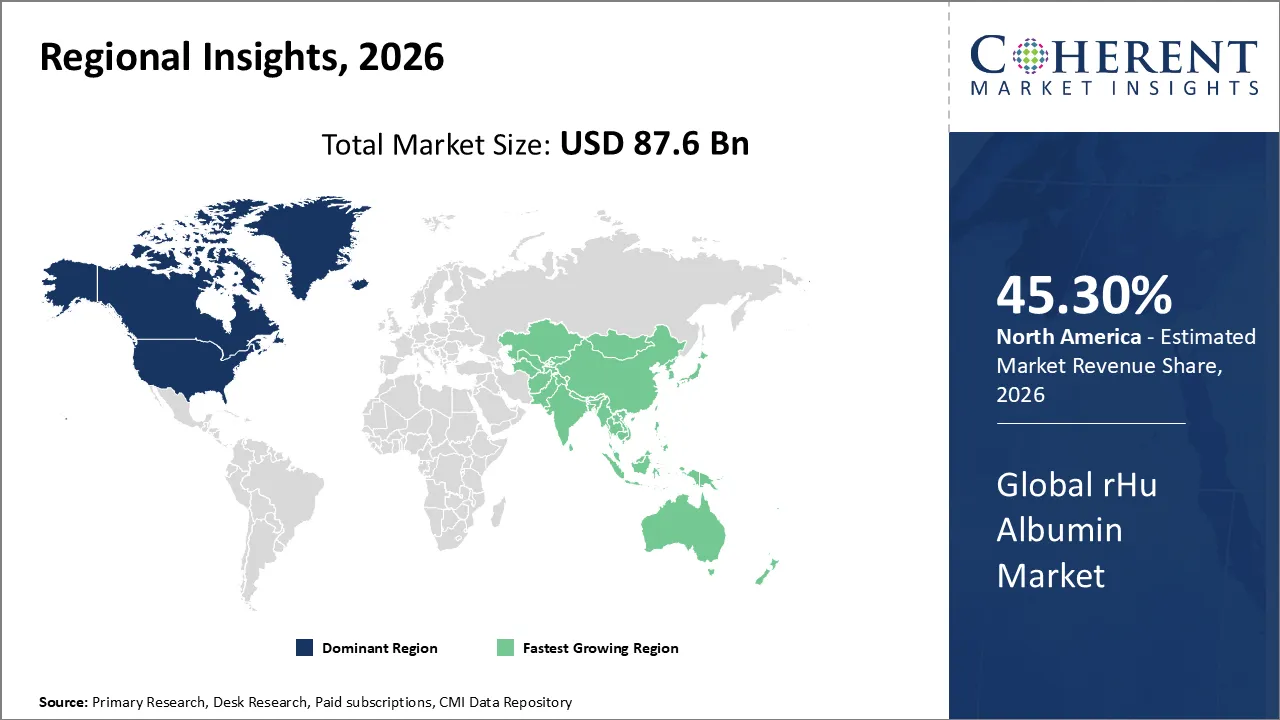
The rHu Albumin Market is estimated to be valued at USD 87.6 Bn in 2026 and is expected to reach USD 277.8 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 13.6% from 2026 to 2033.
Pharmaceutical, biotechnology, and research industries are driving the expansion of the recombinant human albumin (rHu albumin) market. Companies use rHu albumin in drug formulation, vaccine production, cell culture media, and regenerative medicine due to its high purity, safety, and consistent quality compared to plasma-derived albumin. The growth of biologics manufacturing, increasing stem cell research, and the adoption of animal-free cell culture systems further support market development. In addition, advancements in recombinant protein technologies and rising investments in biopharmaceutical research continue to boost global demand for rHu albumin.
|
Current Events |
Description and its impact |
|
Macro-Level Technological Advancements in Recombinant Protein Production |
|
|
Regional Regulatory Changes in Key Pharmaceutical Markets |
|
|
Clinical Demand and Epidemiological Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Pharmaceutical and Biotechnological Industry acquired the prominent market share of 52.7% in 2026. Pharmaceutical and biotechnology companies are fueling the growth of the recombinant human albumin (rHu albumin) market by using it in drug formulations, biologics, vaccines, and cell culture systems. They depend on rHu albumin for its high purity, consistent quality, and safety over plasma-derived alternatives. Expanding biologics production, advancing research in novel therapies, and adopting animal-free manufacturing are driving higher demand. As companies focus on reliable and reproducible materials for development and production, they increasingly integrate recombinant albumin into their processes.
Oryza Sativa hold the largest market share of 42.2% in 2026. Biotechnology and pharmaceutical companies are increasingly adopting Oryza sativa–based expression systems to produce recombinant human albumin because they provide high-purity, animal-free products suitable for biopharmaceutical applications. Rice-derived platforms enable scalable production and help reduce contamination risks associated with plasma-derived sources. Manufacturers use this system in cell culture media, vaccine development, and drug formulation. Growing demand for reliable raw materials in biologics production and ongoing advances in plant-based recombinant technology continue to support the use of Oryza sativa–derived rHu albumin.
Stem Cell Therapy expected to hold largest market share of 38.8% in 2026. Researchers and biotechnology companies are driving demand in the recombinant human albumin (rHu albumin) market by expanding stem cell therapy applications. They use rHu albumin to maintain stable culture conditions, enhance cell viability, protect cells from oxidative stress, and improve culture efficiency in laboratory and clinical environments. The growing emphasis on regenerative medicine and personalized treatments is increasing the need for dependable culture materials. As organizations intensify stem cell research and therapeutic development, they continue to increase the use of recombinant human albumin. For instance, in March 2026, the International Society for Stem Cell Research (ISSCR) launched Stem Cell Medicine: Parkinson's Disease, a new course that trains clinicians on evidence-based stem cell therapies for Parkinson’s disease (PD).
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 45.30% in 2026. Biotechnology and pharmaceutical companies in North America are driving the recombinant human albumin (rHu albumin) market through increased use in biologics, vaccine production, cell culture, and regenerative medicine, valuing its safety and consistent quality. Rising investments in stem cell research, advanced therapies, and animal-free manufacturing practices are accelerating adoption. Leading biopharmaceutical firms, advanced healthcare infrastructure, and strong R&D initiatives further promote innovation and sustain steady demand for recombinant albumin throughout the region.
Pharmaceutical and biotechnology companies across Asia Pacific are driving the growth of the recombinant human albumin (rHu albumin) market by using it in biologics, vaccines, and cell culture, appreciating its high purity and safety. Increasing investments in regenerative medicine, stem cell research, and animal-free manufacturing are fueling demand. Rapid industrialization, expanding healthcare infrastructure, and supportive government policies are promoting local production and research, encouraging innovation and broader adoption of recombinant albumin throughout the region’s developing biopharmaceutical sector.
Pharmaceutical and biotechnology companies in the United States are expanding the recombinant human albumin (rHu albumin) market by incorporating it into drug formulations, biologics, vaccines, and cell culture applications. Researchers increasingly use rHu albumin for its consistent quality, safety, and animal-free production. Growing regenerative medicine, stem cell research, and advanced therapy initiatives are accelerating adoption. Strong healthcare infrastructure, significant R&D investments, and the presence of leading biopharmaceutical firms further drive innovation and maintain steady demand for recombinant albumin throughout the U.S.
Pharmaceutical and biotechnology companies in Japan are driving the growth of the recombinant human albumin (rHu albumin) market by using it in biologics, vaccines, and cell culture applications. Researchers rely on rHu albumin for its high purity, safety, and animal-free production, ensuring consistent laboratory and clinical results. Expanding regenerative medicine, stem cell research, and advanced therapy development are boosting demand. Strong healthcare infrastructure, government support for biotech innovation, and active R&D initiatives further promote local production and broader adoption of recombinant albumin in Japan.
In April 2024, Partner Therapeutics, Inc. (PTx) announced that its partner Nobelpharma received approval from the Japanese Pharmaceuticals and Medical Device Agency for the inhaled use of Leukine (sargramostim), marketed in Japan as Sargmalin, for the treatment of aPAP. Leukine is a glycosylated recombinant human granulocyte-macrophage colony-stimulating factor produced using recombinant DNA technology in yeast.
The industry’s shift toward animal-free and serum-free cell culture systems creates opportunities for recombinant albumin adoption. Biopharmaceutical and research organizations prefer rHu albumin over plasma-derived alternatives to minimize contamination risks and ensure reproducibility. Companies can capitalize by offering products compatible with serum-free media, providing safety certifications, and catering to regulatory requirements, thereby meeting evolving customer preferences in vaccines, biologics, and cell therapy manufacturing.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 87.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.6% | 2033 Value Projection: | USD 277.8 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck KGaA, HiMedia Laboratories Pvt. Ltd., Albumin Bioscience, Albumedix, ScienCell Research Laboratories, Inc., InVitria, Lazuline Biotech Private Limited, Lee Biosolutions, Inc., Abcam Plc, and Shandong Kingtone Biotechnology Co.,Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients