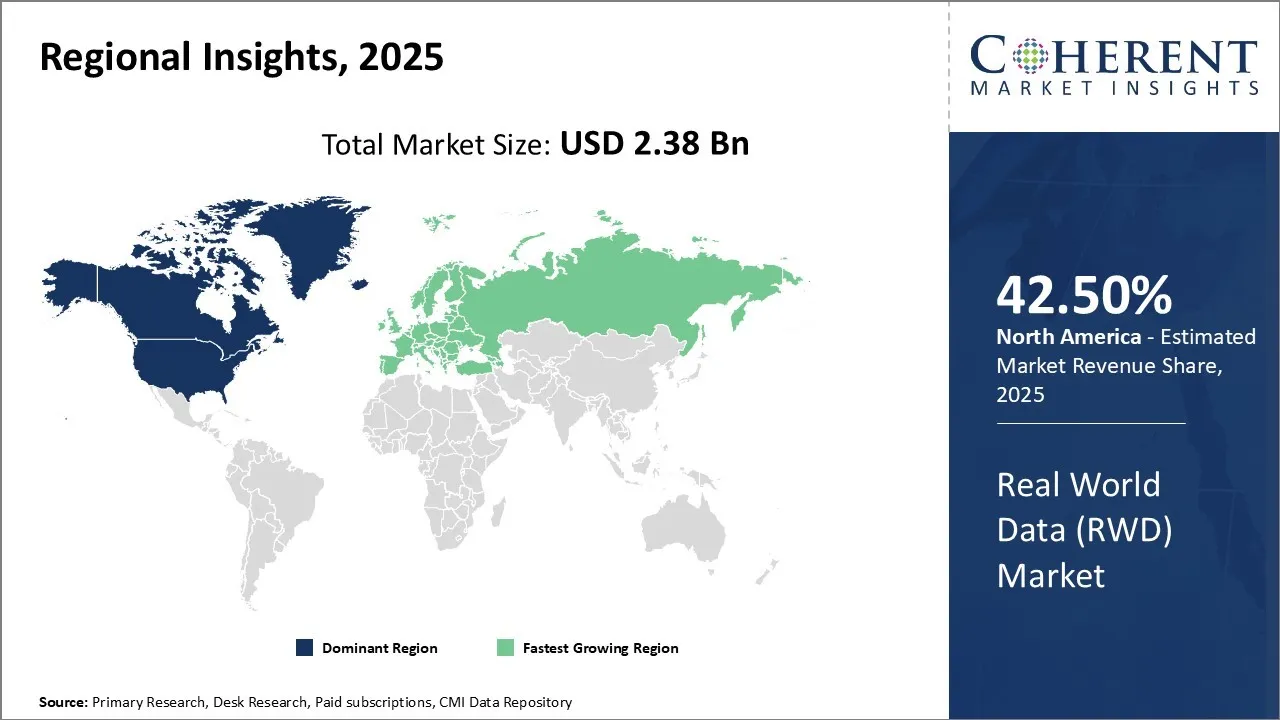
North America is forecasted to lead the global real world data market demand in 2026, holding a market share of approximately 42.5%. This leadership is driven by the region’s advanced healthcare infrastructure, well-established pharmaceutical and biotechnology sectors, and stringent regulatory frameworks governing drug approvals and clinical research.
The U.S. and Canada are key contributors to market growth, supported by increasing prevalence of chronic diseases such as cancer, diabetes, and cardiovascular conditions, which demand comprehensive real world evidence for improved patient outcomes.
The region benefits from sophisticated health IT systems, extensive electronic health record (EHR) adoption, and strong government initiatives promoting the integration of real world data into healthcare decision-making. Additionally, investments in data privacy and security frameworks facilitate the ethical and compliant use of large-scale health datasets, further accelerating RWD market expansion.
Europe is positioned as the second-largest real world data market value in 2026, with an anticipated share of 28.5%. The region’s prominence stems from its robust focus on healthcare innovation and widespread adoption of big data analytics in clinical research and policy formulation. Countries such as Germany, the UK, France, and the Nordic nations are spearheading efforts to harness RWD to enhance healthcare delivery, regulatory evaluations, and pharmacovigilance.
Strong collaborations between public health agencies, research institutions, and private sector players foster a conducive environment for RWD utilization. Additionally, Europe’s progressive regulatory landscape, including frameworks like the General Data Protection Regulation (GDPR), balances data accessibility with patient privacy, enabling trust and transparency in real world evidence generation.
Asia Pacific is emerging as the fastest-growing region in the global real world data market, with a robust CAGR of 10.5% anticipated through 2026. This rapid expansion is fueled by the region’s large and diverse population base, increasing burden of chronic and lifestyle diseases, and expanding healthcare infrastructure. Key markets such as China, India, Japan, and Southeast Asia are witnessing growing government support and strategic initiatives to integrate RWD in clinical research and healthcare policymaking.
The rise in digital health adoption, improvements in health data digitization, and the growing presence of local and multinational pharma companies investing in real world evidence capabilities further accelerate market growth. Additionally, increasing awareness about the benefits of RWD in improving treatment effectiveness and reducing healthcare costs drives its adoption across public and private healthcare sectors.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.73 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.6% | 2033 Value Projection: | USD 7.08 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
IQVIA Holdings Inc., Optum, Inc. (a subsidiary of UnitedHealth Group), Cerner Corporation, Flatiron Health, Inc., IBM Corporation, Tempus Labs Inc., Syneos Health Inc., Evidera, Inc., Palantir Technologies Inc., SAS Institute Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The widespread adoption of electronic health records by healthcare providers has led to the accumulation of vast amounts of patient data. This data, when aggregated and anonymized, can provide valuable insights into real-world patient outcomes, treatment patterns, and disease progression.
For instance, on January 16, 2023, Fujitsu, the information and communications technology equipment and services corporation, and Sapporo Medical University announced the launch of a joint project to realize data portability for patients’ healthcare data, including electronic health records (EHRs) (2) and personal health records (PHRs).
There is a growing emphasis on evidence-based medicine, which relies on real-world evidence (RWE) derived from RWD. Stakeholders in healthcare, including regulators, payers, and healthcare providers, are recognizing the importance of using real-world data to make informed decisions about treatment effectiveness, comparative effectiveness, safety, and cost-effectiveness.
For instance, on January 16, 2023, according to an article published in the Nature Medicine journal, advances in wearable technologies, data science, and machine learning have begun to transform evidence-based medicine, offering a tantalizing glimpse into a future of next-generation ‘deep’ medicine. Moreover, bringing a drug successfully through all phases of drug development into the clinic costs more than 5–2.5 billion dollars. Therefore, innovative strategies are needed to engage patients and generate the necessary evidence to propel new advances.
Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have recognized the value of real-world evidence in regulatory decision-making. They have provided guidelines and frameworks for the use of RWD and real world evidence (RWE) in regulatory submissions, drug approvals, and post-market surveillance, further driving the demand for real-world data.
The advancement of data analytics tools and technologies, such as artificial intelligence (AI), machine learning (ML), and big data analytics, has facilitated the extraction of meaningful insights from large and complex real-world datasets. These technologies enable the analysis of RWD to identify treatment patterns, patient subgroups, adverse events, and other clinically relevant information.
Real-world data can be leveraged to optimize the drug development process and enhance clinical trial design. RWD can be used to identify target populations, understand disease progression, assess treatment effectiveness, and identify potential safety concerns. By integrating real-world evidence into clinical trials, researchers can enhance trial efficiency, recruit appropriate patients, and generate robust outcomes.
For instance, in June 2022, AbbVie Inc., a pharmaceutical company, announced the launch of AbbVie Research Collaborative, a health data platform that enables adults anywhere in the U.S. to partner with AbbVie’s world-class scientific researchers. Collaborative participants contribute their real-world health data, such as genetic, lifestyle, and exercise information, medical records, and more, to provide a more comprehensive picture of their health. Real-world health data empowers researchers to find health and disease patterns they may not see within a clinical trials.
Real-world data enables comparative effectiveness research, which focuses on comparing the benefits and risks of different treatment options in real-world settings. RWD allows for the assessment of treatment effectiveness, safety, and patient outcomes across diverse patient populations, thereby providing valuable insights for healthcare decision-making.
For instance, in August 2020, the Medical Device Innovation Consortium (MDIC) announced the launch of its real world evidence (RWE) Framework for regulatory decision-making for in vitro diagnostics.
There is a growing trend of integrating multiple data sources to gain a comprehensive view of patient health and outcomes. This includes combining electronic health records (EHRs), claims data, patient registries, wearable devices, social media data, and genomic information. Integrating diverse data sources allows for a more holistic analysis and provides richer insights into real-world patient experiences and outcomes.
The global real world data (RWD) market is witnessing rapid advancements in data analytics tools and techniques, including artificial intelligence (AI) and machine learning (ML). These technologies enable more sophisticated analysis of large and complex datasets, uncovering patterns, trends, and correlations that may not be apparent with traditional statistical approaches. AI and ML algorithms can assist in data integration, predictive modeling, risk stratification, and decision support.
For instance, in August 2021, Syneos Health, a pharmaceutical company, partnered with Aetion, a health care technology company to provide a real-world evidence solution and offer analytical-driven data and regulatory-grade data.
*Definition: Real-world data (RWD) refers to data collected from a variety of sources that reflect a patient's health status and/or the delivery of health care in real-world settings. Real-world data is gathered from various sources such as electronic health records (EHRs), claims and billing activities, product and disease registries, patient-related activities in outpatient or inpatient settings, health monitoring devices, and other sources, in contrast to data from traditional clinical trials, which are collected under controlled conditions. Using this data, real-world evidence (RWE) regarding a product's use and any advantages or disadvantages is produced for use in healthcare analysis and decision-making.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients