The global sevoflurane market is estimated to be valued at USD 420.3 Mn in 2026 and is expected to exhibit a CAGR of 4.8% during the forecast period (2026-2033). Sevoflurane is a type of anesthetic that belongs to the general anesthetics class of drugs. Before and during surgery, sevoflurane is used to induce general anesthesia (lack of consciousness). Sevoflurane is to be administered via inhalation. Although, sevoflurane can be used solely, it is frequently combined with other anesthetics. In some patients, this aids in the production of more effective anesthetic.
During surgery, sevoflurane also helps to keep the patient's blood pressure and pulse rate stable. Sevoflurane can also be used to help manage discomfort following surgery.
Many countries such as Thailand, Indonesia, and Singapore are facing problems with regards to transportation and distribution of healthcare products.
These medications allow for precise and rapid management of sedation levels, and drug administration does not necessitate the use of electronic infusion pumps, which are in short supply.
Figure 1. Global Sevoflurane Market Value (USD MN), by Region, 2026

To learn more about this report, Request Free Sample
The increasing number of surgeries is the major factor that is expected to drive the growth of global sevoflurane market over the forecast period.
The emerging number of surgeries is expected to drive the growth of the global sevoflurane market. For instance, according to European Statistical System, 2021, per 100,000 inhabitants, 1094.3 cataract surgeries, 259.6 transluminal coronay angioplasty, 232.5 cholecystectomay, 219.5 inguinal hernia repair surgeries were performed in Belgium in 2018.
Prevalence of Chronic Disorders
Increasing number of chronic disorders like cancer, respiratory diseases, etc. is expected to aid in the growth of the global sevoflurane market over the forecast period. For instance, according to GLOBOCAN 2020, an estimated 19.3 million new cancer cases (18.1 million excluding non-melanoma skin cancer) were diagnosed worldwide, with around 10.0 million cancer deaths (9.9 million excluding non-melanoma skin cancer) in 2020. With an anticipated 2.3 million new cases 11.7% were of female breast cancer followed by lung cancer (11.4 %), colorectal cancer (10.0%), prostate cancer (7.3 %), and stomach cancer (5.6 %) globally.
Sevoflurane Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 420.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 583.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Baxter, Piramal Pharma Solutions, Manus Aktteva Biopharma LLP, Maruishi Pharmaceutical Co.,Ltd., Lunan Pharmaceutical Group, Abbvie, Wellona Pharma, Rewine Pharmaceutical, and Troikaa Pharmaceuticals Limited |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Sevoflurane Market – Restraint
Side effects associated with sevoflurane are expected to hamper the growth of global sevoflurane market over forecast period. Side effects include-
Global Sevoflurane Market – Regional Analysis
On the basis of region, the global sevoflurane market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa.
North America is expected to hold dominant position in the global sevoflurane market over the forecast period, owing to increasing standards of surgical care in the U.S. For instance, in July 2019, at the ACS Quality and Safety Conference, the American College of Surgeons (ACS) announced the introduction of its new Geriatric Surgery Verification (GSV) Program. This new surgical quality improvement initiative incorporates 30 new surgical criteria aimed at systematically improving surgical treatment and outcomes in the elderly.
Moreover, due to increasing number of surgeries, Asia Pacific is expected to witness a significant growth in the global sevoflurane market over the forecast period. For instance, according to the World Journal of Surgery, 2021, annually, 3646 procedures were performed to address the surgical needs of the Indian population, compared to 5000 surgeries per 100,000 people globally. Caesarean section, cataract surgery, fracture and hernia surgeries were the most common surgical procedures.
Figure 2. Global Sevoflurane Market Share, By Application, 2026
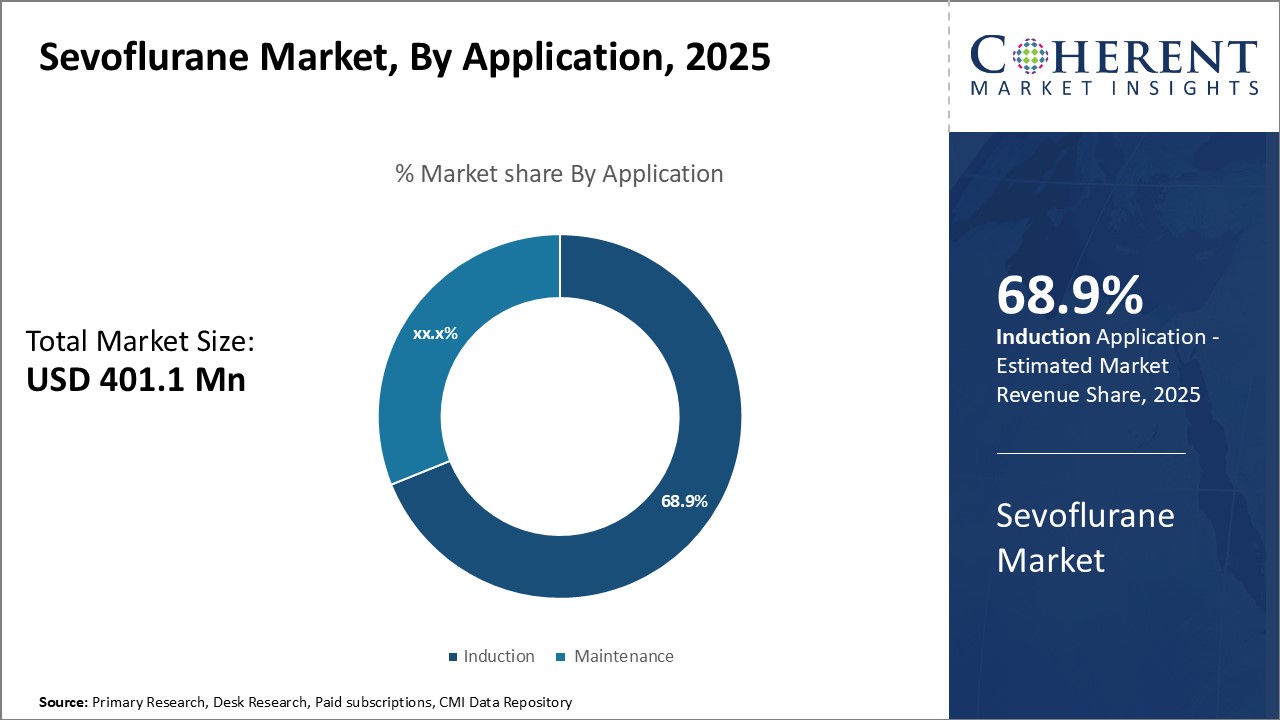
To learn more about this report, Request Free Sample
Recent Developments
In January 2022, Dechra, a company that develops and markets veterinary products, acquired the veterinary-approved inhalant anesthetics isoflurane and sevoflurane from Halocarbon, LLC, a leader in high-purity fluorochemistry for the pharmaceutical, biotechnology, personal care, and agricultural applications.
Global Sevoflurane Market – Competitive Landscape
Major players operating in the global sevoflurane market include Abbott Laboratories, Baxter, Piramal Pharma Solutions, Manus Aktteva Biopharma LLP, Maruishi Pharmaceutical Co.,Ltd., Lunan Pharmaceutical Group, Abbvie, Wellona Pharma, Rewine Pharmaceutical, and Troikaa Pharmaceuticals Limited.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients