Subcutaneous Drug Delivery Market is estimated to be valued at USD 15.70 Bn in 2026 and is expected to reach USD 30.41 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 9.9% from 2026 to 2033.
Analysts’ Views on Global Subcutaneous Drug Delivery Market:
Increasing convenience and safety for both patients and medical professionals offered by subcutaneous devices are major factors leading to the growth of the global subcutaneous drug delivery market. For instance, According to the National Health Institute (NIH), 2020, around 2 million healthcare professionals suffered from infectious diseases as a result of needlestick injuries. The retractable nature of prefilled syringes helps to overcome this problem, thereby acting as an ideal device to reduce the occurrence of needlestick injuries.
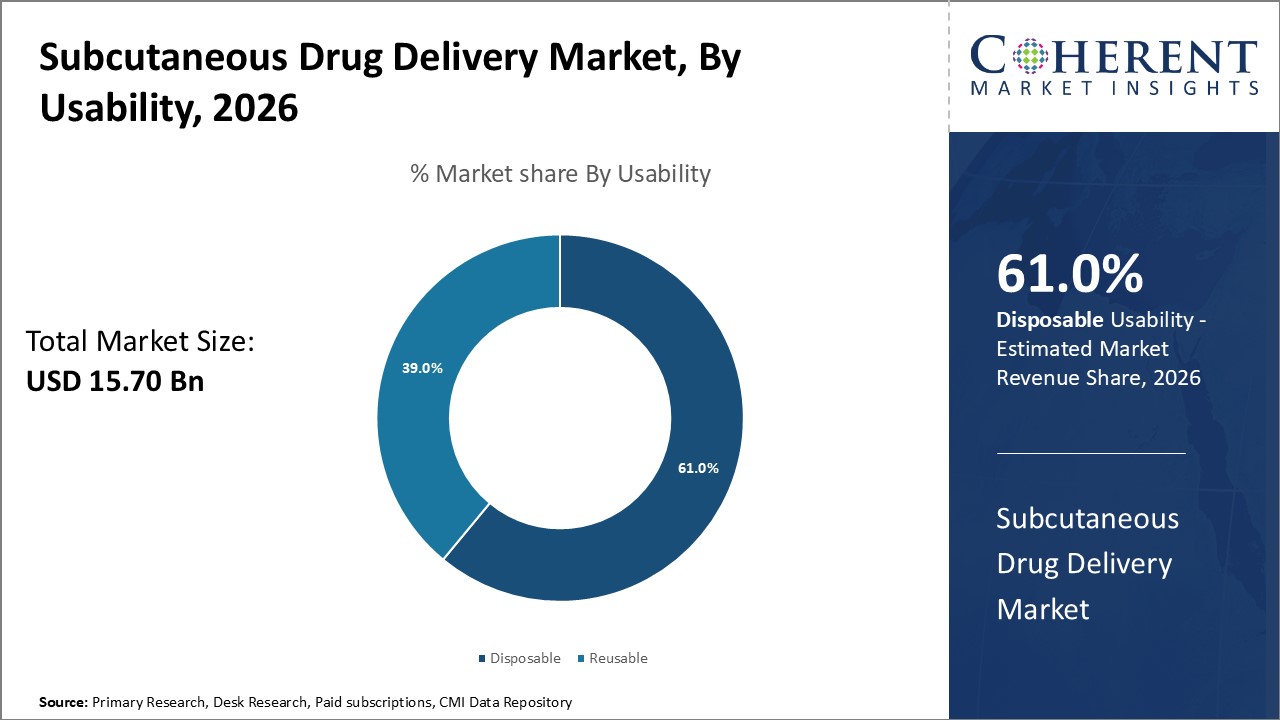
Figure 1. Global Subcutaneous Drug Delivery Market Share (%), By Usability, 2026
To learn more about this report, Request Free Sample
Global Subcutaneous Drug Delivery Market – Drivers
Adoption of inorganic growth strategies by key players
Increasing focus of manufacturers on the adoption of inorganic growth strategies such as collaboration and partnership, in order to strengthen their product portfolio of subcutaneous drug delivery systems is expected to drive the market growth over the forecast period. For instance, in March 2021, the collaboration between Eitan Medical device company, provides drug delivery devices and medical pumps and Stevanato Group, a glass container manufacturing company launched a wearable injection device for the simple and efficient subcutaneous administration of large-volume and high-viscosity medications that provide the interests of both patients and pharmaceutical companies.
Subcutaneous Drug Delivery Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 15.70 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.9% | 2033 Value Projection: | USD 30.41 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton Dickinson and Company., Novo Nordisk A/S, Johnson & Johnson, Novartis International AG, Amgen Inc., Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche AG., Consort Medical plc LaVision Biotec GmbH, Eli Lilly and Company., Merck & Co., Inc, Insulet Corporation., West Pharmaceutical Services, Inc. Gerresheimer AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing focus on preventing the needlestick injuries
Needlestick injuries with injection needles and lancing devices are a key concern of patients with diabetes. Insulin syringe needlestick injuries is a percutaneous wound injury caused by needle tips and can occur while using, handling, disassembling, or disposing of needles and syringes. Increasing awareness regarding safety and concern about needlestick injuries are major factors driving the growth of the subcutaneous drug delivery market.
Preference for safety injection devices is increasing among healthcare professionals and manufacturers, as these devices decrease the chances of needlestick injuries. For instance, the WHO recommended the use of safety syringes with sharp injury protection (SIP) in order to prevent needlestick injuries. Moreover, in May 2021, Innovative Neurons LLC, is a medical device company has announced its plan to commercialize its patented auto retractable multi-needle syringe known as FAST (fast, accessible, safe, and technology), to decrease needlestick injuries.
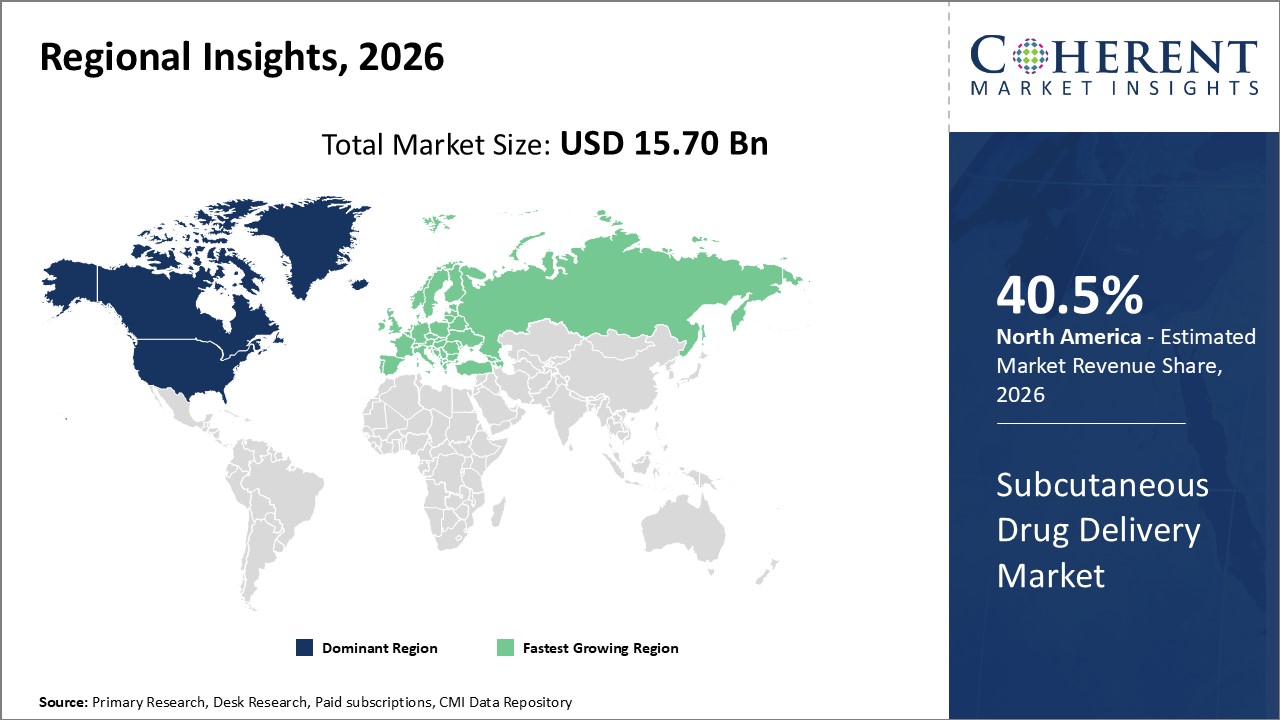
Figure 2. Global Subcutaneous Drug Delivery Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Subcutaneous Drug Delivery Market - Regional Analysis
Increasing product launches by market players is expected to drive the market growth. North America is expected to hold a dominant position in the market, owing to the large healthcare industry in the region. Market players are focused on launching new products in the region. According to the WHO (World Health Organization), over 16 billion injections are administered annually in transitional and developing countries the world over. For instance, in June 2021, Hindustan Syringes & Medical Devices company, launch single-use dispojekt safety needle to prevent injuries.
Supply chain and manufacturing activities in India, China, and the U.S.
According to an article published in the Center for Biosimilars (American Journal of Managed Care), on February 27, 2020, manufacturers such as Novartis AG who have manufacturing facilities in Austria, Europe, are particularly at risk on the supply side.
Global Subcutaneous Drug Delivery Market - Segmentation
Global Subcutaneous Drug Delivery market is segmented into Usability, Disease Indication, By Delivery System, By Distribution Channel, and By Region.
Based on Usability, the market is segmented into disposable and reusable. Out of which, the reusable segment dominated the global subcutaneous drug delivery market during the forecast period. For instance, in May 2026, Jabil Inc., a medical device company, announced the launch of the Qfinity Autoinjector Platform. It is a reusable drug delivery system with wide application.
Based on Disease Indication, the market is segmented into auto-immune disorders, neurological disorders, oncology disorders, hormonal disorders, allergy & infectious disorders, ophthalmic disorders, and others. Out of which, the auto-immune disorder segment dominated the global subcutaneous drug delivery market. For instance, in November 2020, according to the autoimmune registry inc (ari), between 15 and 30 million people in the U.S. suffer from autoimmune disease. It mostly affects women between the ages of 20 and 40, making it the largest class of illnesses in the country.
Based on Delivery System, the market is segmented into prefilled syringes, auto injector, pen injectors, disposable, and others. Out of the which pen injector segment dominated to the global subcutaneous drug delivery market, For instance, on February 2, 2023, according to AstraZeneca ,British-Swedish multinational pharmaceutical and biotechnology company, has announced received approval from U.S FDA for Tezspire (tezepelumab) self-administration in a prefilled, single-use pen for patients 12 years of age and older for asthma.
Based on Distribution Channel, the market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies. Out of which, hospital pharmacies segment is expected to dominate the market over the forecast period.
Based on Region, the global subcutaneous drug delivery market is segmented into North America, Latin America, Europe, Middle East, Asia Pacific, and Africa. Out of which, North America is expected to dominate the global subcutaneous drug delivery market over the forecast period, owing to increasing usage of auto injector and prefilled syringes in the market.
Global Subcutaneous Drug Delivery Market Cross Sectional Analysis:
Due to the increasing number of products being launched for epilepsy, the auto injector holds a dominant position in the delivery system segment in North America. For instance, in August 2022, according rafa laboratories, a pharmaceutical company, announced received approval from the FDA to launch a 10-mg midazolam autoinjector for the treatment of status epilepticus in adults.
Global Subcutaneous Drug Delivery Market: Key Developments
On January 13, 2023, MTD Group announced the FDA clearance obtained by its affiliate Pikdare S.p.A. for DropSafe Sicura passive safety needle with a completely passive mechanism that can be used to inject vaccines and other drugs into the muscle and under the skin.
In October 2022, scPharmaceuticals, a pharmaceutical company, announced the FDA approval for Furoscix (furosemide injection), the first and only self-administered, subcutaneous loop diuretic for the at-home treatment of congestion in chronic heart failure.
Global Subcutaneous Drug Delivery Market- Key Trends
Increasing the inorganic strategy of development and collaboration by major key players to boost the subcutaniouse drug dilevery market.
In November 2022, Kindeva Drug Delivery, Its capabilities include formulation, product development, scale-up manufacturing, and commercial manufacturing company is merging with ex-Pfizer subsidiary Meridian Medical Technologies to create a globetrotting drug-device contract development and manufacturing organization.
Global Subcutaneous Drug Delivery Market: Restraint
Increasing the number of product recalls from FDA
One factor impeding the growth of the worldwide subcutaneous medication delivery market is the lack of approved goods due to an increase in product recalls by different companies. Due to an increase in consumer complaints, the product has been discovered to be discoloured. For instance, on January 9, 2023, Spectrum Laboratory Products, Inc. a chemical manufacturing company, announced U.S.FDA gives alerts recall for EpiPen JR Auto-Injector, a bulk active epinephrine pharmaceutical ingredient (API) used to manufacture or compound prescription products.
Global Subcutaneous Drug Delivery Market: Key Players
Major market players include Becton Dickinson and Company., Novo Nordisk A/S, Johnson & Johnson, Novartis International AG, Amgen Inc., Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche AG., Consort Medical plc, LaVision Biotec GmbH, Eli Lilly and Company, Merck & Co., Inc, Insulet Corporation., West Pharmaceutical Services, Inc. Gerresheimer AG.
*Definition: A subcutaneous injection is a method of administering medication. Subcutaneous is the Latin word for skin. The tissue layer between the skin and the muscle is the target of this kind of injection, which uses a short needle. When administered this way, medication is typically absorbed over a longer length of time than when it is injected into a vein, sometimes up to 24 hours. When other administration techniques may not be as efficient, this kind of injection is performed. For instance, certain drugs cannot be administered orally because the stomach's acid and enzymes would destroy them.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients