Urea cycle disorders (UCDs) are a group of inherited defects of six enzymes and two transporters that constitute the urea cycle in the periportal liver cells. The age of onset symptoms vary between the different types of UCDs. UCDs often occur during the period of infancy due to triggered postnatal catabolism. The diagnosis of urea cycle disorder is done by the analysis of urine and blood to check for abnormal metabolites and high ammonia levels. Frequent blood tests are done to monitor ammonia levels in urea cycle disorder patients. The treatment of urea cycle disorders consists of dietary management to limit ammonia production. A liver transplant can be an effective treatment for urea cycle disorder for the proper functioning of urea cycle.
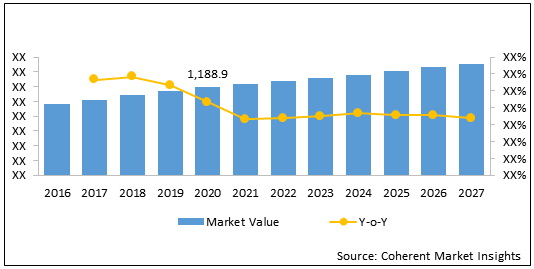
The global urea cycle disorders treatment market is estimated to be valued at US$ 1,188.9 million in 2020 and is expected to exhibit a CAGR of 3.5 % during the forecast period (2020-2027).
Figure 1. Global Urea Cycle Disorders Treatment Market Value (US$ Mn), 2016-2027
To learn more about this report, Request Free Sample
The increasing prevalence of urea cycle disorder is expected to drive growth of the global urea cycle disorders treatment market over the forecast period. For instance, according to the article published in the Orphanet Journal of Rare Diseases in June 2017, in 2017 the estimated prevalence of ornithine transcarbamylase deficiency ranges between 1/56,500 to 1/113,000 live births across the world.
Moreover, the increasing initiatives by key players operating in the market for supporting patients suffering from UCD is expected to fuel growth of the global urea cycle disorders treatment market in the near future.
For instance, in March 2017, Horizon Pharma plc. launched UCD in Common, an initiative for people suffering from urea cycle disorder, in order to offer supportive, educational, and interactive resources to the patients living with UCD, their families, caregivers, and healthcare professionals.
Urea Cycle Disorders Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2019 | Market Size in 2020: | US$ 1,188.9 Mn |
| Historical Data for: | 2016 to 2019 | Forecast Period: | 2020 to 2027 |
| Forecast Period 2020 to 2027 CAGR: | 3.5 % | 2027 Value Projection: | US$ 1,510.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bausch Health Companies, Inc., Recordati Rare Diseases Inc., Lucane Pharma SA, Acer Therapeutics, Ultragenyx Pharmaceutical Inc., Aeglea BioTherapeutics, Arcturus Therapeutics Holdings Inc., Orpharma Pty Ltd., Selecta Biosciences, Inc., Abbott Laboratories, NESTLÉ S.A., DANONE S.A., and Mead Johnson & Company, LLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Urea Cycle Disorders Treatment Market – Impact of Coronavirus (COVID-19)
Following the outbreak of COVID-19 in December 2019, the disease has spread to over 100 countries across the globe and the World Health Organization declared it as a public health emergency. According to the World Health Organization’s report, manifestation of coronavirus (COVID-19) has resulted in over 162 million infected individuals worldwide as of 17th May, 2021.
The coronavirus (COVID-19) pandemic and resulting lockdown in various countries across the globe has impacted the financial status of major companies in urea cycle disorders treatment market. The private healthcare sector is one of the sectors, which has been majorly impacted by the COVID-19 pandemic. The pandemic has negatively impacted the global urea cycle disorders treatment market in various aspects such as, research and development, production, and supply of medicines. The pandemic has also affected the growth of pharmaceutical businesses of various companies across the globe due to lockdowns implemented by governments of several countries.
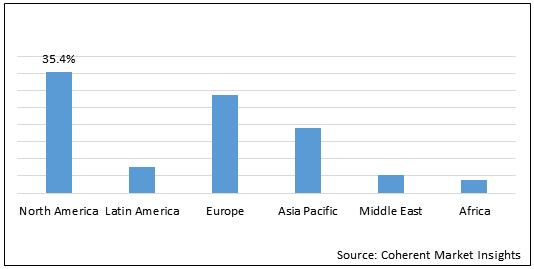
Figure 2. Global Urea Cycle Disorders Treatment Market Share (%), By Region, 2020
To learn more about this report, Request Free Sample
North America region is expected to hold dominant position in the global urea cycle disorders treatment market over the forecast period, owing to increasing research and development by academic institutes in gene therapy for the treatment of the urea cycle disorders.
For instance, in March 2019, University of Calgary Cumming School of Medicine (CSM) and Alberta Health Services (AHS) collaboratively initiated the clinical trial study on treatment of urea cycle disorders with the help of gene therapy. The clinical trial study was conducted at Clinical Trials Unit (CTU) at the Foothills Medical Centre (FMC), a hospital in Canada.
Moreover, due to long-term therapy, patients may suffer from some complications, which include fussiness, sleepiness or sluggishness, vomiting, low body temperature, problems with posture, seizures, and others. Thus, such complications of long-term therapy is expected to hinder the market growth over the forecast period.
Key Players
Major players operating in the global urea cycle disorders treatment market include Bausch Health Companies, Inc., Recordati Rare Diseases Inc., Lucane Pharma SA, Acer Therapeutics, Ultragenyx Pharmaceutical Inc., Aeglea BioTherapeutics, Arcturus Therapeutics Holdings Inc., Orpharma Pty Ltd., Selecta Biosciences, Inc., Abbott Laboratories, NESTLÉ S.A., DANONE S.A., and Mead Johnson & Company, LLC
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients