U.S. Implantable Medical Devices Market is estimated to be valued at USD 23.07 Bn in 2026 and is expected to reach USD 35.38 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.3% from 2026 to 2033.
The U.S. Implantable Medical Devices Market is witnessing robust growth, driven by the rising prevalence of chronic diseases, technological advancements, and an aging population. Implant types such as orthopaedic, cardiovascular, and neurostimulators are in high demand due to increasing surgical procedures and improved clinical outcomes. Orthopaedic implants lead the segment, fuelled by joint replacement and trauma-related surgeries.
|
Current Event |
Description and its impact |
|
Resurgence of Elective Procedures in Post-Pandemic U.S. Healthcare |
|
|
FDA Regulatory Support for Advanced and Digital Implants |
|
|
Surge in Strategic M&A and Partnerships Among U.S. Medtech Firms |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The industry is witnessing significant growth driven by the rising occurrence of various disorders in cardiovascular, ophthalmology, orthopaedic, plastic surgery, dental, and neurology fields. Among these, heart diseases, including cardiovascular disease (CVD), congenital heart defects (CHDs), and stroke, emerge as the primary cause of global mortalities.
According to the World Health Organization (WHO), approximately 17.9 million deaths occur each year due to these heart conditions, accounting for around 31% of all global fatalities. Notably, CHD affects around 7.4 million individuals, while stroke claims the lives of approximately 6.7 million people annually.
Projections suggest that by 2030, CVDs will be responsible for an estimated 23.6 million deaths worldwide. Unhealthy dietary habits, excessive alcohol and tobacco consumption, and sedentary lifestyles are major contributing factors to the increasing incidence and prevalence of heart diseases. With the continued upward trend in these conditions, there exists a significant market opportunity for future growth.
In August 2024, Reuters reported that Zimmer Biomet exceeded profit expectations in Q2 as demand for hip and knee implants surged. The company also acquired AI-driven guidance firm OrthoGrid Systems to enhance surgical precision.
Manufacturers are focused on technological advances in implantable devices by adopting nanotechnology as the future of their products which expected to drive the growth of the U.S. Implantable medical devices market in near future. The advancements in this field will greatly improve the effectiveness of implantable devices.
By successfully launching technologically advanced products early, manufacturers can secure a significant market share and become leaders in the market. For instance, in December 2025, Abbott, a multinational medical devices and health care company, announced the U.S. Food and Drug Administration (FDA) approval of the company's Eterna spinal cord stimulation (SCS) system– the smallest implantable device, rechargeable spinal cord stimulator currently available on the market for the treatment of chronic pain.
Eterna SCS utilizes Abbott's proprietary low-dose BurstDR stimulation, the only SCS waveform technology with the highest level of clinical evidence (1A evidence), proven to reduce pain 23% more than traditional waveform technology approaches. In May 2025, Reuters reported that the FDA granted “breakthrough device” designation to Neuralink’s brain-computer interface implant, aimed at restoring speech for individuals with paralysis—a regulatory milestone likely to accelerate development and potential approval.
The hospitals segment is expected to capture a dominant 60.8% share of the U.S. implantable medical devices market in 2026, owing to their central role in conducting complex surgical implant procedures. Equipped with specialized infrastructure, advanced diagnostics imaging services, and highly trained surgical teams, hospitals remain the preferred setting for orthopaedic, cardiovascular, and neurological implantations.
Additionally, hospitals benefit from favourable reimbursement frameworks and increasing adoption of robotic-assisted surgeries, which enhance procedural precision and patient outcomes. The rise in chronic conditions and age-related degenerative diseases is further boosting surgical volumes in hospital settings, solidifying their leading position in the implantable devices ecosystem.
To learn more about this report, Request Free Sample
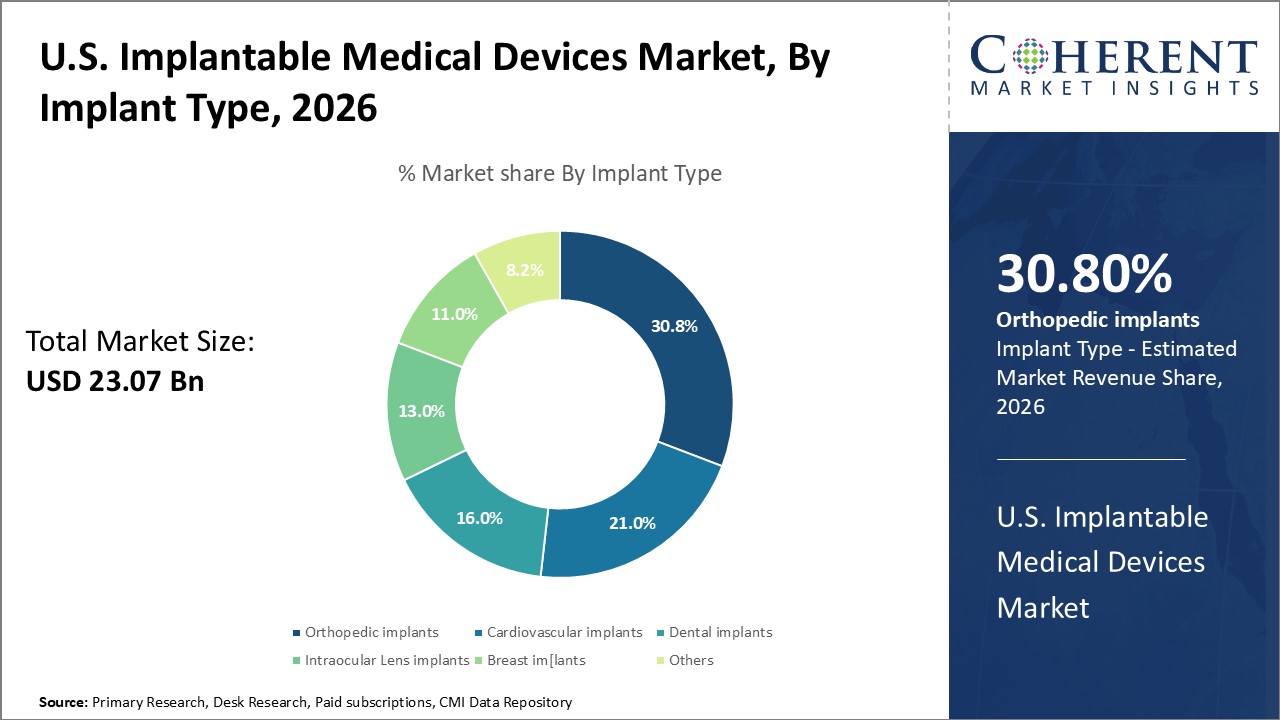
Orthopaedic implants are projected to account for a leading 30.8% share of the U.S. implantable medical devices market in 2026. This segment's growth is fuelled by the aging population, increasing incidence of osteoarthritis, and higher demand for joint reconstruction and spinal implants. Advancements in biomaterials, 3D printing, and minimally invasive surgical techniques are enhancing implant performance, durability, and patient satisfaction.
The rise in sports-related injuries and trauma cases has also contributed to the expanding orthopaedic implant market. With continuous innovation and growing patient preference for improved mobility solutions, orthopaedic implants are set to maintain a strong foothold in the overall implantable devices landscape.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 23.07 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.3% | 2033 Value Projection: | USD 35.38 Bn |
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Alcon Laboratories, Biomet Incorporated, Bausch and Lomb Incorporated, Boston Scientific Corporation, Johnson and Johnson, Medtronic Plc, St. Jude Medical Incorporated, Smith and Nephew PLC, Stryker Corporation, Synthes Incorporated, Zimmer Holdings Incorporated. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients